Boyles Law Relate to Charles Law Lab Introduction

Boyles Law Relate to Charles’ Law Lab Introduction Lab Exit Ticket

Graph of Charles’ Law Lab relating Temperature and Volume http: //www. grc. nasa. gov/WWW/K-12/airplane/glussac. html

2. Proportionality • Inversely proportional relationship means as one variable increase the other decreases. • Proportional relationship means that as one variable increases the other increases.

3. Does the graph of Charles’ Law show an inversely proportional or (directly) proportional relationship? • Proportional, Why? • As temperature increases, volume also increases • As temperature decreases, volume also decreases

4. According to the Kinetic Molecular Theory of Gases, the actual volume occupied by a gas is extremely small compared to the volume it occupies? • Real World: Yes, the actual gas molecule is extremely small as compared to the volume it occupies, but it does occupy a volume. • Calculation: Individual molecules are very small and negligible to the calculation.

Compressibility • This is a crowding effect of gases when the volume is decreased

Pressure • Pressure is not the same as force. • Pressure is a force over an area. • Example: psi = Pounds per in 2 http: //hypertextbook. com/phys ics/thermal/pressuretemperature/ Photograph by : Larry Mac. Dougal/Can. West News Service

Measuring Pressure • A barometer measures atmospheric pressure. www. aniscon. com/&h=300&w=315&sz=59&hl=en&start=17&um=1&tbnid=U 44 e. ZVI 6 n 9 D 3 M: &tbnh=111&tbnw=117&prev=/images%3 Fq%3 DBarometer%26 um%3 D 1%26 hl%3 Den%26 rlz%3 D 1 T 4 DKUS_en. US 213 US 214%26 sa%3 DN

Units of Pressure • • • Kilopascals (k. Pa), Atomospheres (atm), Millimeters of mercury (mm of Hg), • Torr (torr) • • • Helpful Conversions 1 atm = 760 mm Hg 1 atm = 760 torr 1 mm Hg = 1 torr 1 atm = 101. 325 k. Pa

Volume • • • Liters (L), Milliliters (m. L), Centimeters (cm 3) • Helpful conversions – 1000 m. L = 1 L – 1 m. L = 1 cm 3

Boyle’s Law: • Pressure and volume are inversely proportional • Formula: P 1 V 1 = P 2 V 2
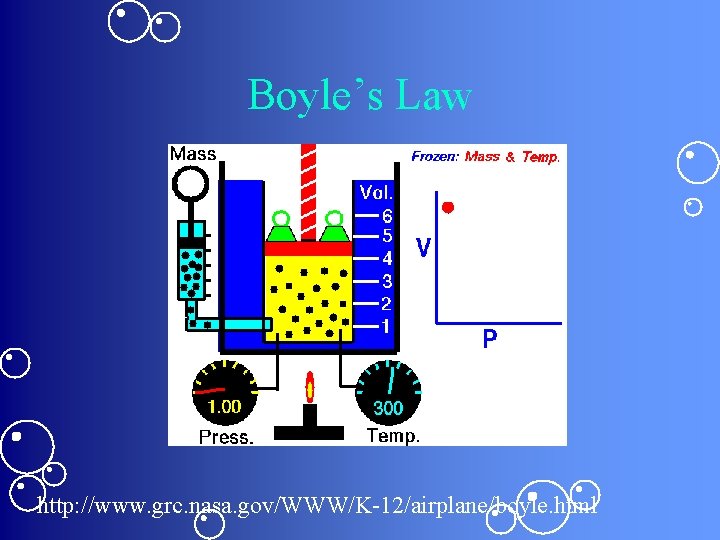
Boyle’s Law http: //www. grc. nasa. gov/WWW/K-12/airplane/boyle. html

Lab Overview 1. Pressure = Books of Uniform Mass 2. Find the Average Volume for Each Mass 3. Graph the Average Volume vs. Pressure 4. Exit Ticket

Lab Parts

Draw up 35 m. L (or cc) in syringe and cap it—notice we will read the measurement from the bottom of the plunger. What about sig figs?

Be sure to put the cap in the bottom. Some kits have black caps and some have orange caps.

Slide the other end of the syringe into the wooden platform.

The uniform masses are textbooks.

• You will add one book at a time. • Record the volume (correct sig figs) after each book is added. • When you have all 6 books on the apparatus, you will then remove one book at a time and record the volume after each book is removed.

Lab Safety • Goggles • Lab Apron • Common Sense!

QUESTIONS? ?
- Slides: 21