Boyles Law Pressure Volume BOYLE 1662 Temperature and

Boyle’s Law • Pressure • Volume

BOYLE - 1662 Temperature and Amount are constant Look at relationship between P and V What happenes when you increase the pressure on the syringe? As pressure increases, volume decreases
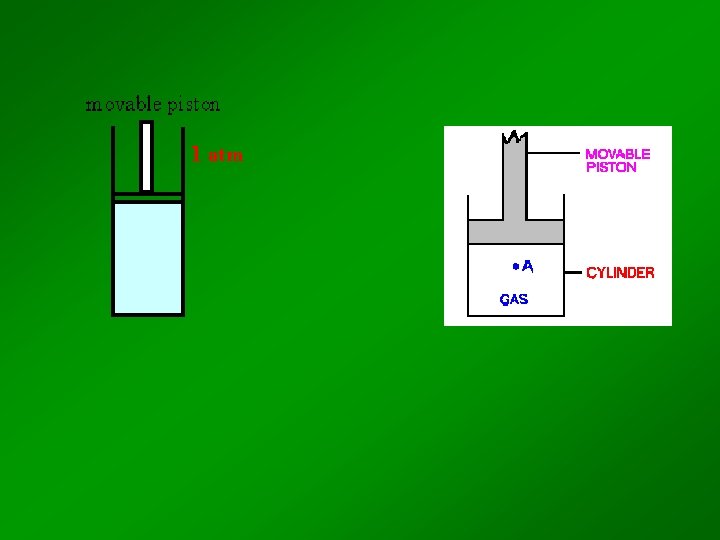
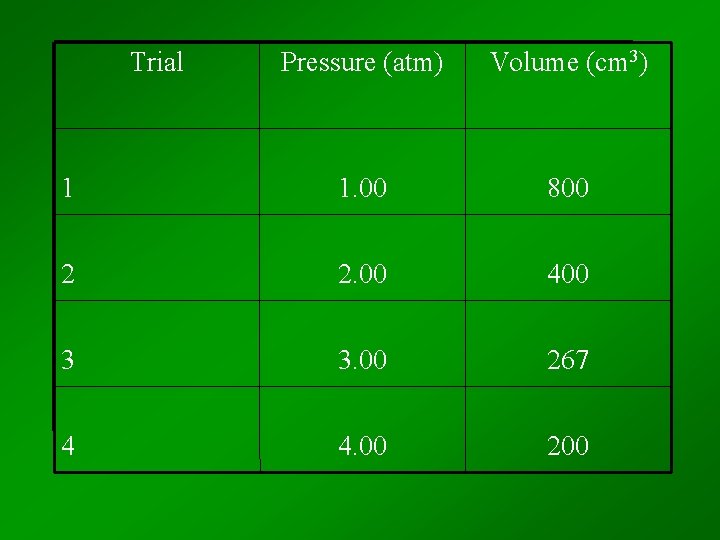
Trial Pressure (atm) Volume (cm 3) 1 1. 00 800 2 2. 00 400 3 3. 00 267 4 4. 00 200
Scientists plot data because a picture shows relationships better than lists of numbers. There are lots of “pictures” that scientists & mathematicians recognize.

Inverse Relationships • Graph of 2 variables is a hyperbola • Properties: – Relationship is inverse – Product of the 2 variables is a constant
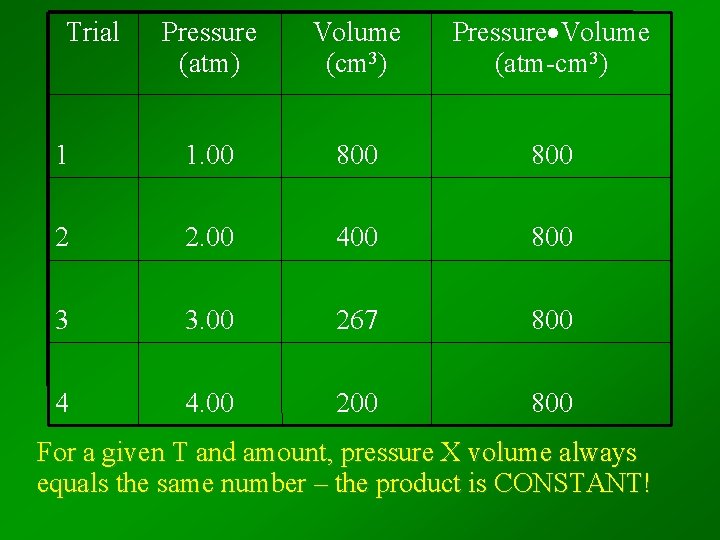
Trial Pressure (atm) Volume (cm 3) Pressure Volume (atm-cm 3) 1 1. 00 800 2 2. 00 400 800 3 3. 00 267 800 4 4. 00 200 800 For a given T and amount, pressure X volume always equals the same number – the product is CONSTANT!
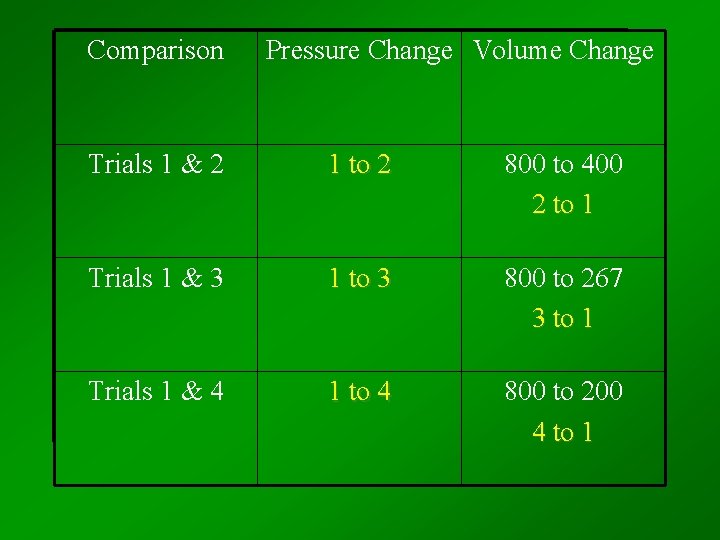
Comparison Pressure Change Volume Change Trials 1 & 2 1 to 2 800 to 400 2 to 1 Trials 1 & 3 1 to 3 800 to 267 3 to 1 Trials 1 & 4 1 to 4 800 to 200 4 to 1

Boyle’s Law - words • The volume of a sample of gas is inversely proportional to its pressure, at constant temperature
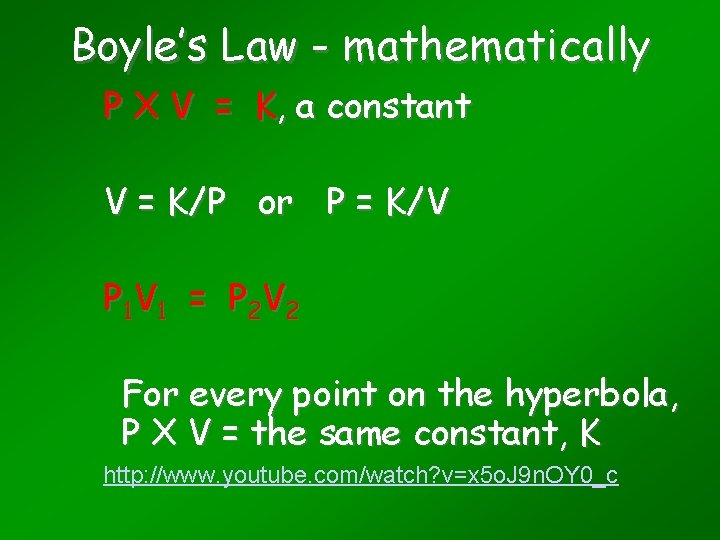
Boyle’s Law - mathematically P X V = K, a constant V = K/P or P = K/V P 1 V 1 = P 2 V 2 For every point on the hyperbola, P X V = the same constant, K http: //www. youtube. com/watch? v=x 5 o. J 9 n. OY 0_c
- Slides: 10