Boyles Law P V P V inversely proportional
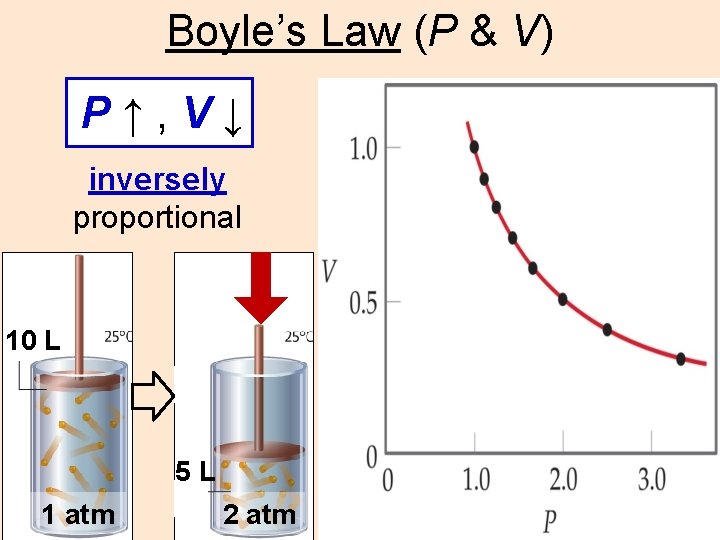
Boyle’s Law (P & V) P↑, V↓ inversely proportional 10 L 5 L 1 atm 2 atm
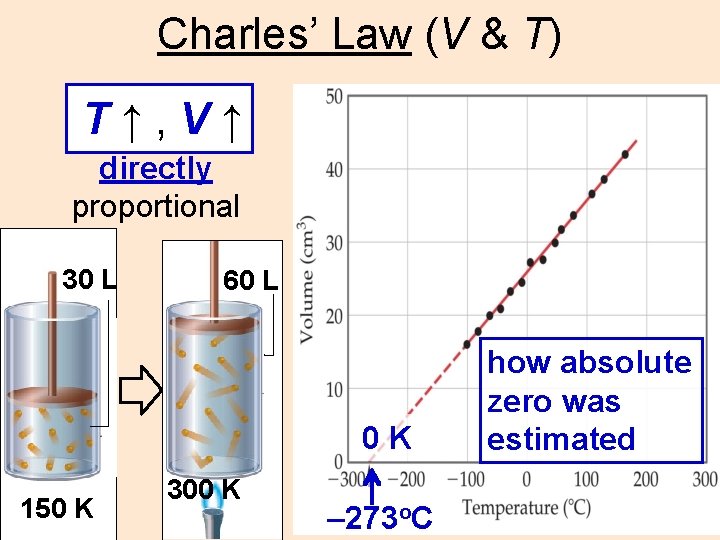
Charles’ Law (V & T) T↑, V↑ directly proportional 30 L 60 L 0 K 150 K 300 K – 273 o. C how absolute zero was estimated
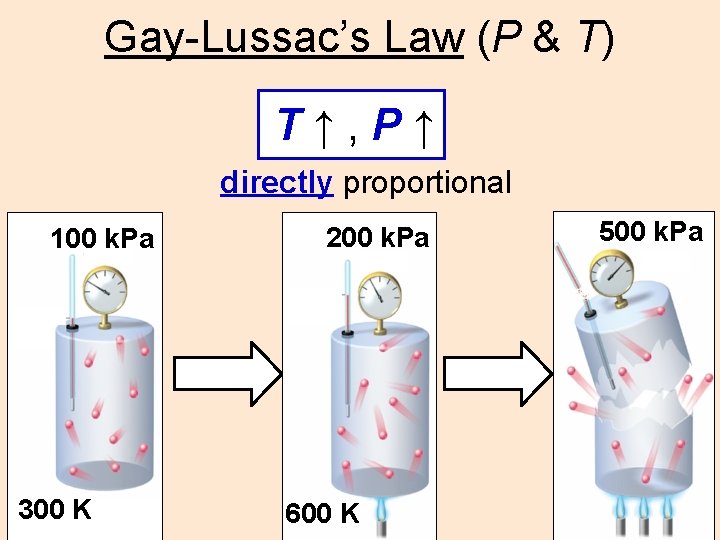
Gay-Lussac’s Law (P & T) T↑, P↑ directly proportional 100 k. Pa 300 K 200 k. Pa 600 K 500 k. Pa
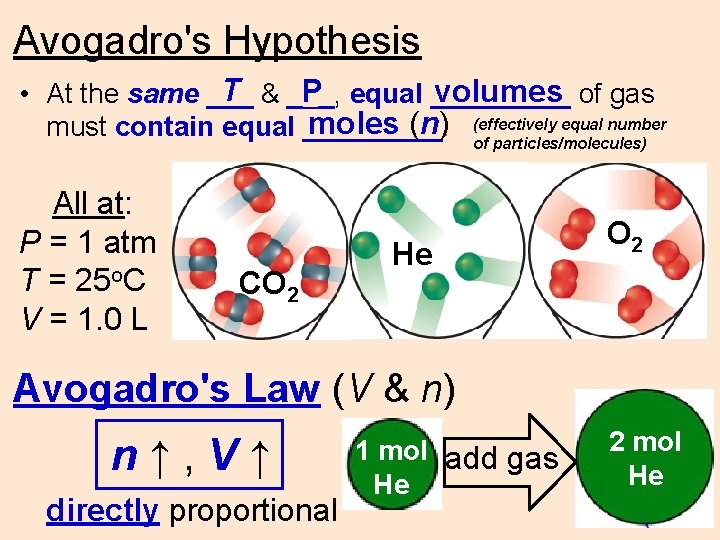
Avogadro's Hypothesis T & ___, volumes of gas P equal _____ • At the same ___ equal number moles (n) (effectively must contain equal _____. of particles/molecules) All at: P = 1 atm T = 25 o. C V = 1. 0 L CO 2 He O 2 Avogadro's Law (V & n) n↑, V↑ directly proportional 1 mol add gas He 2 mol He
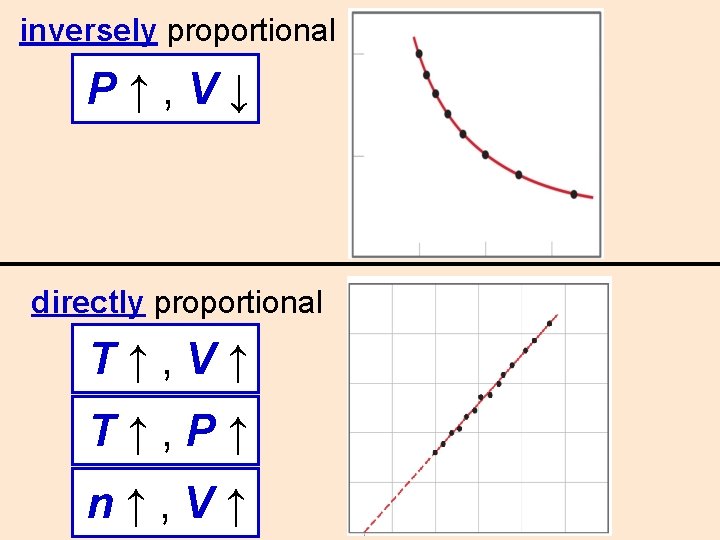
inversely proportional P↑, V↓ directly proportional T↑, V↑ T↑, P↑ n↑, V↑
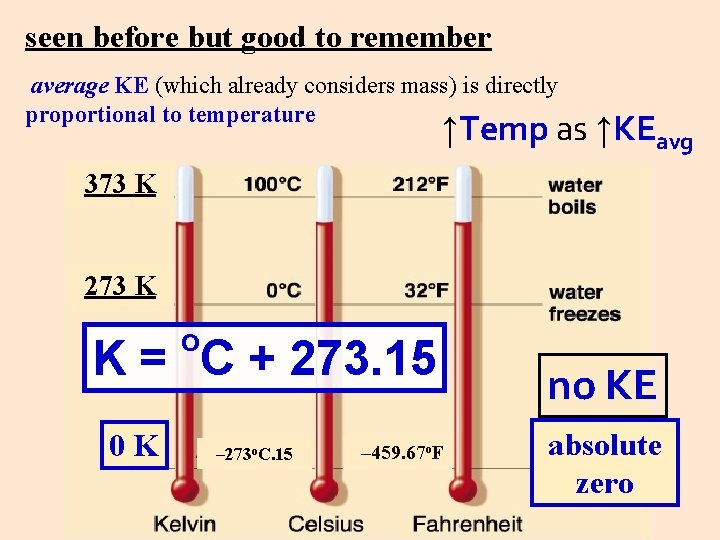
seen before but good to remember average KE (which already considers mass) is directly proportional to temperature ↑Temp as ↑KEavg 373 K 273 K o K = C + 273. 15 0 K – 273 o. C. 15 – 459. 67 o. F no KE absolute zero

Quick Quiz! 1) A balloon becomes slightly smaller when moved from a mountaintop to the seashore. Which gas law explains this behavior? A) Gay- Lussac’s law T↑, P↑ B) Boyle’s law P↑, V↓ C) Charles’s law T↑, V↑ D) Brock’s law Work from bell to bell!

Quick Quiz. 2) If the temperature of a gas in a flexible container is doubled, the volume will be… A) halved. B) doubled. C) the same. D) stupid anyway. T↑, V↑

Quick Quiz. 3) If the pressure of a gas in a flexible container is halved, the volume will be… A) halved. B) doubled. C) the same. D) squirrel! P↑, V↓ P↓, V↑ OR
- Slides: 9