BOTULISM Los Angeles County Department of Public Health

BOTULISM Los Angeles County Department of Public Health Acute Communicable Disease Control Program David E. Dassey MD, MPH and Public Health Laboratory Bioterrorism Response Unit Patricia Bolivar MS, CLS, SM (ASCP)

Objectives • Case report • Botulinum toxins • Clinical forms of botulism • Clinical diagnosis & differential • Laboratory confirmation • Treatment • Case report - conclusion

Case Report • 34 y/o female nursing student – Generalized weakness – Bulbar palsies • Admitting MD contacted Public Health – Release of botulinum antitoxin for treatment – Approval for botulism toxin testing by PH Lab 4 Serum & stool - direct toxin screen 4 Stool – culture for clostridia

Botulism • Symmetrical cranial nerve palsies • Descending, symmetric flaccid paralysis • of voluntary muscles Progression to respiratory compromise – Total paralysis – Death • German Botulismus: sausage poisoning, from Latin botulus (sausage)

Botulism • Neurotoxins produced by Clostridium – C. botulinum: toxins A, B, E, F, G [human disease] – C. botulinum: toxins C, D [non-human disease] – C. butyricum: toxin E – C. baratii: toxin F • Obligate anaerobic, spore-former • Toxin production in low-acid, p. H>4. 6 • All toxins are heat labile

Botulism Toxins • Toxin Type A, B and E most common in • • human cases Toxin Type F occurs infrequently in human cases Toxin Types C and D are associated with avian and animal botulism – Toxin production is phage mediated • Toxin Type G has been recovered from humans, however role in disease is unclear. – Toxin production is plasmid mediated

Botulism Toxins • Dichain polypeptide – zinc-containing metalloprotease – 100 -kd "heavy" chain – joined by a single disulfide bond to a – 50 -kd "light" chain • Distinguished by neutralization of biological activity with type-specific antisera (A – G) – Mouse bioassay
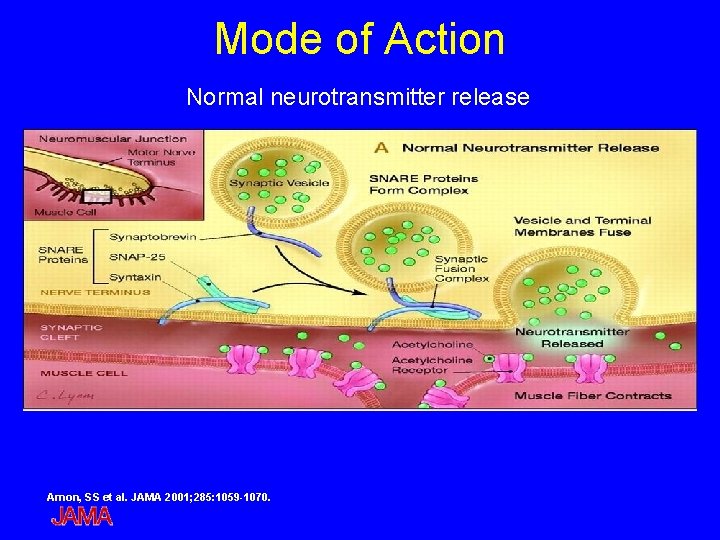
Mode of Action Normal neurotransmitter release Arnon, SS et al. JAMA 2001; 285: 1059 -1070.
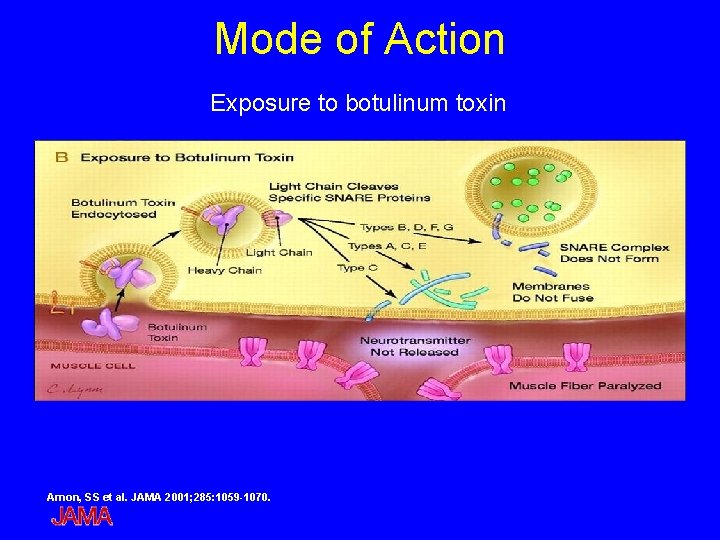
Mode of Action Exposure to botulinum toxin Arnon, SS et al. JAMA 2001; 285: 1059 -1070.

Toxin Lethal Dose • Lethal human oral dose for Bo. NT type A • • estimated to be between 100 – 1, 000 ng equivalent to 5, 000 to 50, 000 mouse lethal injected dose (MLD). Food implicated in cases of foodborne botulism have contained toxin as high as 10, 000 MLD/gram Some culture supernatants tested contain over 1, 000 MLD/ml

Growth and Toxin Production • C. botulinum grows under anaerobic, • low salt, low acid, low water activity Inhibited by – temp <4°C or >121°C – p. H <4. 5 • Spores inactivated – 121°C under pressure of 15 -20 lb/in² • Toxin destroyed by – Heating >85°C for 5 min

Naturally Occurring Disease Forms • Naturally occurring – Food-borne – Wound – Infant – Intestinal – Other/Undetermined

Other Disease Forms • Unintentional (iatrogenic) – Following toxin injection for therapeutic or cosmetic purposes • Intentional act of terrorism – Aerosolization, absorption through mucous membranes or break in skin – Distributed on food items
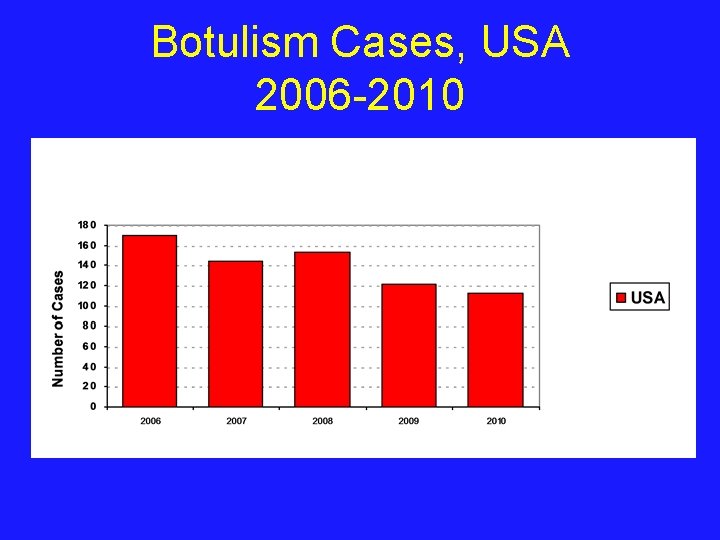
Botulism Cases, USA 2006 -2010
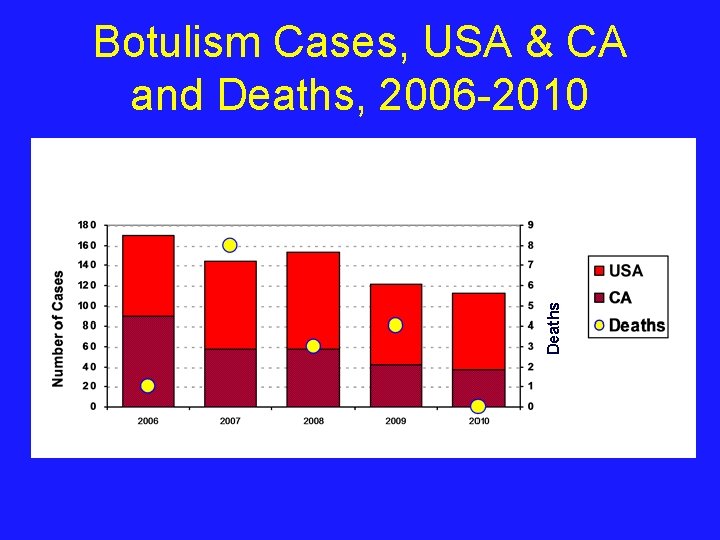
Deaths Botulism Cases, USA & CA and Deaths, 2006 -2010
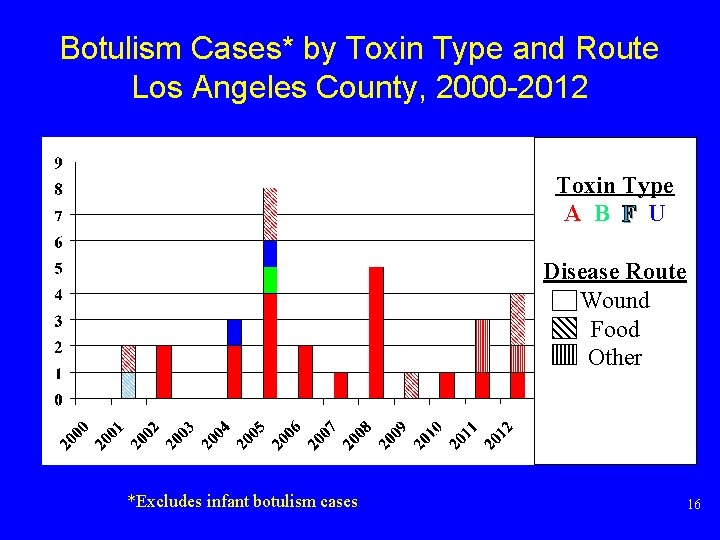
Botulism Cases* by Toxin Type and Route Los Angeles County, 2000 -2012 Toxin Type A B F U Disease Route Wound Food Other *Excludes infant botulism cases 16

Botulism, Foodborne – Case Definition • Case Classification – Probable: clinically compatible case with epidemiologic link (eg, ingestion of home-canned food within previous 48 hours) – Confirmed: clinically compatible case that is laboratory confirmed or that occurs among persons who ate the same food as persons who have laboratory-confirmed botulism • Laboratory Criteria for Diagnosis – Detection of botulinum toxin in serum, stool, or patient's food, or isolation of C. botulinum from stool

Foodborne Botulism • Incubation dependent on quantity and rate of absorption of toxin – as early as 2 – 8 hours after meal consumption – typical incubation is 12 -72 hours after consumption – GI symptoms may occur • Mild cases may not be detected – Botulism from Chopped Garlic: Delayed Recognition of a Major Outbreak. Ann Intern Med. 1 March 1988
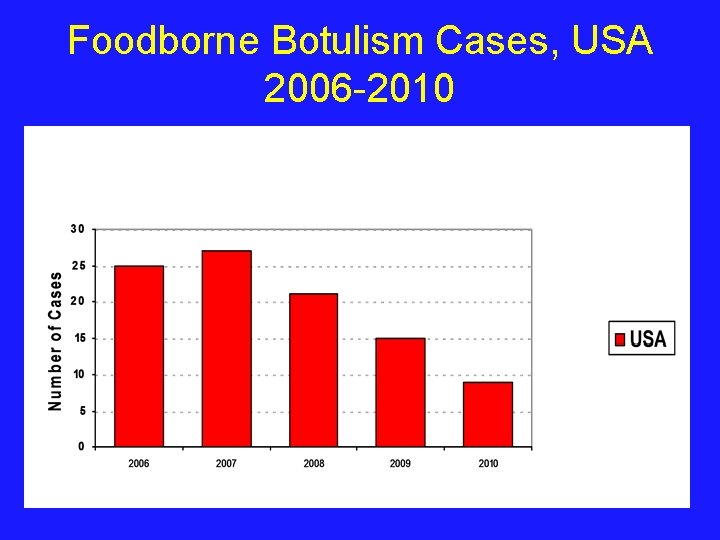
Foodborne Botulism Cases, USA 2006 -2010
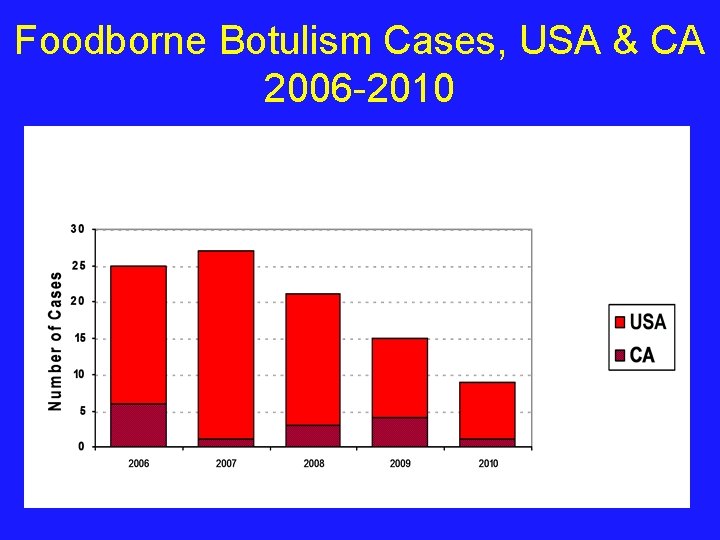
Foodborne Botulism Cases, USA & CA 2006 -2010
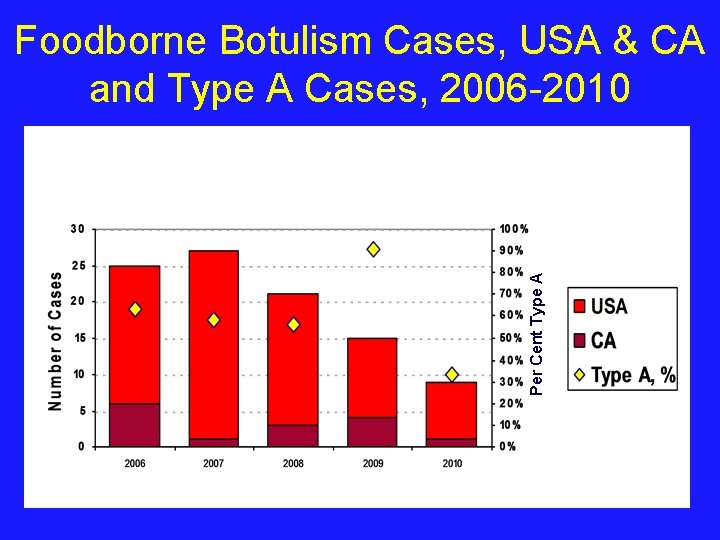
Per Cent Type A Foodborne Botulism Cases, USA & CA and Type A Cases, 2006 -2010
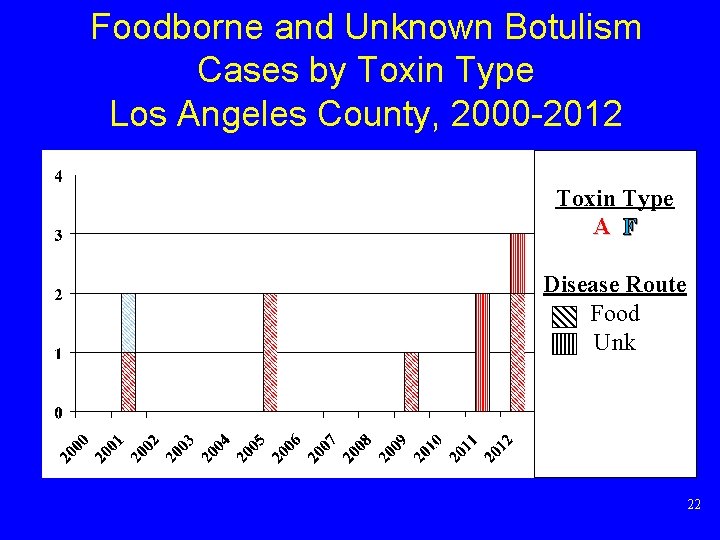
Foodborne and Unknown Botulism Cases by Toxin Type Los Angeles County, 2000 -2012 Toxin Type A F Disease Route Food Unk 22

Foodborne Botulism Vehicles • • Home-canned or home processed foods Low-acid (p. H >4. 6) – – – – Vegetables Relish, salsa Peppers Meats Fish Fermented, salted fish Whale, seal • • Baked potatoes in foil Garlic in oil Sautéed onions in butter sauce Cheese sauce Pot pie Canned chili “Pruno”

Botulism, Wound – Case Definition • • Case Classification – Confirmed: clinically compatible case that is laboratory confirmed in a patient who has no suspected exposure to contaminated food and who has a history of a fresh, contaminated wound during the 2 weeks before onset of symptoms, or a history of injection drug use within the 2 weeks before onset of symptoms – Probable: a clinically compatible case in a patient who has no suspected exposure to contaminated food and who has either a history of a fresh, contaminated wound during the 2 weeks before onset of symptoms, or a history of injection drug use within the 2 weeks before onset of symptoms Laboratory Criteria for Diagnosis – Detection of botulinum toxin in serum, or isolation of C. botulinum from wound

Wound Botulism • • Growth of C. botulinum in wounds with toxin production in vivo Neurological presentation is indistinguishable from other forms of botulism, tho more insidious Majority of current cases associated with injection drug use – skin popping No gastrointestinal involvement • Type A – 80% Type B – 20% • •
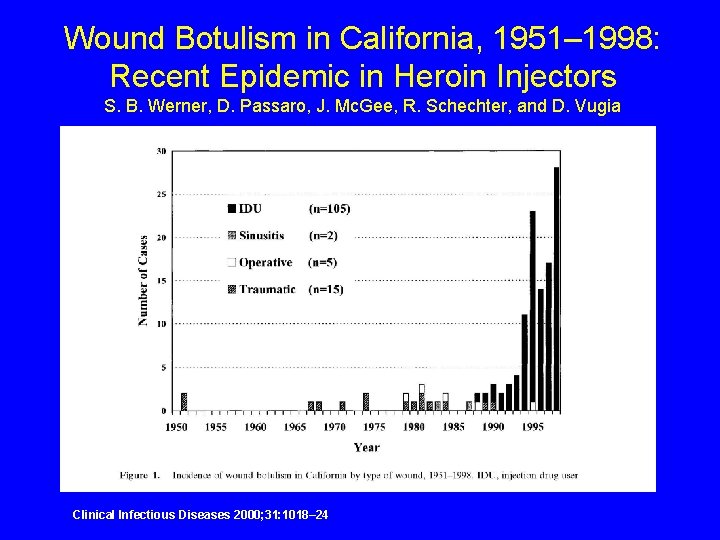
Wound Botulism in California, 1951– 1998: Recent Epidemic in Heroin Injectors S. B. Werner, D. Passaro, J. Mc. Gee, R. Schechter, and D. Vugia Clinical Infectious Diseases 2000; 31: 1018– 24

Wound Botulism in California, 1951– 1998: Recent Epidemic in Heroin Injectors S. B. Werner, D. Passaro, J. Mc. Gee, R. Schechter, and D. Vugia Clinical Infectious Diseases 2000; 31: 1018– 24
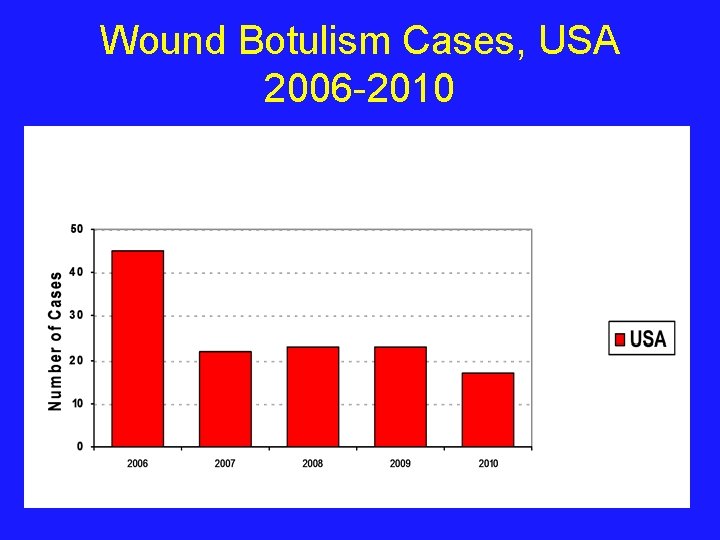
Wound Botulism Cases, USA 2006 -2010
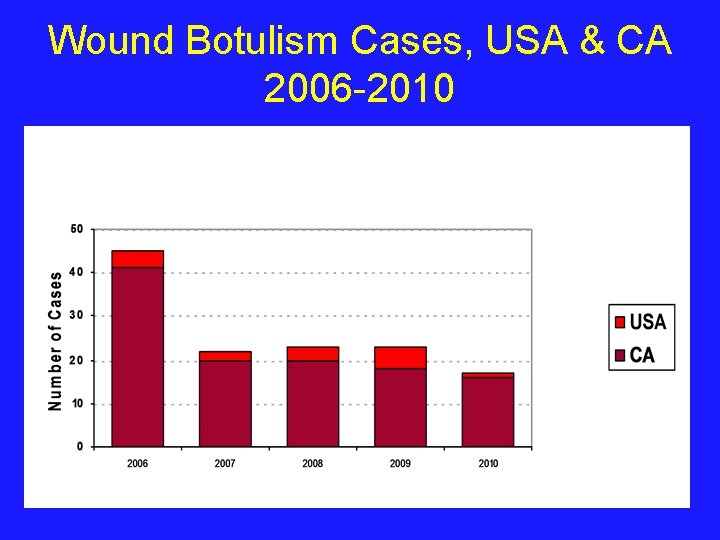
Wound Botulism Cases, USA & CA 2006 -2010
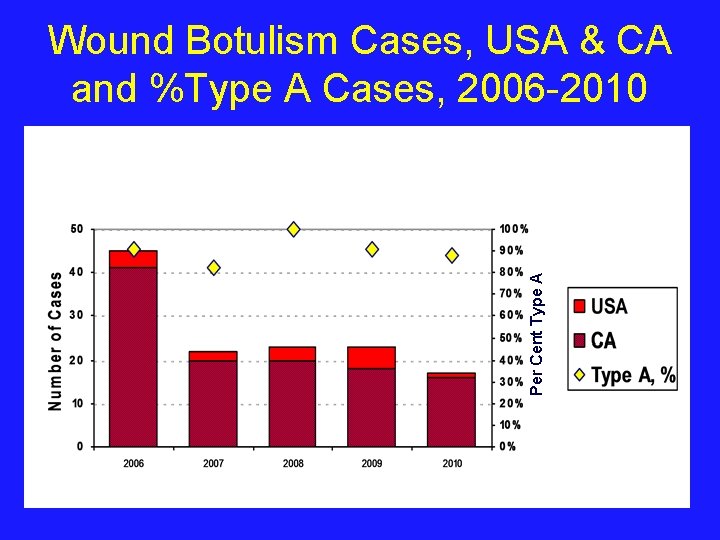
Per Cent Type A Wound Botulism Cases, USA & CA and %Type A Cases, 2006 -2010
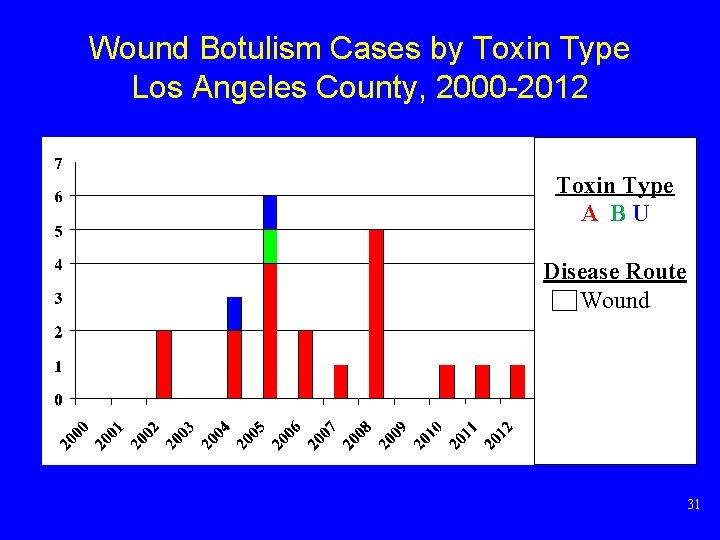
Wound Botulism Cases by Toxin Type Los Angeles County, 2000 -2012 Toxin Type A BU Disease Route Wound 31

Infant (Intestinal) Botulism • Most common form of botulism reported – 50% type A, 50% type B – Intestinal tract becomes colonized with spores of C. botulinum with subsequent production of toxin – Lethargy, poor feeding, floppy head with progression to more severe disease if not treated • Adult intestinal botulism – GI anatomical defect, rare
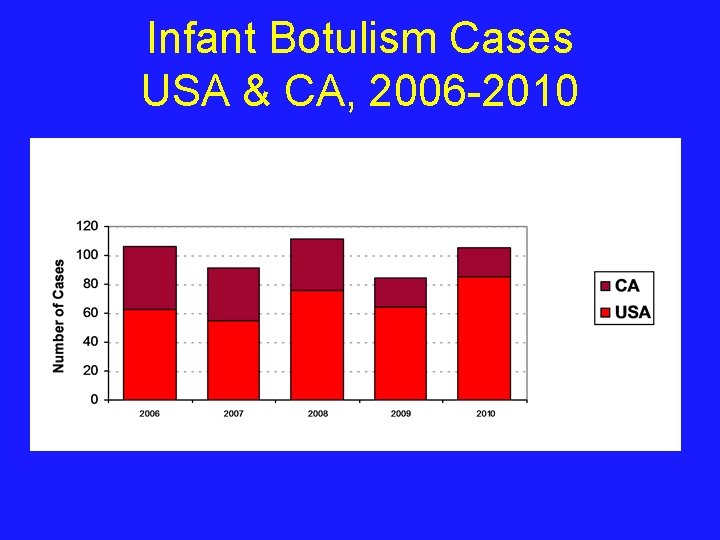
Infant Botulism Cases USA & CA, 2006 -2010

Botulism, Other – Case Definition • Case Classification – Confirmed: a clinically compatible case that is laboratory-confirmed in a patient aged greater than or equal to 1 year who has no history of ingestion of suspect food and has no wounds • Laboratory Criteria for Diagnosis – Detection of botulinum toxin in clinical specimen, or isolation of C. botulinum from clinical specimen

Iatrogenic Botulism • Therapeutic use of botulinum toxins – – Strabismus Cervical dystonia Blepharospasm Spasticity (not FDA approved) • Cosmetic uses – Botulism Disaster Uncovers Fake Botox Market 2004 South Florida outbreak

Botulinum Toxin As Possible Bioweapon • Inhalational botulism – Japanese biological warfare group 4 Unit 731, 1930 s – Germany, WW-II – Cold War 4 Soviet Union, Aralsk-7 4 USA - ended in 1970 – Aum Shinrikyō cult 4 Attempted on at least 3 occasions 1990 -1995 – Iran, Iraq, North Korea, Syria

Features That Suggest Deliberate Release of Botulinum Toxin • Outbreak of a large number of cases of acute • • • flaccid paralysis with prominent bulbar palsies Outbreak with an unusual botulinum toxin type (ie, type C, D, F, or G, or type E toxin not acquired from an aquatic food) Outbreak among cases with a common geographic factor (eg, airport, work location) but without a common dietary exposure Multiple simultaneous outbreaks with no common source

Clinical Presentation • Bilateral descending flaccid paralysis beginning with cranial nerves – – – Diplopia Difficulty in swallowing, dysarthria Vertigo, dizziness, unsteadiness Neck and extremity muscle weakness Chest, diaphragm involvement lead to respiratory paralysis 4 Fatal if supportive therapy not provided • Alert, normal vital signs, afebrile • Normal sensory exam

Clinical Diagnosis • Autonomic findings – Dry mouth, sore throat, anhydrosis – Constipation • GI (foodborne only) – Nausea, vomiting may precede neuro signs • Absence of cranial nerve palsies nearly always rules out botulism • History of – Home-canned or spoiled food – Similar illness in persons sharing food – Injection or wound ĉ/ŝ visible abscess

Incubation Period • Dependent on rate and amount of toxin • • absorbed More rapid in foodborne botulism Wound botulism is generally very insidious – Days to weeks of very minor symptoms

Workup • Detailed history • Complete physical exam, particularly • looking for minor wounds Thorough neurological exam – Normal sensory • Head – MRI, CT • Lumbar puncture for CSF

Workup • Edrophonium (Tensilon) challenge test – Falsely positive in 25% • EMG – Decreased action potentials in affected muscles – Repetitive stimulation @ high frequency (20 -50 Hz) yields increased amplitude (facilitation) • Appropriate toxicological studies

Differential Diagnosis major conditions • Guillain-Barré & Miller-Fisher Syndromes – Ascending / Descending paralysis 4 MFS: ophthalmoplegia, ataxia, areflexia – Pain, parasthesias – Elevated CSF protein (delayed) – Electromyography 4 Marked slowing of NCV 4 No MAP augmentation (facilitation) at hi frequency 20 -50 Hz – Anti-ganglioside antibodies Clinical Infectious Diseases 2000; 31: 1018– 24

Differential Diagnosis major conditions • Myasthenia gravis – Muscle fatigability, resolves with edrophonium test 4~25% mild botulism cases also respond – EMG - decrement in MAP with rapid stimulation at 3 Hz • Cerebrovascular accident of midbrain – May not be visualized early Clinical Infectious Diseases 2000; 31: 1018– 24

Differential Diagnosis minor conditions • • • Polio, other • encephalitides • Tick paralysis • Wernicke encephalopathy • Eaton Lambert myasthenic syndrome Electrolyte abnormalities • Clinical Infectious Diseases 2000; 31: 1018– 24 Paralytic shellfish poisoning Carbon monoxide poisoning Organophosphate poisoning Aminoglycoside paralysis – gentamicin, tobramycin, streptomycin, etc. Poisoning with belladonalike alkaloids

Laboratory Diagnosis • Toxin detection in clinical samples – Serum, stool, vomitus, wound tissue or exudates – Collect specimens from patients prior to administering anti-toxin • Food – detection of toxin or a toxigenic organism in implicated food item • Culture and isolation of toxigenic organism from wound, feces, gastric contents

Laboratory Diagnosis • Mouse toxicity and neutralization bioassay for toxin • Diffusion-in-gel ELISA – ABEF proteins • Real time PCR for botulinum toxin gene • Mass spectrometry – toxins AB

Mouse Bioassay • Confirmatory test • Detects functionally active toxin • Sensitivity: 10 – 30 pg • Requires extensive animal use • Results obtained within 1 - 4 days
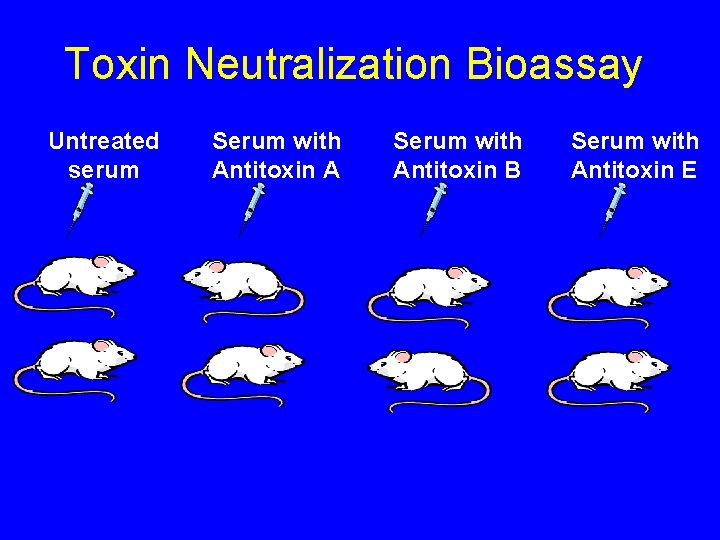
Toxin Neutralization Bioassay Untreated serum Serum with Antitoxin A Serum with Antitoxin B Serum with Antitoxin E
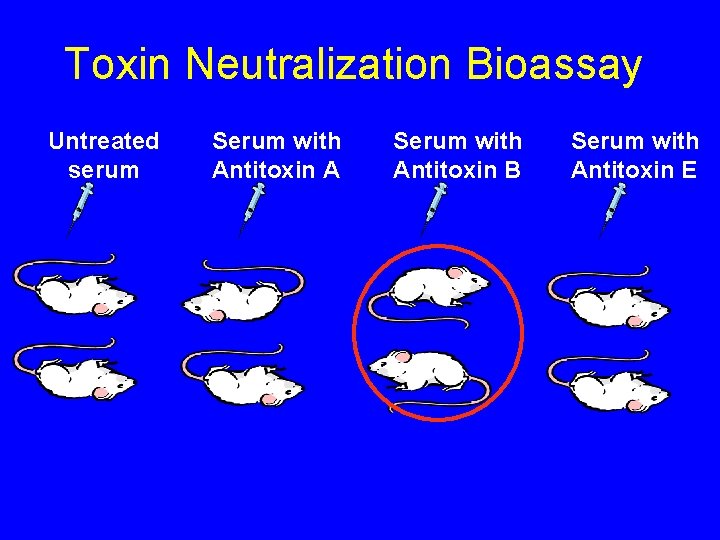
Toxin Neutralization Bioassay Untreated serum Serum with Antitoxin A Serum with Antitoxin B Serum with Antitoxin E
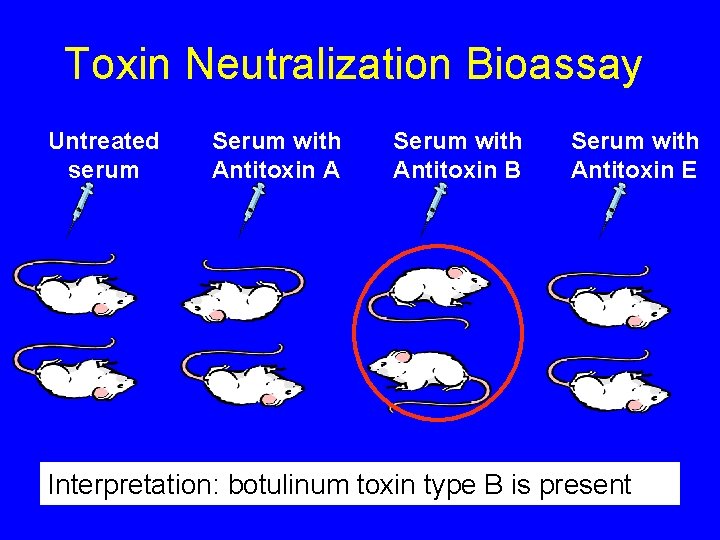
Toxin Neutralization Bioassay Serum with Antitoxin B Serum with Antitoxin E 1 B 2 E Serum with Antitoxin A 1 E Untreated serum 1 U 1 A 2 U 2 A Interpretation: botulinum toxin type B is present

Botulinum Toxin DIG-ELISA • Presumptive test for toxins ABEF • Detects toxin protein structural elements • Sensitivity <10 pg • Animals are not required • Results obtained within 4. 5 h

Real-time PCR for Detection of Neurotoxin Genes (types A-G) • Presumptive identification and differentiation of Bo. NT – A, B, C, D, E, F, or G • Clostridia in enrichment broths from – Foods – Environmental samples

Evolving Diagnostics • Matrix-assisted laser desorption/ ionization - time of flight (MALDI-TOF) mass spectrometry – capable of detecting and differentiating botulinum toxins, A, B, E and F – Concentrations starting from 5 pg/m. L – Clinical, food and environmental samples

LA County 2000 -2007 • 54 reports of suspected botulism cases – 18 confirmed botulism cases – 17 unconfirmed cases of clinical botulism – 19 patients with other/unk diagnoses • 32 (62%) reports were in IDU – 14 confirmed botulism cases – 14 unconfirmed cases of clinical botulism – 4 patients with other/unk diagnoses 55

LA County 2000 -2007 • 54 reports of suspected botulism cases – 18 confirmed botulism cases – 17 unconfirmed cases of clinical botulism – 19 patients with other/unk diagnoses • 32 (62%) reports were in IDU – 14 confirmed botulism cases – 14 unconfirmed cases of clinical botulism – 4 patients with other/unk diagnoses 56

LA County 2000 -2007 • 54 reports of suspected botulism cases – 18 confirmed botulism cases – 17 unconfirmed cases of clinical botulism – 19 patients with other/unk diagnoses • 32 (62%) reports were in IDU – 14 confirmed botulism cases – 14 unconfirmed cases of clinical botulism – 4 patients with other/unk diagnoses 57

LA County 2000 -2007 • 54 reports of suspected botulism cases – 18 confirmed botulism cases – 17 unconfirmed cases of clinical botulism – 19 patients with other/unk diagnoses • 32 (62%) reports were in IDU – 14 confirmed botulism cases – 14 unconfirmed cases of clinical botulism – 4 patients with other/unk diagnoses 58

LA County 2000 -2007 • 54 reports of suspected botulism cases – 18 confirmed botulism cases – 17 unconfirmed cases of clinical botulism – 19 patients with other/unk diagnoses • 32 (62%) reports were in IDU – 14 confirmed botulism cases – 14 unconfirmed cases of clinical botulism – 4 patients with other/unk diagnoses 59

Alternative Diagnoses for 19 Botulism Suspects, 2000 -2007 • Reported botulism suspects that did not confirm – 9 Guillain-Barré / MF syndrome 42 with campylobacteriosis 41 with unspecified diarrheal illness – 2 circulatory: brain stem; multi-infarcts – 2 neoplasia: brain stem, cervical chord – 4 inflammatory: vasculitis, paraneoplastic syndrome, encephalitis, polyneuropathy – 2 no other diagnosis or unknown 60

Report to Public Health Immediately • Authorization for specific confirmatory • • testing in Public Health Lab Release of antitoxin for treatment Rule out possible foodborne outbreak or intentional release of toxin • REPORT to Public Health AS SOON AS BOTULISM IS CONSIDERED

Treatment • Antitoxin released upon consultation with PH physician – stored by CDC at LAX Airport – ABCDEFG heptavalent antitoxin • Treatment is NEVER dependent on results of specific botulism toxin tests.

Treatment • Investigational Heptavalent Botulinum Antitoxin (HBAT) to Replace Licensed Botulinum Antitoxin AB and Investigational Botulinum Antitoxin E 4 MMWR, March 19, 2010 / 59(10); 299 4 IND and post-treatment data required

Treatment • Botulinum antitoxin – Most effective given early in course of illness – Neutralizes only circulating toxin – Does not reverse neurological symptoms 4 Motor recovery with regrowth of nerve endings – Circulating half life of antitoxin is 5 – 8 days 4 Rarely is second dose needed – Determine if patient is hypersensitive to equine derived products with skin prick test

Treatment • Foodborne botulism – Cathartic to empty GI tract • Wound botulism – Debridement only after antitoxin administration – Antibiotic coverage • Respiratory and supportive therapy – Patient is alert, not comatose !

Treatment of Infant Cases • Baby. BIG® – Botulism Immune Globulin Intravenous (Human) (BIG-IV) (Baby-BIG) – human-derived anti-botulism toxin antibodies manufactured by California DPH – approved by US FDA for treatment of infant botulism types A and B – Released by State on consultation – 510 -231 -7600 [24 -hour hotline]

Case Follow-Up - 1 • Suspected case lived with married couple – Recent immigrants from Middle East – Denied current symptoms, denied home canned food • Environmental Spec. and PHN found – 30 gallons (5 opened) of home-pickled eggplant, onions, garlic – All were embargoed for possible toxin tests.

Case Follow-Up - 2 • Husband admitted to different hospital 12 hours later with dysphonia, dysphagia – Additional jar of eggplant at bedside • Wife had been admitted 6 weeks earlier for 3 weeks with neck weakness – 8 months pregnant – Diagnosed with myasthenia gravis, r/o botulism 4 Not reported by attending MD or hospital lab – Normal infant delivered 2 weeks later

Case Follow-Up - 3 • Eggplant + for botulinum toxin type A – All jars were destroyed • Index case +toxin A in serum and stool – Prolonged hospitalization, complications and rehabilitation • Husband hospitalized ~ 5 days • Both were preventable if original suspect case had been reported.

How to Report ? For emergent communicable disease reports, outbreaks, or unusual disease occurrences (possible zebras): Acute Communicable Disease Control (213) 240 -7941

References • • • Botulism in the United States: a clinical and epidemiological review. Shapiro RL, Hathaway C, Swerdlow DL. Ann Intern Med 1998; 129(3): 221 -8. Wound botulism in California, 1951 -1998: recent epidemic in heroin injectors. Werner SB, Passaro D, Mc. Gee J, Schechter R, Vugia DJ. Clin Infect Dis. 2000 Oct; 31(4): 1018 -24. Botulinum toxin as a biological weapon - medical and public health management. Arnon SS, et al. for the Working Group on Civilian Biodefense. JAMA. 2001; 285: 1059 -1070. Botulism. Sobel J. Clin Infect Dis. 2005 Oct 15; 41(8): 1167 -73. CDC: Botulism in the United States, 1899 -1996. Handbook for epidemiologists, clinicians, and laboratory workers. Atlanta, GA. Centers for Disease Control and Prevention, 1998.


Confirmed Botulism Cases Los Angeles County, 2000 -2012 Year Foodborne Wound Other 2000 - - 2001 2 A F - 2002 - 2 AA 2003 - - 2004 - 3 AA U 2005 2 AA 6 AAAA B U 2006 - 2 AA 2007 - 1 A 2008 - 5 AAAAA - 2009 1 A - - 2010 - 1 A - 2011 - 1 AA 2012 2 AA Total 7 1 A 22 1 A 3 73
- Slides: 73