Boston Scientific Vessix Renal Denervation System Taped Case

Boston Scientific Vessix™ Renal Denervation System: Taped Case Farrell O. Mendelsohn, MD Director, Center for Therapeutic Angiogenesis Cardiology PC Birmingham, Alabama

Farrell O. Mendelsohn, MD Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below Affiliation/Financial Relationship Company Boston Scientific / Vessix Vascular Consulting Fees Medtronic Consulting Fees

Disclosures IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the on-label use of referenced technologies as well as current standards of care for certain conditions. Of course, patients and their medical circumstances vary, so the clinical considerations and procedural steps described may not be appropriate for every patient or case. As always, decisions surrounding patient care depend on the physician’s professional judgment in light of all available information for the case at hand. BSC does not promote or encourage the use of its devices outside their approved labeling. The presenter’s experience with BSC products may not be interpreted or relied upon to support clinical claims about BSC devices or product comparison claims regarding BSC and competitive devices. The experiences of other users may vary. This presentation contains several case studies. Results from case studies are not predictive of results in other cases. Results in other cases may vary.

Vessix™ Clinical Program REDUCE-HTN Post Market Study Clinical Study Overview Postmarket surveillance clinical study for medication-resistant hypertension • Single cohort, international, multi-center, prospective, treatment registry • Up to 150 subjects at 25 clinical sites in EU, AU, and NZ • Adults with systolic blood pressure > 160 mm. Hg on three or more antihypertensive drugs at maximum tolerated doses • Efficacy Endpoint – Reduction of office based systolic and diastolic blood pressure at six months post procedure compared to baseline. • Safety Endpoint – Absence of flow limiting stenosis in the renal artery at six months post procedure (measured by DUS) *The Vessix™ Renal Denervation System is not available for sale in the US.

Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US

Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US
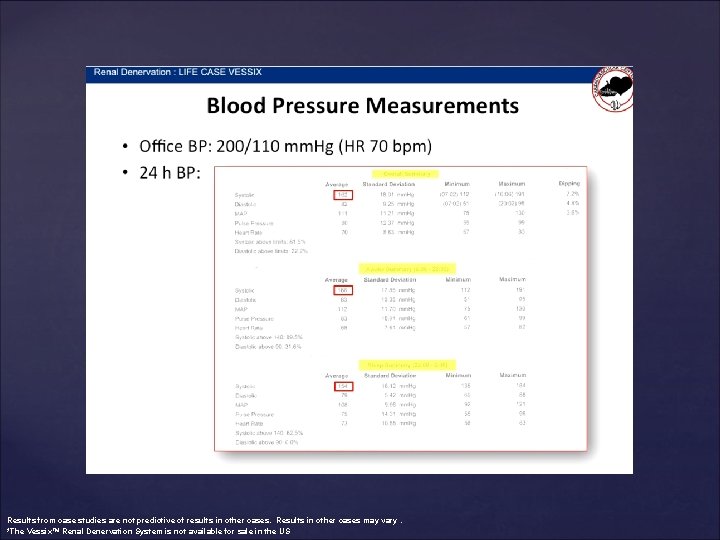
Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US
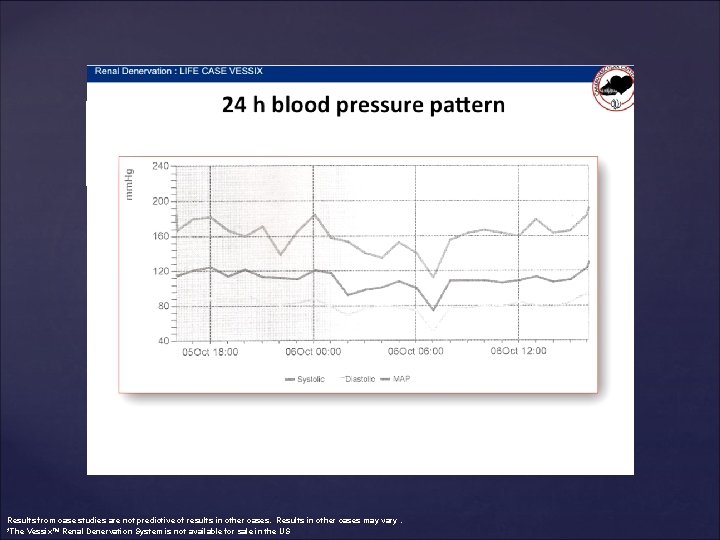
Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US
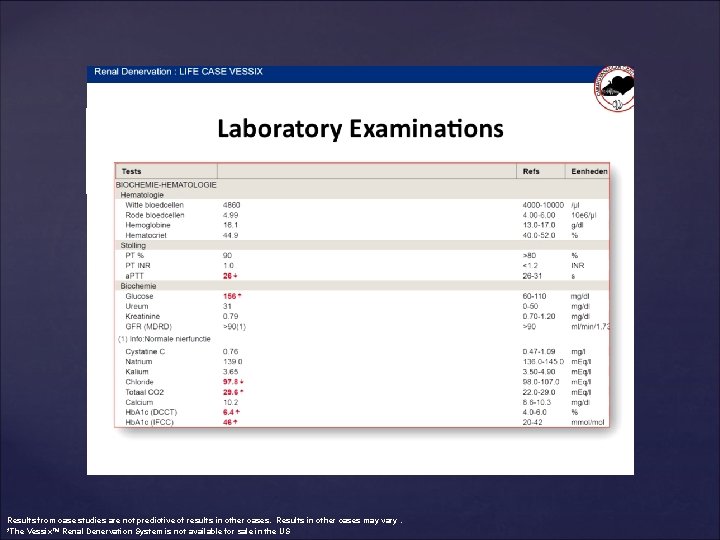
Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US
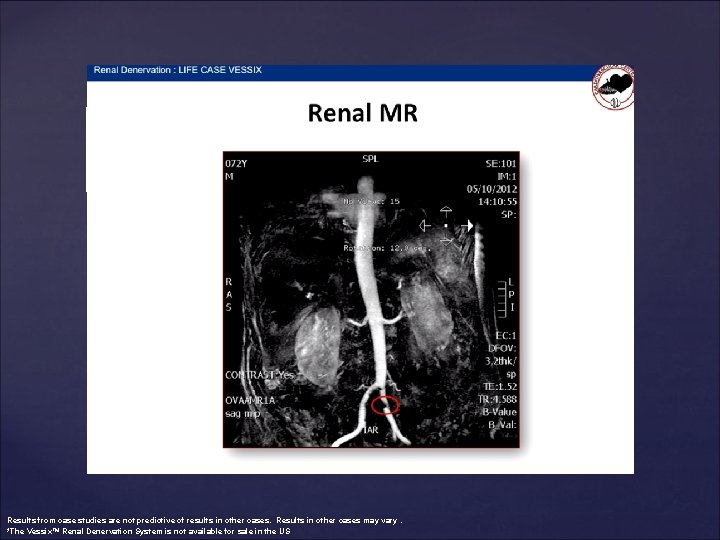
Results from case studies are not predictive of results in other cases. Results in other cases may vary. *The Vessix™ Renal Denervation System is not available for sale in the US

Taped Case

Thank you
- Slides: 13