BORNHABER CYCLES Lattice enthalpy For an ionic compound

BORN-HABER CYCLES

Lattice enthalpy For an ionic compound the lattice enthalpy is the heat energy released when one mole of solid in its standard state is formed from its ions in the gaseous state. Eg. Na+(g) + Cl-(g) Na. Cl(s) - L. E cannot be determined directly - enthalpy cycle (Hess’s Law) links the data. MOLAR HEATS OF FORMATION - enthalpy cycle is based on the formation of the compound from its elements in standard states.
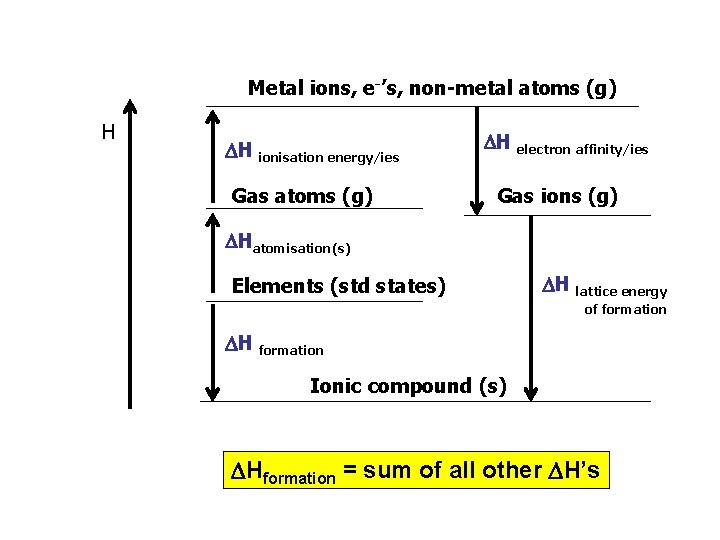
Metal ions, e-’s, non-metal atoms (g) H H ionisation energy/ies Gas atoms (g) H electron affinity/ies Gas ions (g) Hatomisation(s) Elements (std states) H lattice energy of formation H formation Ionic compound (s) Hformation = sum of all other H’s
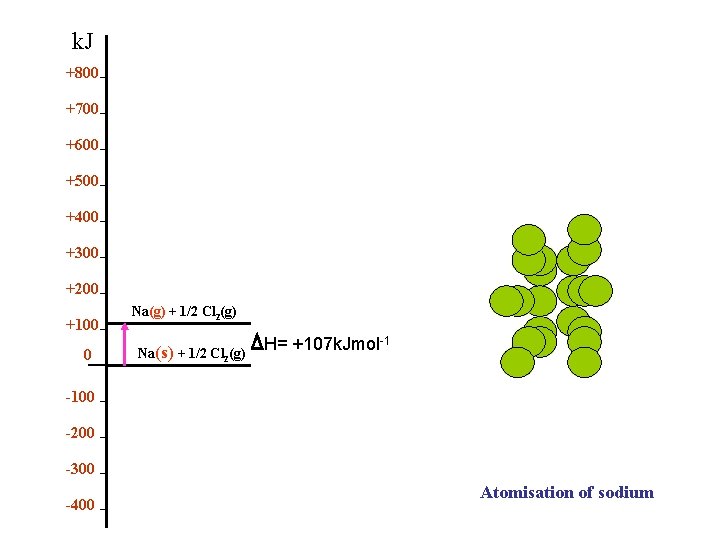
k. J +800 +700 +600 +500 +400 +300 +200 +100 0 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) H= +107 k. Jmol-1 -100 -200 -300 -400 Atomisation of sodium
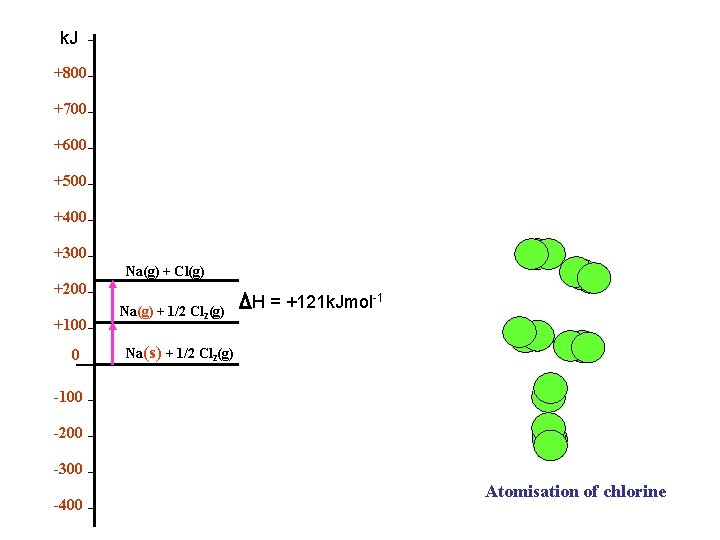
k. J +800 +700 +600 +500 +400 +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) H = +121 k. Jmol-1 Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 Atomisation of chlorine
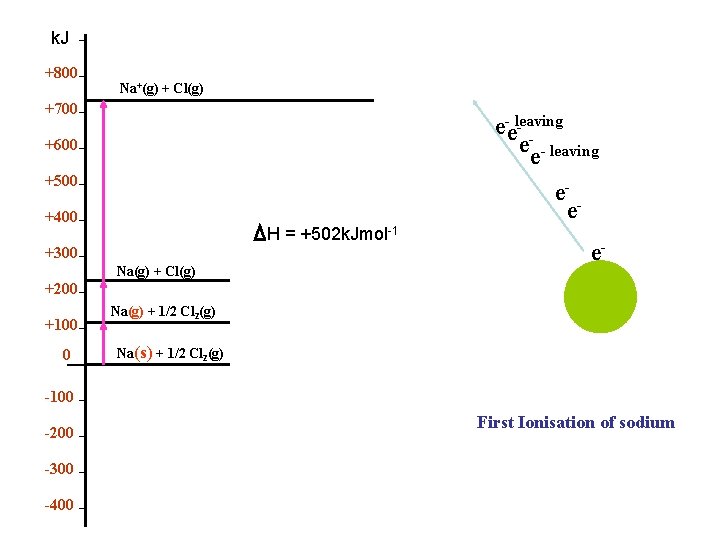
k. J +800 Na+(g) + Cl(g) +700 e-eleaving e-e- leaving e- e +600 +500 +400 H = +502 k. Jmol-1 +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) e+ Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 First Ionisation of sodium
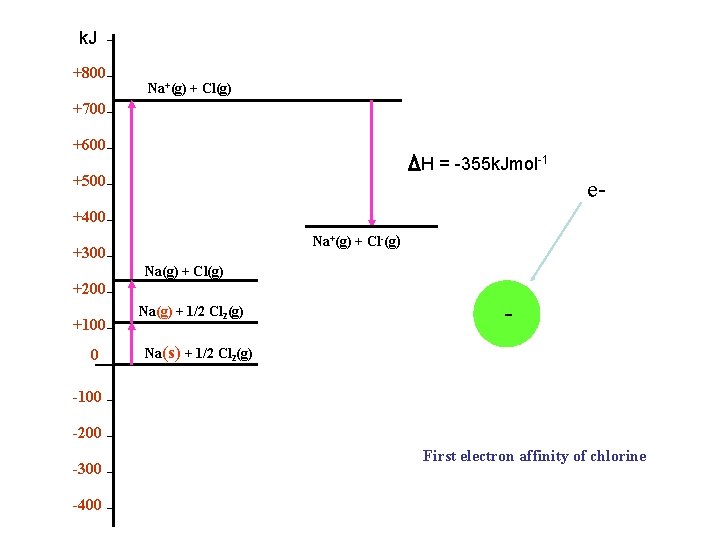
k. J +800 Na+(g) + Cl(g) +700 +600 H = -355 k. Jmol-1 +500 e- +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) - e- Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 First electron affinity of chlorine
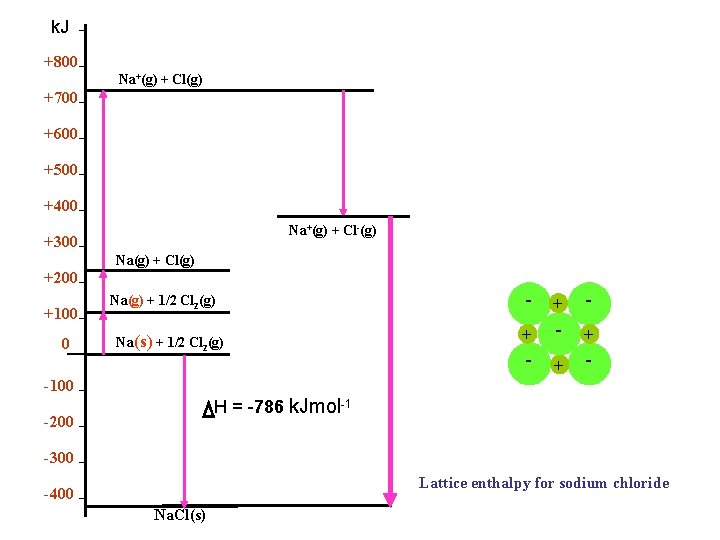
k. J +800 Na+(g) + Cl(g) +700 +600 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) -100 - + - + - H = -786 k. Jmol-1 -200 -300 Lattice enthalpy for sodium chloride -400 Na. Cl(s)
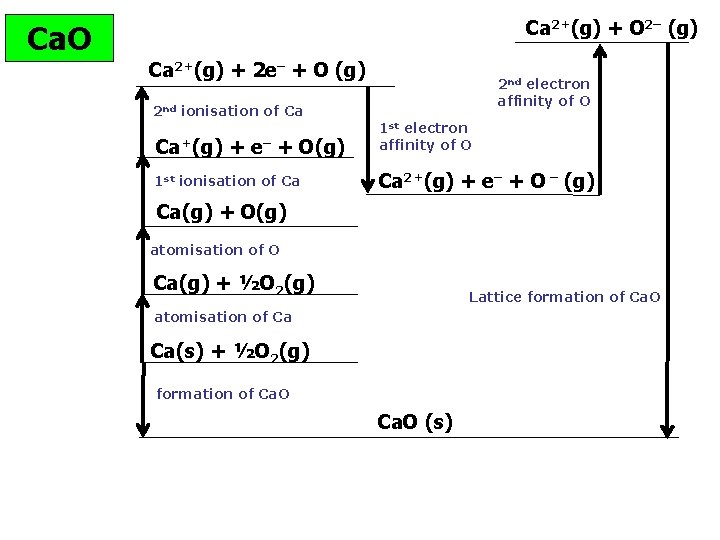
Ca. O Ca 2+(g) + O 2– (g) Ca 2+(g) + 2 e– + O (g) 2 nd ionisation of Ca 2 nd electron affinity of O Ca+(g) + e– + O(g) 1 st electron affinity of O 1 st ionisation of Ca Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca Ca(s) + ½O 2(g) formation of Ca. O (s)

Ca 2+(g) + O 2– (g) Ca. O Ca 2+(g) + 2 e– + O (g) 1150 2 nd ionisation of Ca Ca+(g) 590 248 + e– + O(g) 1 st ionisation of Ca – 142 1 st electron affinity of O – 635 844 Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca 193 2 nd electron affinity of O X Ca(s) + ½O 2(g) formation of Ca. O (s) Hf Ca. O= – 635; Atomisation calcium = +193; Atomisation oxygen = +248 1 st IE Ca = +590; 2 nd IE Ca = +1150 1 st e- affinity of oxygen = – 142 k. J mol-1 2 nd e- affinity= +844

Ca 2+(g) + O 2– (g) Ca. O Ca 2+(g) + 2 e– + O (g) 1150 2 nd ionisation of Ca Ca+(g) 590 248 + e– + O(g) 1 st ionisation of Ca – 142 1 st electron affinity of O – 635 844 Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca 193 2 nd electron affinity of O X Ca(s) + ½O 2(g) formation of Ca. O (s) = 193 + 248 + 590 + 1150 – 142 + 844 + Hlattice = – 635 – 193 – 248 – 590 – 1150 + 142 – 844 = – 3518 k. J mol-1 © www. chemsheets. co. uk A 2 1015 19 -May-2016 – 635

Mg. Br 2 Mg 2+(g) + 2 e– + 2 Br (g) 2 nd ionisation of Mg Mg+(g) + e– + 2 Br(g) 1 st ionisation of Mg 2 x electron affinity of Br Mg 2+(g) 2 Br – (g) Mg(g) + 2 Br(g) 2 x atomisation of Br Mg(g) + Br 2(l) Lattice formation of Mg. Br 2 atomisation of Mg Mg(s) + Br 2(l) formation of Mg. Br 2 (s)

Mg. Br 2 Mg 2+(g) + 2 e– + 2 Br (g) 1450 736 2(112) 150 X 2 x electron affinity of Br 2 nd ionisation of Mg Mg+(g) + e– + 2 Br(g) 1 st ionisation of Mg 2(– 342) Mg 2+(g) 2 Br – (g) Mg(g) + 2 Br(g) 2 x atomisation of Br Mg(g) + Br 2(l) Lattice formation of Mg. Br 2 atomisation of Mg – 2394 Mg(s) + Br 2(l) formation of Mg. Br 2 (s) Hformation = 150 + 2(112) + 736 + 1450 + 2(– 342) – 2394 Hformation = – 518 k. J mol-1 © www. chemsheets. co. uk A 2 1015 19 -May-2016
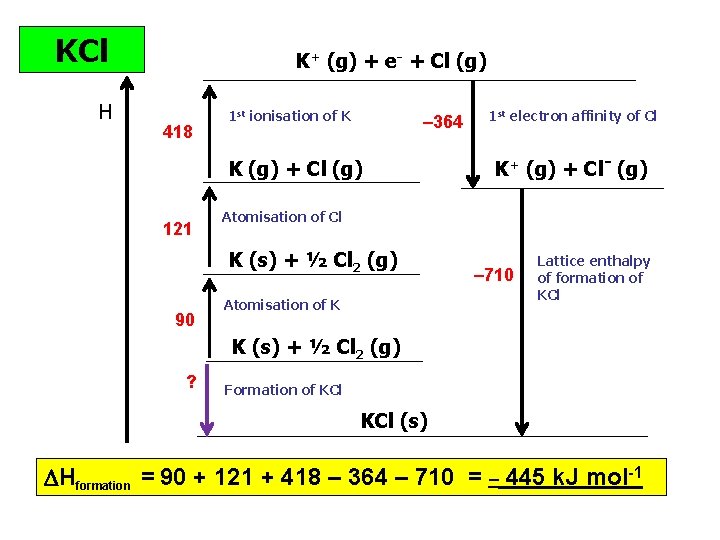
KCl H K+ (g) + e- + Cl (g) 418 1 st ionisation of K – 364 K (g) + Cl (g) 121 K+ (g) + Cl- (g) Atomisation of Cl K (s) + ½ Cl 2 (g) 90 1 st electron affinity of Cl Atomisation of K – 710 Lattice enthalpy of formation of KCl K (s) + ½ Cl 2 (g) ? Formation of KCl (s) Hformation = 90 + 121 + 418 – 364 – 710 = – 445 k. J mol-1
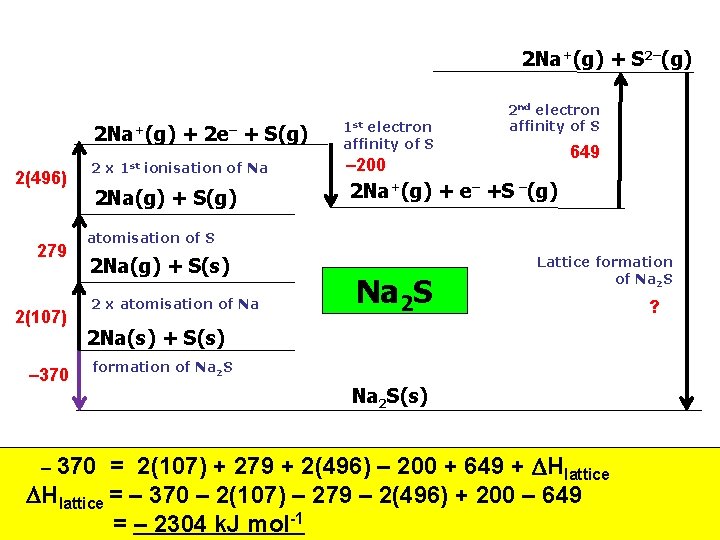
2 Na+(g) + S 2–(g) 2 Na+(g) 2(496) 279 2(107) – 370 2 x 1 st + 2 e– + S(g) ionisation of Na 2 Na(g) + S(g) 1 st electron affinity of S 2 nd electron affinity of S 649 – 200 2 Na+(g) + e– +S –(g) atomisation of S 2 Na(g) + S(s) 2 x atomisation of Na Na 2 S Lattice formation of Na 2 S 2 Na(s) + S(s) formation of Na 2 S(s) = 2(107) + 279 + 2(496) – 200 + 649 + Hlattice = – 370 – 2(107) – 279 – 2(496) + 200 – 649 = – 2304 k. J mol-1 – 370 ?
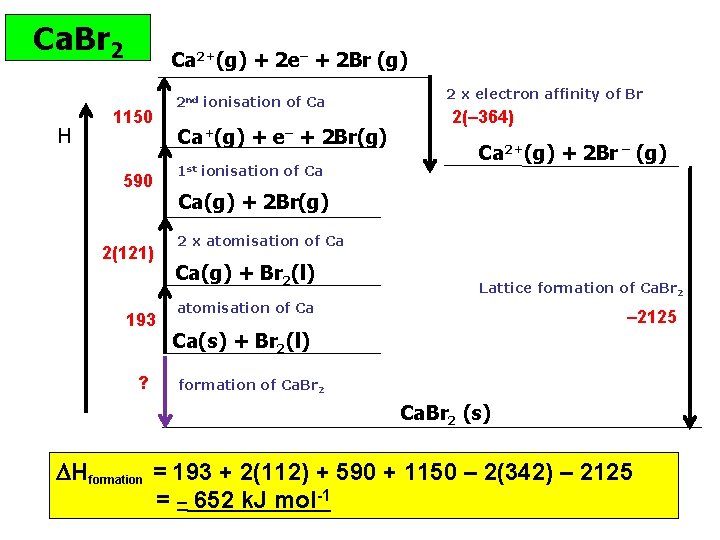
Ca. Br 2 H Ca 2+(g) + 2 e– + 2 Br (g) 1150 590 2(121) 193 ? 2 nd ionisation of Ca Ca+(g) + e– + 2 Br(g) 1 st ionisation of Ca 2 x electron affinity of Br 2(– 364) Ca 2+(g) + 2 Br – (g) Ca(g) + 2 Br(g) 2 x atomisation of Ca Ca(g) + Br 2(l) Lattice formation of Ca. Br 2 atomisation of Ca – 2125 Ca(s) + Br 2(l) formation of Ca. Br 2 (s) Hformation = 193 + 2(112) + 590 + 1150 – 2(342) – 2125 = – 652 k. J mol-1
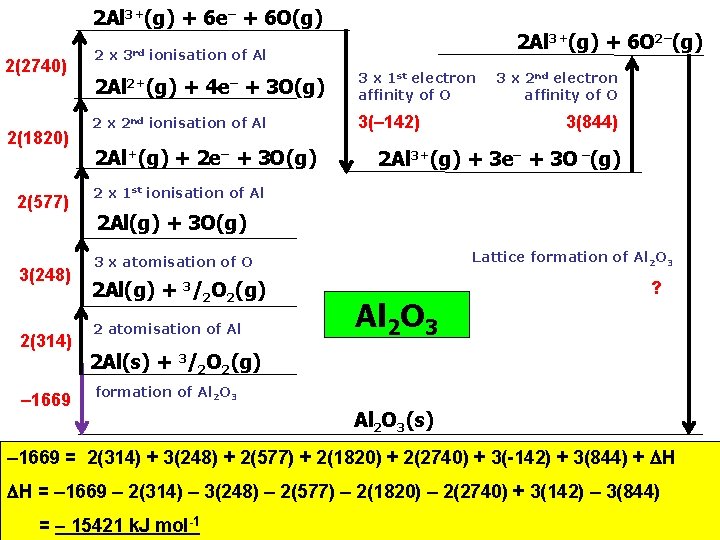
2 Al 3+(g) + 6 e– + 6 O(g) 2(2740) 2(1820) 2(577) 3(248) 2(314) – 1669 2 x 3 rd 2 Al 3+(g) + 6 O 2–(g) ionisation of Al 2+(g) + 4 e– + 3 O(g) 2 x 2 nd ionisation of Al 2 Al+(g) + 2 e– + 3 O(g) 3 x 1 st electron affinity of O 3(– 142) 3 x 2 nd electron affinity of O 3(844) 2 Al 3+(g) + 3 e– + 3 O –(g) 2 x 1 st ionisation of Al 2 Al(g) + 3 O(g) Lattice formation of Al 2 O 3 3 x atomisation of O 2 Al(g) + 3/2 O 2(g) 2 atomisation of Al Al 2 O 3 ? 2 Al(s) + 3/2 O 2(g) formation of Al 2 O 3(s) – 1669 = 2(314) + 3(248) + 2(577) + 2(1820) + 2(2740) + 3(-142) + 3(844) + H H = – 1669 – 2(314) – 3(248) – 2(577) – 2(1820) – 2(2740) + 3(142) – 3(844) = – 15421 k. J mol-1
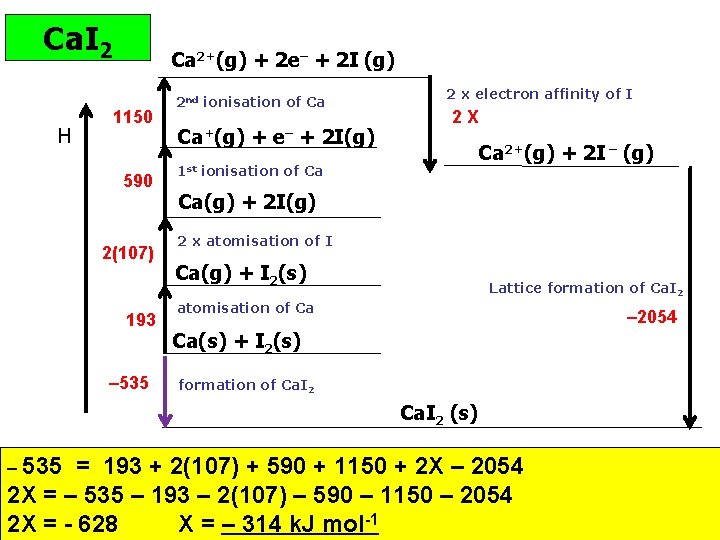
Ca. I 2 H Ca 2+(g) + 2 e– + 2 I (g) 1150 590 2(107) 193 – 535 2 nd ionisation of Ca Ca+(g) + e– + 2 I(g) 2 x electron affinity of I 2 X Ca 2+(g) + 2 I – (g) 1 st ionisation of Ca Ca(g) + 2 I(g) 2 x atomisation of I Ca(g) + I 2(s) Lattice formation of Ca. I 2 atomisation of Ca – 2054 Ca(s) + I 2(s) formation of Ca. I 2 (s) – 535 = 193 + 2(107) + 590 + 1150 + 2 X – 2054 2 X = – 535 – 193 – 2(107) – 590 – 1150 – 2054 2 X = - 628 X = – 314 k. J mol-1
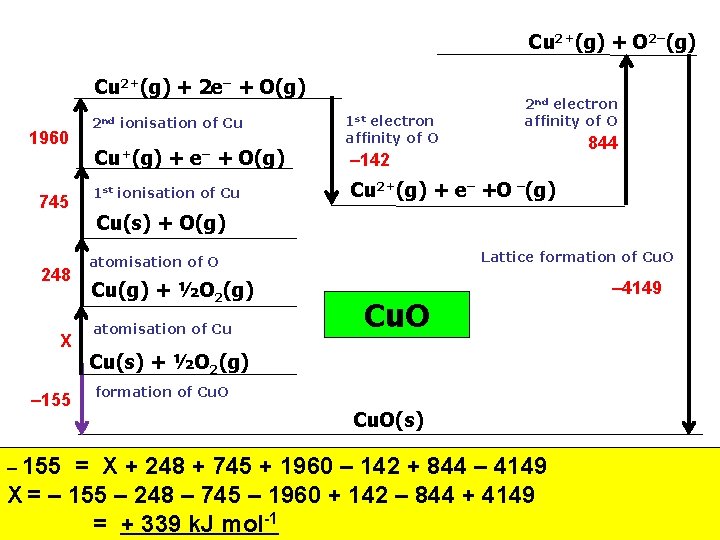
Cu 2+(g) + O 2–(g) Cu 2+(g) + 2 e– + O(g) 1960 745 248 X – 155 2 nd ionisation of Cu Cu+(g) + e– + O(g) 1 st ionisation of Cu 1 st electron affinity of O 2 nd electron affinity of O 844 – 142 Cu 2+(g) + e– +O –(g) Cu(s) + O(g) Lattice formation of Cu. O atomisation of O Cu(g) + ½O 2(g) atomisation of Cu Cu. O Cu(s) + ½O 2(g) formation of Cu. O(s) = X + 248 + 745 + 1960 – 142 + 844 – 4149 X = – 155 – 248 – 745 – 1960 + 142 – 844 + 4149 = + 339 k. J mol-1 – 4149

Ag. I Ag+ (g) + e- + I (g) H 732 1 st ionisation of Ag – 314 Ag (g) + I (g) 107 Atomisation of Ag Ag (s) + ½ I 2 (s) – 62 Ag+ (g) + I- (g) Atomisation of I Ag (s) + ½ I 2 (s) 289 1 st electron affinity of I Formation of Ag. I (s) – 62 = 289 + 107 + 732 – 314 + HLE = – 289 – 107 – 732 + 314 – 62 = – 876 k. J mol-1 ? Lattice enthalpy of formation of Ag. I
- Slides: 20