BORNHABER CYCLES BORNHABER CYCLES At the end of

BORN-HABER CYCLES

BORN-HABER CYCLES At the end of this lesson you will know The following terms: 1. Lattice Enthalpy, 2. Ionization energy, 3. Electron affinity, 4. Atomization 2. How to construct a Bohr-Haber Diagram 3. How to calculate any aspect within the diagram 4 minutes on Salt production from the its elements (Formation) https: //www. youtube. com/watch? v=w. K 9_Dtw. Pa. V 0 Berkley salt production https: //www. youtube. com/watch? v=d 2 gei. GKFve. E

Definitions The change in enthalpy when one mole of a substance in its standard state is formed from its elements in their standard states. Standard enthalpy of formation
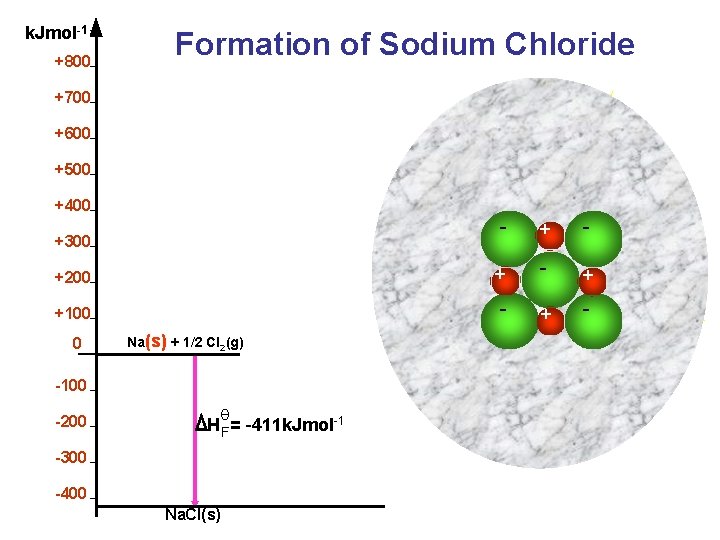
k. Jmol-1 +800 Formation of Sodium Chloride +700 +600 +500 +400 - +200 + - + +100 - +300 0 Na(s) + 1/2 Cl 2(g) -100 -200 θ H F = -411 k. Jmol-1 -300 -400 Na. Cl(s)

Definitions The energy released when one mole of an ionic compound is formed from its constituent ions in their gaseous states. Atomization Energy The energy required to break one mole of bonds between 2 atoms Is called Bond Energy for Cl 2 it is 244 k. J/mol. Cl 2 2 Cl I am sure you can see the PROBLEM here , if you look above
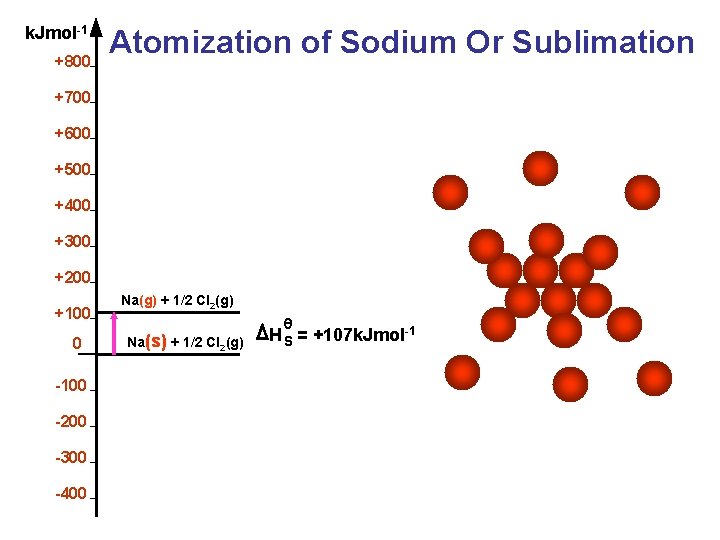
k. Jmol-1 +800 Atomization of Sodium Or Sublimation +700 +600 +500 +400 +300 +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl (g) 2 θ H S = +107 k. Jmol-1
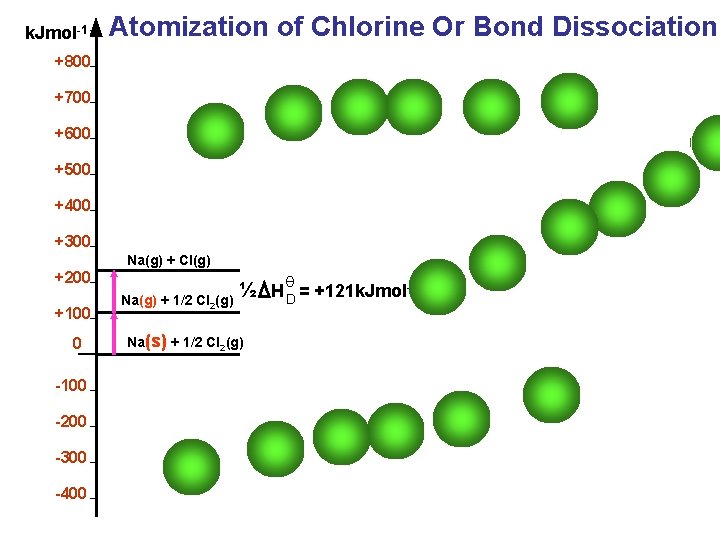
k. Jmol-1 Atomization of Chlorine Or Bond Dissociation +800 +700 +600 +500 +400 +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) θ ½ H D = +121 k. Jmol-1 Na(s) + 1/2 Cl 2(g)

Definitions The energy required to remove the outermost electron of each atom in one mole of an element in its gaseous state. X (g) X+ (g) + e − First ionization energy
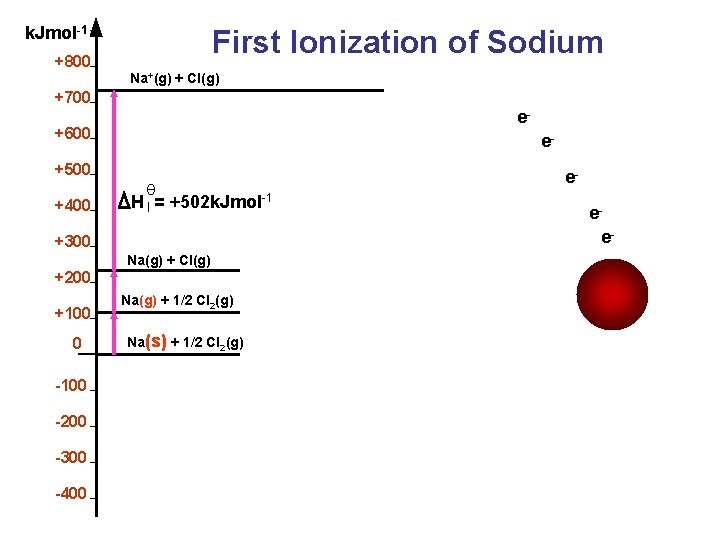
k. Jmol-1 +800 First Ionization of Sodium Na+(g) + Cl(g) +700 e- +600 e- +500 +400 θ H I = +502 k. Jmol-1 +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) eee+

Definitions The energy released or absorbed when an electron is added to each atom in one mole of a substance in its gaseous state. X (g) + e − → X− (g) Electron affinity
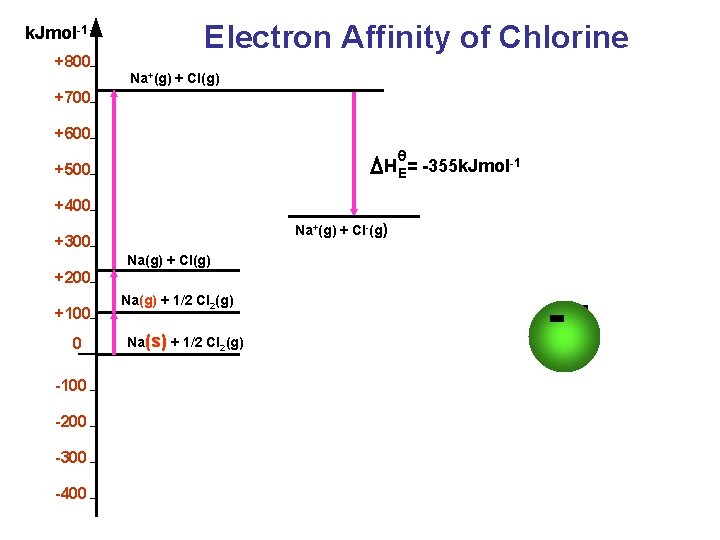
k. Jmol-1 +800 Electron Affinity of Chlorine Na+(g) + Cl(g) +700 +600 θ H E= -355 k. Jmol-1 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 -100 -200 -300 -400 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) -e
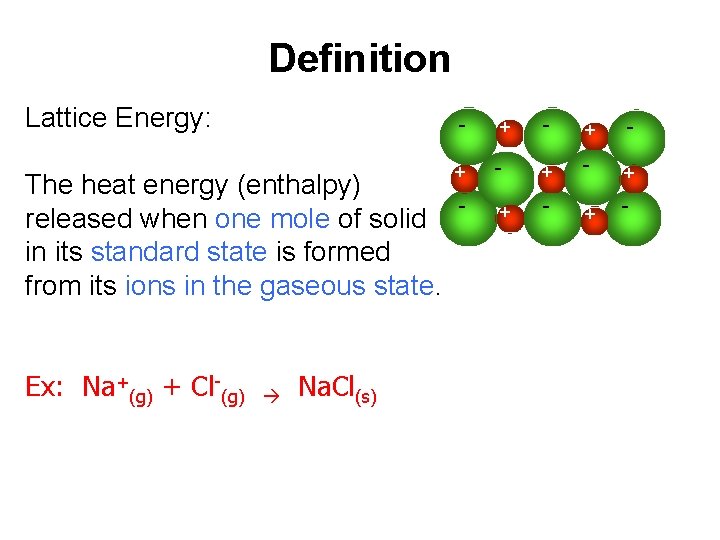
Definition Lattice Energy: The heat energy (enthalpy) released when one mole of solid in its standard state is formed from its ions in the gaseous state. Ex: Na+(g) + Cl-(g) Na. Cl(s) - + - + - + - - +
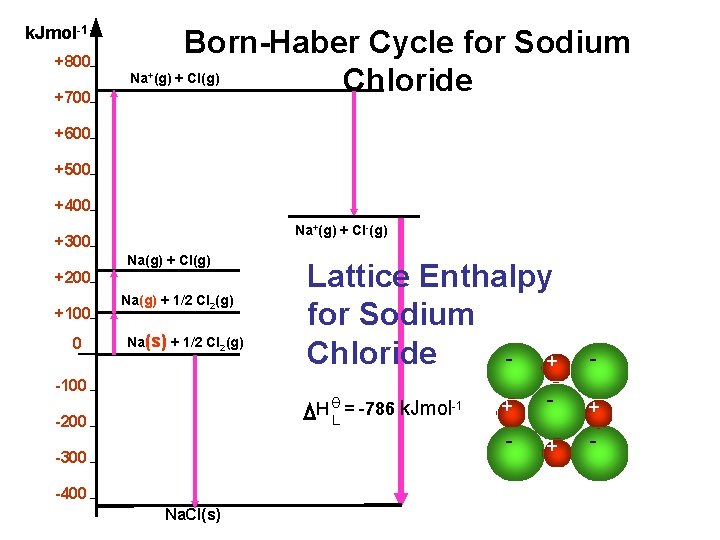
k. Jmol-1 +800 +700 Born-Haber Cycle for Sodium Na (g) + Cl(g) Chloride + +600 +500 +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) -100 Lattice Enthalpy for Sodium Chloride + - + - θ H = -786 k. Jmol-1 L -200 -300 -400 Na. Cl(s)
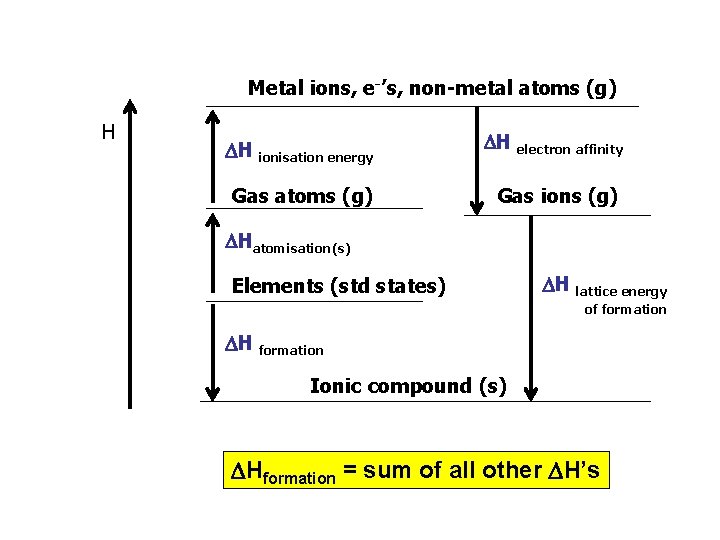
Metal ions, e-’s, non-metal atoms (g) H H ionisation energy H electron affinity Gas atoms (g) Gas ions (g) Hatomisation(s) Elements (std states) H lattice energy of formation H formation Ionic compound (s) Hformation = sum of all other H’s
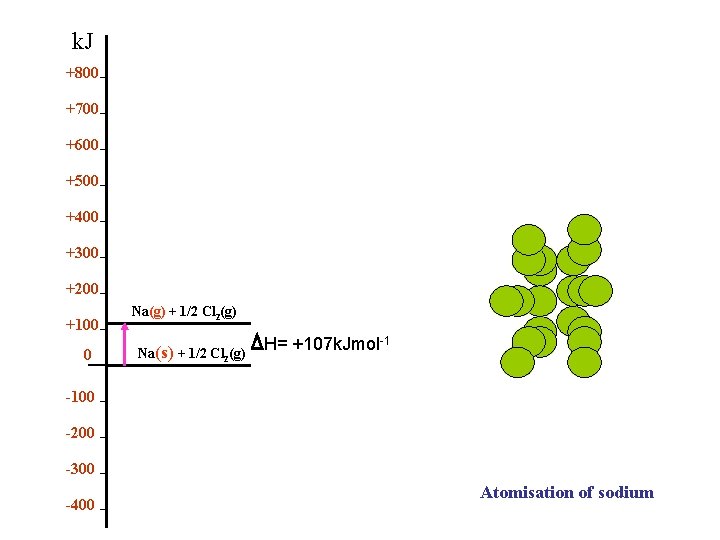
k. J +800 +700 +600 +500 +400 +300 +200 +100 0 Na(g) + 1/2 Cl 2(g) Na(s) + 1/2 Cl 2(g) H= +107 k. Jmol-1 -100 -200 -300 -400 Atomisation of sodium
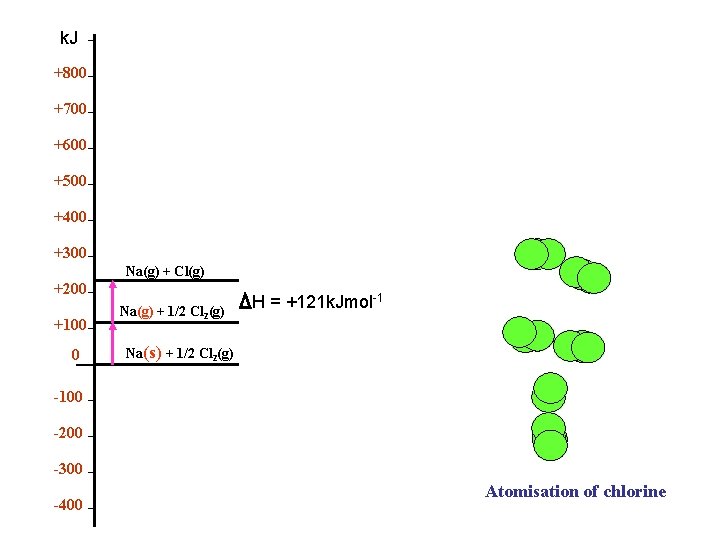
k. J +800 +700 +600 +500 +400 +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) H = +121 k. Jmol-1 Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 Atomisation of chlorine
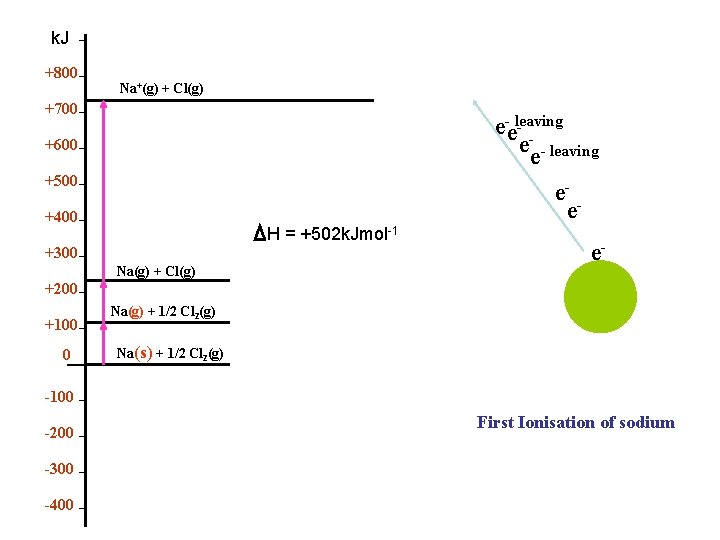
k. J +800 Na+(g) + Cl(g) +700 e-eleaving e-e- leaving e- e +600 +500 +400 H = +502 k. Jmol-1 +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) e+ Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 First Ionisation of sodium
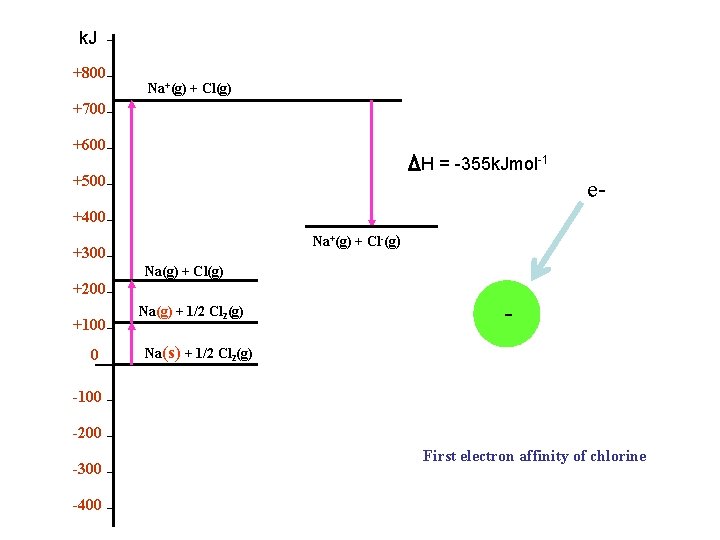
k. J +800 Na+(g) + Cl(g) +700 +600 H = -355 k. Jmol-1 +500 e- +400 Na+(g) + Cl-(g) +300 Na(g) + Cl(g) +200 +100 0 Na(g) + 1/2 Cl 2(g) - e- Na(s) + 1/2 Cl 2(g) -100 -200 -300 -400 First electron affinity of chlorine
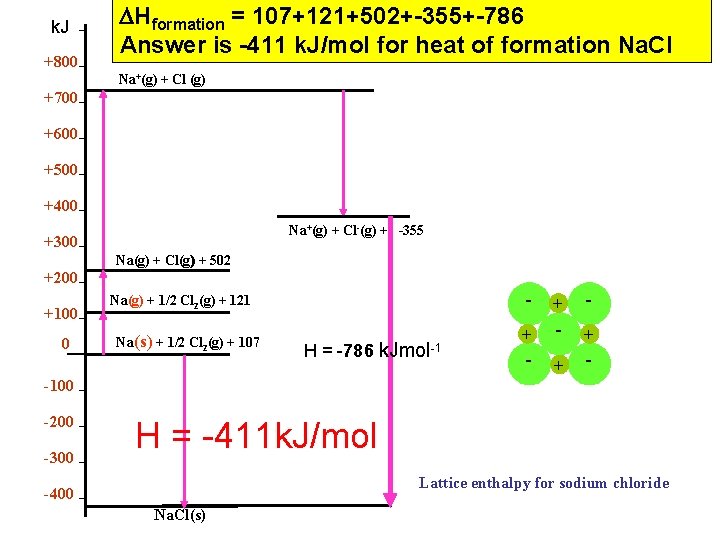
k. J +800 Hformation = 107+121+502+-355+-786 Answer is -411 k. J/mol for heat of formation Na. Cl Na+(g) + Cl (g) +700 +600 +500 +400 Na+(g) + Cl-(g) + -355 +300 Na(g) + Cl(g) + 502 +200 +100 0 Na(g) + 1/2 Cl 2(g) + 121 Na(s) + 1/2 Cl 2(g) + 107 H = -786 k. Jmol -1 - + - + - -100 -200 -300 H = -411 k. J/mol Lattice enthalpy for sodium chloride -400 Na. Cl(s)

Ok, so we just found the heat of formation for Na. Cl Using Born-Haber. Now lets try another (Ca. O) and find something different like Lattice Energy.
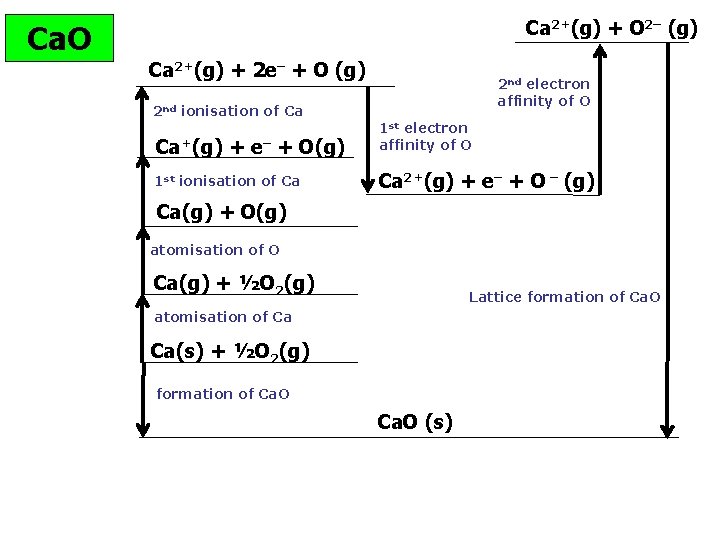
Ca. O Ca 2+(g) + O 2– (g) Ca 2+(g) + 2 e– + O (g) 2 nd ionisation of Ca 2 nd electron affinity of O Ca+(g) + e– + O(g) 1 st electron affinity of O 1 st ionisation of Ca Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca Ca(s) + ½O 2(g) formation of Ca. O (s)

Ca 2+(g) + O 2– (g) Ca. O Ca 2+(g) + 2 e– + O (g) 1150 2 nd ionisation of Ca Ca+(g) 590 248 + e– + O(g) 1 st ionisation of Ca 2 nd electron affinity of O – 142 1 st electron affinity of O 844 Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca 193 – 635 X Ca(s) + ½O 2(g) formation of Ca. O (s) Hf Ca. O= – 635; Atomisation calcium = +193; Atomisation oxygen = +248 1 st IE Ca = +590; 2 nd IE Ca = +1150 1 st e- affinity of oxygen = – 142 2 nd e- affinity= +844

Ca 2+(g) + O 2– (g) Ca. O Ca 2+(g) + 2 e– + O (g) 1150 2 nd ionisation of Ca Ca+(g) 590 248 + e– + O(g) 1 st ionisation of Ca – 142 1 st electron affinity of O – 635 844 Ca 2+(g) + e– + O – (g) Ca(g) + O(g) atomisation of O Ca(g) + ½O 2(g) Lattice formation of Ca. O atomisation of Ca 193 2 nd electron affinity of O X Ca(s) + ½O 2(g) formation of Ca. O (s) = 193 + 248 + 590 + 1150 – 142 + 844 + Hlattice = – 635 – 193 – 248 – 590 – 1150 + 142 – 844 = – 3518 k. J mol-1 © www. chemsheets. co. uk A 2 1015 19 -May-2016 – 635

Ok, so we just found the heat of Lattice Energy For Ca. O Now try Mg. Br 2 and Find it’s Lattice energy By drawing and Using Born-Haber Diagram.

Mg. Br 2 Mg 2+(g) + 2 e– + 2 Br (g) 2 nd ionisation of Mg Mg+(g) + e– + 2 Br(g) 1 st ionisation of Mg 2 x electron affinity of Br Mg 2+(g) 2 Br – (g) Mg(g) + 2 Br(g) 2 x atomisation of Br Mg(g) + Br 2(l) Lattice formation of Mg. Br 2 atomisation of Mg Mg(s) + Br 2(l) formation of Mg. Br 2 (s)

Mg. Br 2 Mg 2+(g) + 2 e– + 2 Br (g) 1450 736 2(112) 150 X 2 x electron affinity of Br 2 nd ionisation of Mg Mg+(g) + e– + 2 Br(g) 1 st ionisation of Mg 2(– 342) Mg 2+(g) 2 Br – (g) Mg(g) + 2 Br(g) 2 x atomisation of Br Mg(g) + Br 2(l) Lattice formation of Mg. Br 2 atomisation of Mg – 2394 Mg(s) + Br 2(l) formation of Mg. Br 2 (s) Hformation = 150 + 2(112) + 736 + 1450 + 2(– 342) – 2394 Hformation = – 518 k. J mol-1 © www. chemsheets. co. uk A 2 1015 19 -May-2016

Next, find Lattice energy for Na 2 S
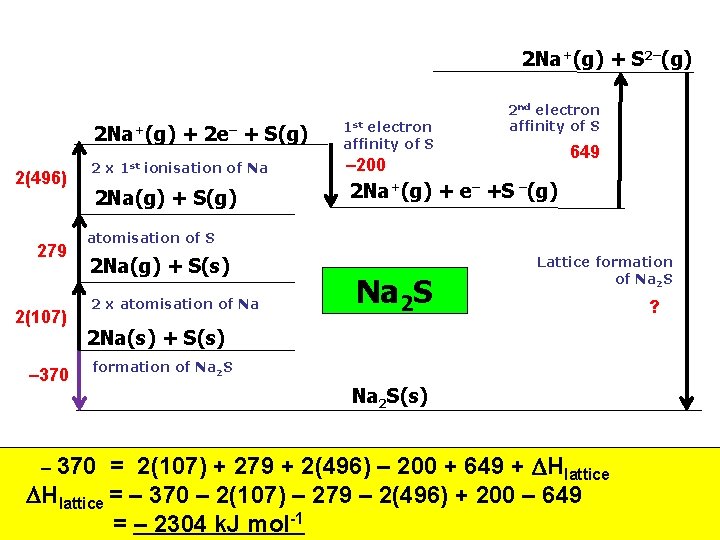
2 Na+(g) + S 2–(g) 2 Na+(g) 2(496) 279 2(107) – 370 2 x 1 st + 2 e– + S(g) ionisation of Na 2 Na(g) + S(g) 1 st electron affinity of S 2 nd electron affinity of S 649 – 200 2 Na+(g) + e– +S –(g) atomisation of S 2 Na(g) + S(s) 2 x atomisation of Na Na 2 S Lattice formation of Na 2 S 2 Na(s) + S(s) formation of Na 2 S(s) = 2(107) + 279 + 2(496) – 200 + 649 + Hlattice = – 370 – 2(107) – 279 – 2(496) + 200 – 649 = – 2304 k. J mol-1 – 370 ?

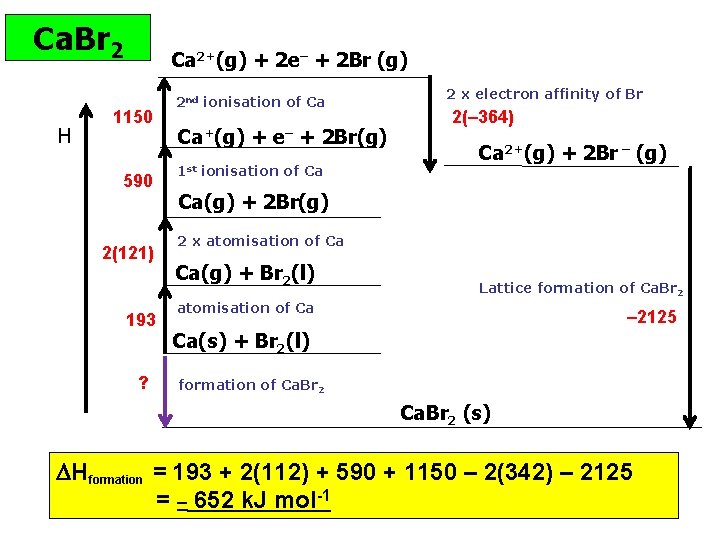
Ca. Br 2 H Ca 2+(g) + 2 e– + 2 Br (g) 1150 590 2(121) 193 ? 2 nd ionisation of Ca Ca+(g) + e– + 2 Br(g) 1 st ionisation of Ca 2 x electron affinity of Br 2(– 364) Ca 2+(g) + 2 Br – (g) Ca(g) + 2 Br(g) 2 x atomisation of Ca Ca(g) + Br 2(l) Lattice formation of Ca. Br 2 atomisation of Ca – 2125 Ca(s) + Br 2(l) formation of Ca. Br 2 (s) Hformation = 193 + 2(112) + 590 + 1150 – 2(342) – 2125 = – 652 k. J mol-1
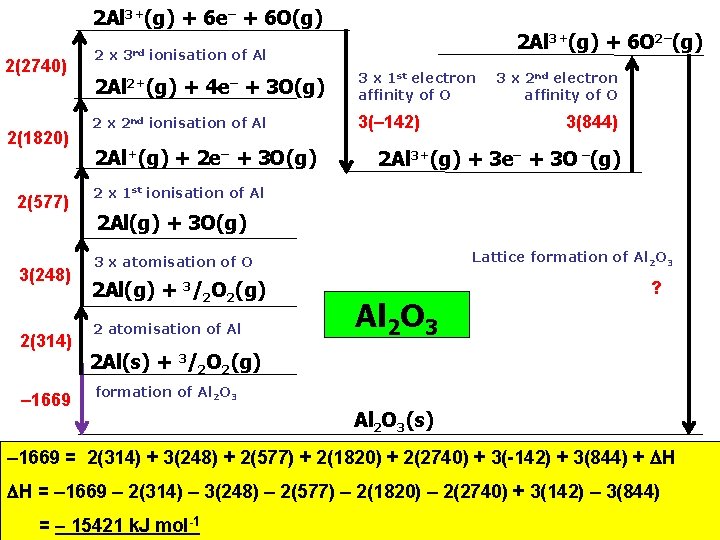
2 Al 3+(g) + 6 e– + 6 O(g) 2(2740) 2(1820) 2(577) 3(248) 2(314) – 1669 2 x 3 rd 2 Al 3+(g) + 6 O 2–(g) ionisation of Al 2+(g) + 4 e– + 3 O(g) 2 x 2 nd ionisation of Al 2 Al+(g) + 2 e– + 3 O(g) 3 x 1 st electron affinity of O 3(– 142) 3 x 2 nd electron affinity of O 3(844) 2 Al 3+(g) + 3 e– + 3 O –(g) 2 x 1 st ionisation of Al 2 Al(g) + 3 O(g) Lattice formation of Al 2 O 3 3 x atomisation of O 2 Al(g) + 3/2 O 2(g) 2 atomisation of Al Al 2 O 3 ? 2 Al(s) + 3/2 O 2(g) formation of Al 2 O 3(s) – 1669 = 2(314) + 3(248) + 2(577) + 2(1820) + 2(2740) + 3(-142) + 3(844) + H H = – 1669 – 2(314) – 3(248) – 2(577) – 2(1820) – 2(2740) + 3(142) – 3(844) = – 15421 k. J mol-1
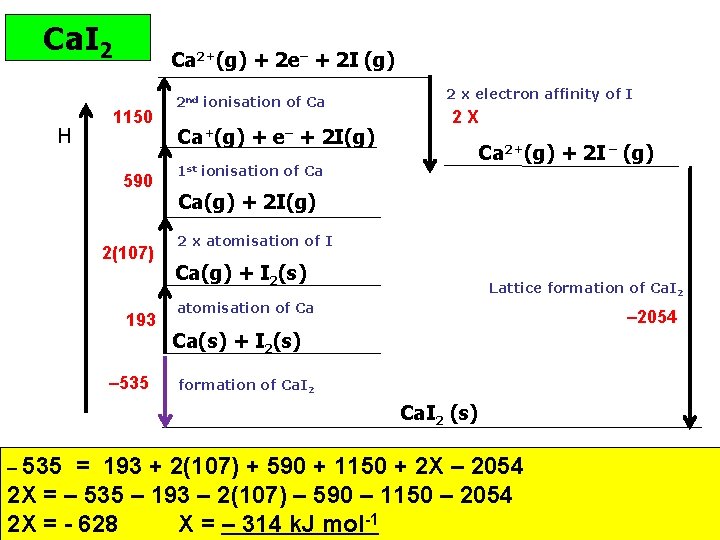
Ca. I 2 H Ca 2+(g) + 2 e– + 2 I (g) 1150 590 2(107) 193 – 535 2 nd ionisation of Ca Ca+(g) + e– + 2 I(g) 2 x electron affinity of I 2 X Ca 2+(g) + 2 I – (g) 1 st ionisation of Ca Ca(g) + 2 I(g) 2 x atomisation of I Ca(g) + I 2(s) Lattice formation of Ca. I 2 atomisation of Ca – 2054 Ca(s) + I 2(s) formation of Ca. I 2 (s) – 535 = 193 + 2(107) + 590 + 1150 + 2 X – 2054 2 X = – 535 – 193 – 2(107) – 590 – 1150 – 2054 2 X = - 628 X = – 314 k. J mol-1
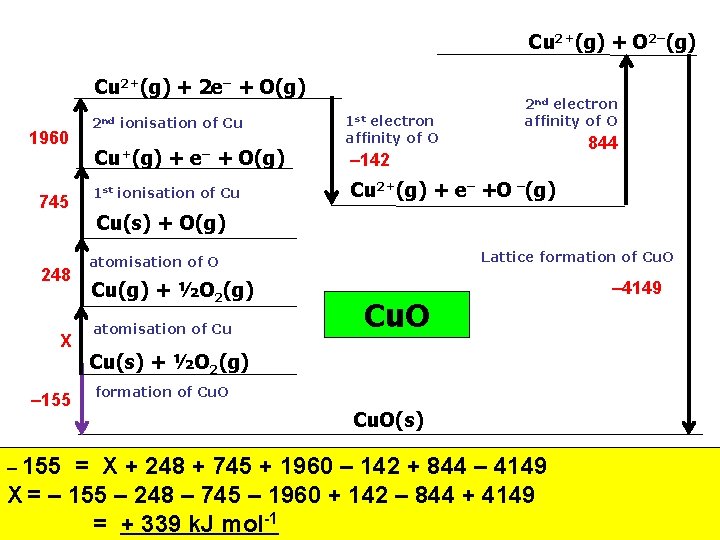
Cu 2+(g) + O 2–(g) Cu 2+(g) + 2 e– + O(g) 1960 745 248 X – 155 2 nd ionisation of Cu Cu+(g) + e– + O(g) 1 st ionisation of Cu 1 st electron affinity of O 2 nd electron affinity of O 844 – 142 Cu 2+(g) + e– +O –(g) Cu(s) + O(g) Lattice formation of Cu. O atomisation of O Cu(g) + ½O 2(g) atomisation of Cu Cu. O Cu(s) + ½O 2(g) formation of Cu. O(s) = X + 248 + 745 + 1960 – 142 + 844 – 4149 X = – 155 – 248 – 745 – 1960 + 142 – 844 + 4149 = + 339 k. J mol-1 – 4149

Ag. I Ag+ (g) + e- + I (g) H 732 1 st ionisation of Ag – 314 Ag (g) + I (g) 107 Atomisation of Ag Ag (s) + ½ I 2 (s) – 62 Ag+ (g) + I- (g) Atomisation of I Ag (s) + ½ I 2 (s) 289 1 st electron affinity of I Formation of Ag. I (s) – 62 = 289 + 107 + 732 – 314 + HLE = – 289 – 107 – 732 + 314 – 62 = – 876 k. J mol-1 ? Lattice enthalpy of formation of Ag. I

BORN-HABER CYCLES You should now know The following terms: 1. A) Lattice Enthalpy, B) Ionization energy, C) Electron affinity, D) Atomization 2. How to construct a Bohr-Haber Diagram 3. How to calculate any aspect within the diagram
- Slides: 35