BORNHABER CYCLES A guide for A level students

BORN-HABER CYCLES A guide for A level students KNOCKHARDY PUBLISHING 2008 SPECIFICATIONS

BORN-HABER CYCLES INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A 2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use the material at home for revision purposes or it may be used for classroom teaching using an interactive white board. Accompanying notes on this, and the full range of AS and A 2 topics, are available from the KNOCKHARDY SCIENCE WEBSITE at. . . www. knockhardy. org. uk/sci. htm Navigation is achieved by. . . either clicking on the grey arrows at the foot of each page or using the left and right arrow keys on the keyboard

BORN-HABER CYCLES CONTENTS • Lattice Enthalpy • Definition of enthalpy changes • Born-Haber cycle for sodium chloride • Calculation of Lattice Enthalpy • Born-Haber cycle for magnesium chloride

Lattice Enthalpy Definition(s) THERE ARE TWO DEFINITIONS OF LATTICE ENTHALPY 1. Lattice Formation Enthalpy ‘The enthalpy change when ONE MOLE of an ionic lattice is formed from its isolated gaseous ions. ’ Example Na+(g) + Cl¯(g) Na+ Cl¯(s) 2. Lattice Dissociation Enthalpy ‘The enthalpy change when ONE MOLE of an ionic lattice dissociates into isolated gaseous ions. ’ Example Na+ Cl¯(s) Na+(g) + Cl¯(g) MAKE SURE YOU CHECK WHICH IS BEING USED

Lattice Enthalpy Definition(s) 1. Lattice Formation Enthalpy ‘The enthalpy change when ONE MOLE of an ionic lattice is formed from its isolated gaseous ions. ’ Values Example highly EXOTHERMIC strong electrostatic attraction between oppositely charged ions a lot of energy is released as the bond is formed relative values are governed by the charge density of the ions. Na+(g) + Cl¯(g) Na+(g) + Cl–(g) Na. Cl(s) Na+ Cl¯(s)

Lattice Enthalpy Definition(s) 2. Lattice Dissociation Enthalpy ‘The enthalpy change when ONE MOLE of an ionic lattice dissociates into isolated gaseous ions. ’ Values Example highly ENDOTHERMIC strong electrostatic attraction between oppositely charged ions a lot of energy must be put in to overcome the attraction relative values are governed by the charge density of the ions. Na+ Cl¯(s) Na+(g) + Cl–(g) Na. Cl(s) + Cl¯(g)

Calculating Lattice Enthalpy SPECIAL POINTS you CANNOT MEASURE LATTICE ENTHALPY DIRECTLY it is CALCULATED USING A BORN-HABER CYCLE

Calculating Lattice Enthalpy SPECIAL POINTS you CANNOT MEASURE LATTICE ENTHALPY DIRECTLY it is CALCULATED USING A BORN-HABER CYCLE greater charge densities of ions = greater attraction = larger lattice enthalpy

Calculating Lattice Enthalpy SPECIAL POINTS you CANNOT MEASURE LATTICE ENTHALPY DIRECTLY it is CALCULATED USING A BORN-HABER CYCLE greater charge densities of ions = greater attraction = larger lattice enthalpy Effects Melting point the higher the lattice enthalpy, the higher the melting point of an ionic compound Solubility solubility of ionic compounds is affected by the relative values of Lattice and Hydration Enthalpies

Lattice Enthalpy Values Cl¯ Br¯ F¯ O 2 - Na+ -780 -742 -918 -2478 K+ -711 -679 -817 -2232 Rb+ -685 -656 -783 Mg 2+ -2256 Ca 2+ -2259 -3791 Units: k. J mol-1 Smaller ions will have a greater attraction for each other because of their higher charge density. They will have larger Lattice Enthalpies and larger melting points because of the extra energy which must be put in to separate the oppositely charged ions.

Lattice Enthalpy Values Cl¯ Br¯ F¯ O 2 - Na+ -780 -742 -918 -2478 K+ -711 -679 -817 -2232 Rb+ -685 -656 -783 Mg 2+ -2256 Ca 2+ -2259 -3791 Smaller ions will have a greater attraction for each other because of their higher charge density. They will have larger Lattice Enthalpies and larger melting points because of the extra energy which must be put in to separate the oppositely charged ions. Na+ Cl¯ K+ Cl¯ The sodium ion has the same charge as a potassium ion but is smaller. It has a higher charge density so will have a more effective attraction for the chloride ion. More energy will be released when they come together.

Born-Haber Cycle For Sodium Chloride k. J mol-1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> Na. Cl(s) – 411 Enthalpy of sublimation of sodium Na(s) + 108 Enthalpy of atomisation of chlorine ½Cl 2(g) ——> Cl(g) + 121 Ist Ionisation Energy of sodium Na(g) ——> Na+(g) + e¯ + 500 Electron Affinity of chlorine Cl(g) + e¯ ——> Cl¯(g) – 364 Na+(g) + Cl¯(g) ——> Na. Cl(s) ? Lattice Enthalpy of Na. Cl ——> Na(g)
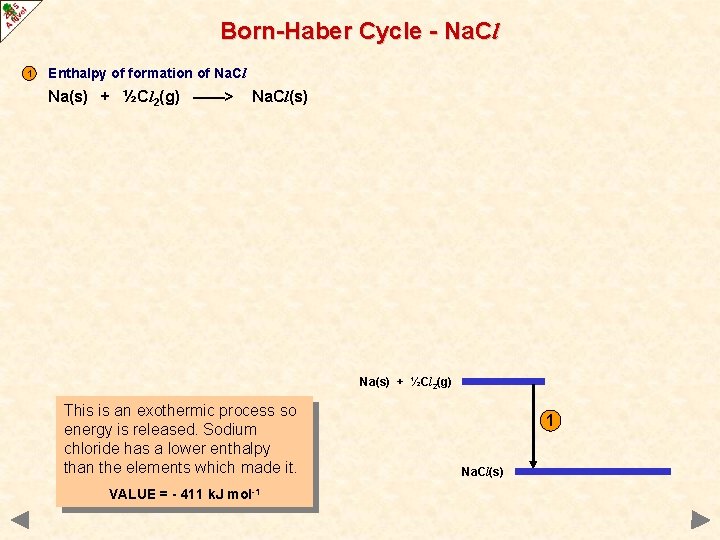
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> Na. Cl(s) Na(s) + ½Cl 2(g) This is an exothermic process so energy is released. Sodium chloride has a lower enthalpy than the elements which made it. VALUE = - 411 k. J mol-1 1 Na. Cl(s)
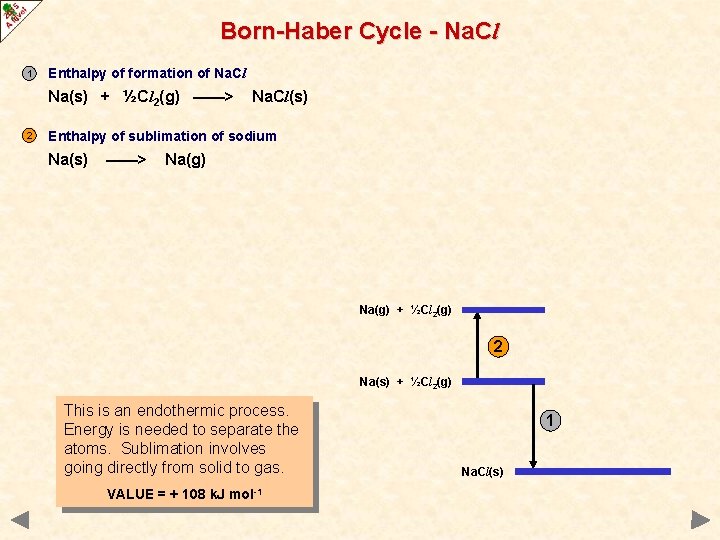
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> 2 Na. Cl(s) Enthalpy of sublimation of sodium Na(s) ——> Na(g) + ½Cl 2(g) 2 Na(s) + ½Cl 2(g) This is an endothermic process. Energy is needed to separate the atoms. Sublimation involves going directly from solid to gas. VALUE = + 108 k. J mol-1 1 Na. Cl(s)
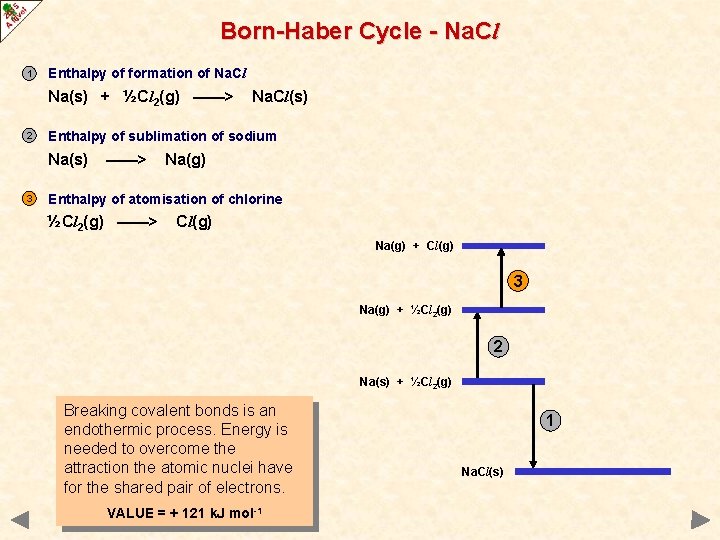
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> 2 Enthalpy of sublimation of sodium Na(s) 3 Na. Cl(s) ——> Na(g) Enthalpy of atomisation of chlorine ½Cl 2(g) ——> Cl(g) Na(g) + Cl(g) 3 Na(g) + ½Cl 2(g) 2 Na(s) + ½Cl 2(g) Breaking covalent bonds is an endothermic process. Energy is needed to overcome the attraction the atomic nuclei have for the shared pair of electrons. VALUE = + 121 k. J mol-1 1 Na. Cl(s)
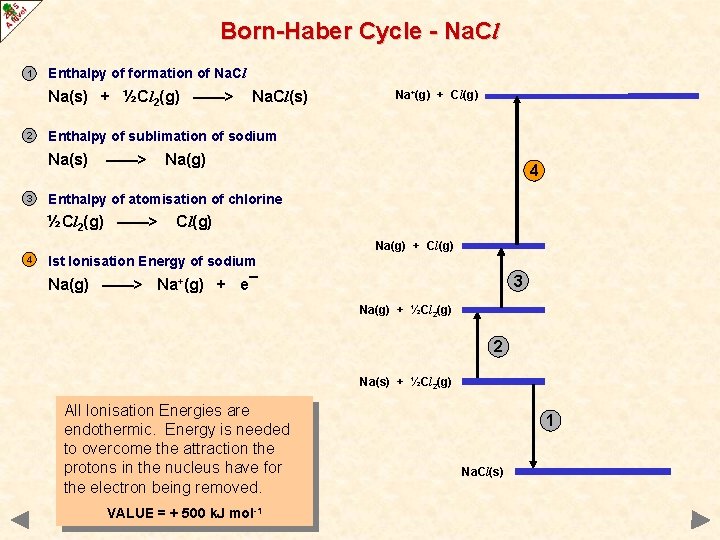
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> 2 Na+(g) + Cl(g) Enthalpy of sublimation of sodium Na(s) 3 Na. Cl(s) ——> Na(g) 4 Enthalpy of atomisation of chlorine ½Cl 2(g) ——> Cl(g) Na(g) + Cl(g) 4 Ist Ionisation Energy of sodium 3 Na(g) ——> Na+(g) + e¯ Na(g) + ½Cl 2(g) 2 Na(s) + ½Cl 2(g) All Ionisation Energies are endothermic. Energy is needed to overcome the attraction the protons in the nucleus have for the electron being removed. VALUE = + 500 k. J mol-1 1 Na. Cl(s)
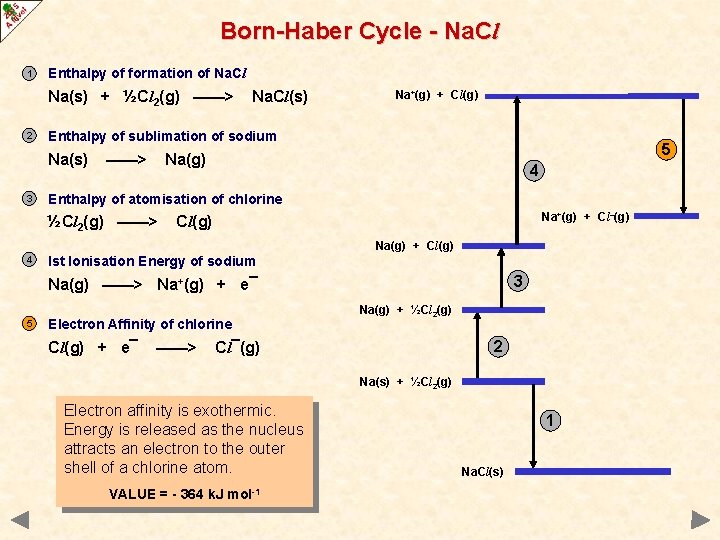
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> 2 Na+(g) + Cl(g) Enthalpy of sublimation of sodium Na(s) 3 Na. Cl(s) ——> 5 Na(g) 4 Enthalpy of atomisation of chlorine ½Cl 2(g) ——> Na+(g) + Cl–(g) Cl(g) Na(g) + Cl(g) 4 Ist Ionisation Energy of sodium 3 Na(g) ——> Na+(g) + e¯ 5 Electron Affinity of chlorine Cl(g) + e¯ ——> Na(g) + ½Cl 2(g) 2 Cl¯(g) Na(s) + ½Cl 2(g) Electron affinity is exothermic. Energy is released as the nucleus attracts an electron to the outer shell of a chlorine atom. VALUE = - 364 k. J mol-1 1 Na. Cl(s)
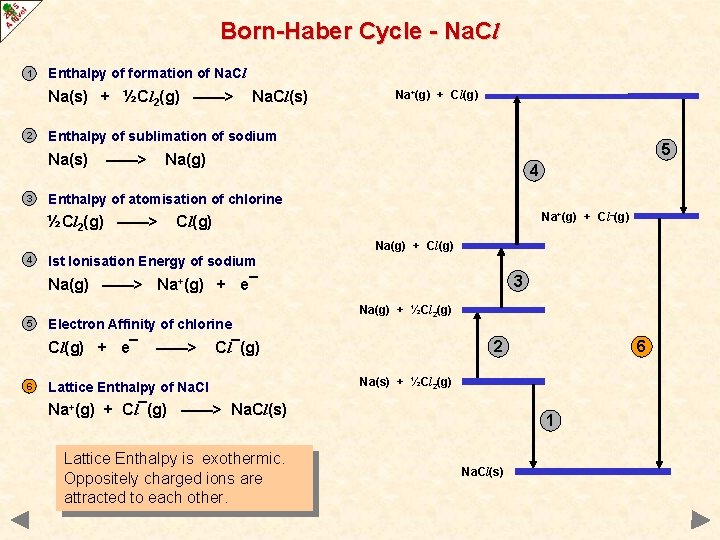
Born-Haber Cycle - Na. Cl 1 Enthalpy of formation of Na. Cl Na(s) + ½Cl 2(g) ——> 2 Na+(g) + Cl(g) Enthalpy of sublimation of sodium Na(s) 3 Na. Cl(s) ——> 5 Na(g) 4 Enthalpy of atomisation of chlorine ½Cl 2(g) ——> Na+(g) + Cl–(g) Cl(g) Na(g) + Cl(g) 4 Ist Ionisation Energy of sodium 3 Na(g) ——> Na+(g) + e¯ 5 Electron Affinity of chlorine Cl(g) + e¯ 6 ——> Na(g) + ½Cl 2(g) 2 Cl¯(g) Lattice Enthalpy of Na. Cl Na(s) + ½Cl 2(g) Na+(g) + Cl¯(g) ——> Na. Cl(s) Lattice Enthalpy is exothermic. Oppositely charged ions are attracted to each other. 6 1 Na. Cl(s)
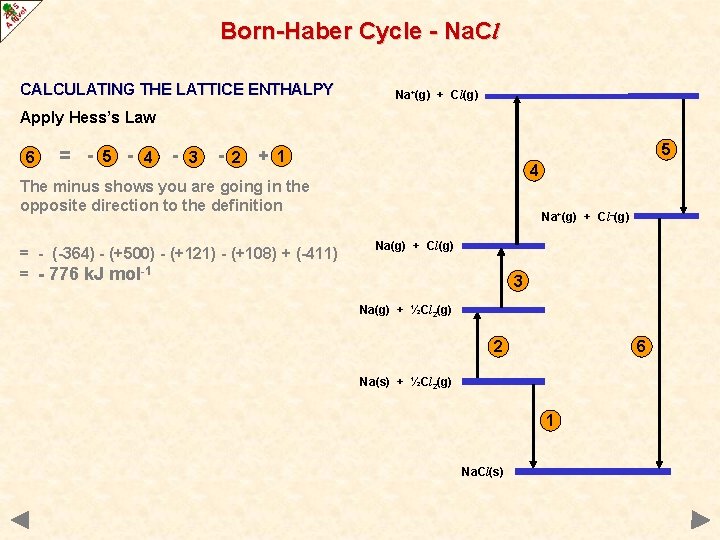
Born-Haber Cycle - Na. Cl CALCULATING THE LATTICE ENTHALPY Na+(g) + Cl(g) Apply Hess’s Law 6 = - 5 - 4 - 3 - 2 5 + 1 4 The minus shows you are going in the opposite direction to the definition = - (-364) - (+500) - (+121) - (+108) + (-411) = - 776 k. J mol-1 Na+(g) + Cl–(g) Na(g) + Cl(g) 3 Na(g) + ½Cl 2(g) 2 6 Na(s) + ½Cl 2(g) 1 Na. Cl(s)
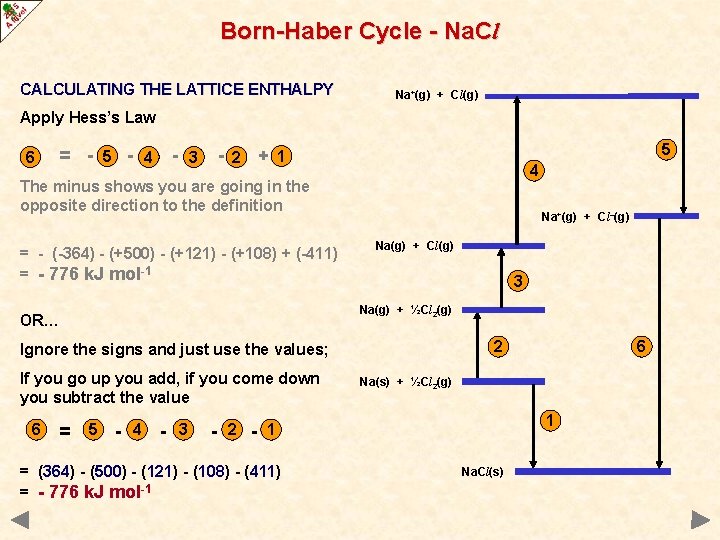
Born-Haber Cycle - Na. Cl CALCULATING THE LATTICE ENTHALPY Na+(g) + Cl(g) Apply Hess’s Law 6 = - - 5 - 4 3 - 5 + 1 2 4 The minus shows you are going in the opposite direction to the definition = - (-364) - (+500) - (+121) - (+108) + (-411) = - 776 k. J mol-1 Na+(g) + Cl–(g) Na(g) + Cl(g) 3 Na(g) + ½Cl 2(g) OR… 2 Ignore the signs and just use the values; If you go up you add, if you come down you subtract the value 6 = 5 - 4 - 3 - 2 - Na(s) + ½Cl 2(g) 1 1 = (364) - (500) - (121) - (108) - (411) = - 776 k. J mol-1 6 Na. Cl(s)
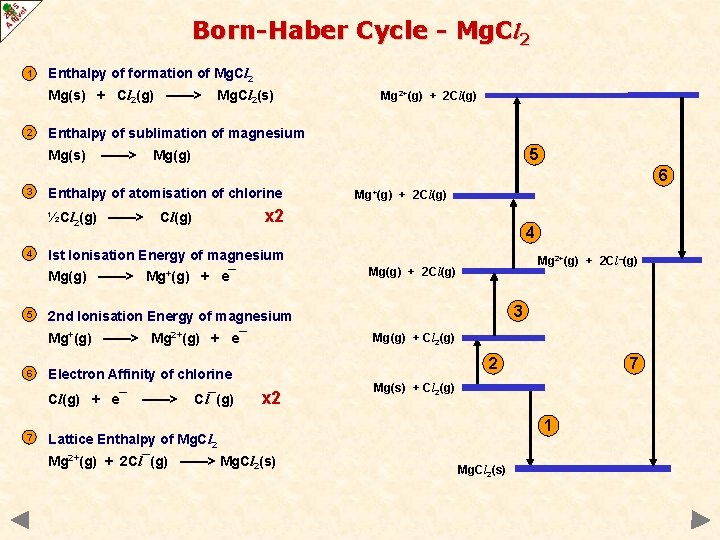
Born-Haber Cycle - Mg. Cl 2 1 Enthalpy of formation of Mg. Cl 2 Mg(s) + Cl 2(g) ——> 2 Mg. Cl 2(s) Mg 2+(g) + 2 Cl(g) Enthalpy of sublimation of magnesium Mg(s) ——> 5 Mg(g) 6 3 Enthalpy of atomisation of chlorine ½Cl 2(g) ——> 4 Mg 2+(g) + 2 Cl–(g) Mg(g) + 2 Cl(g) 3 2 nd Ionisation Energy of magnesium Mg(g) + Cl 2(g) 2 Electron Affinity of chlorine Cl(g) + e¯ 7 4 Ist Ionisation Energy of magnesium Mg+(g) ——> Mg 2+(g) + e¯ 6 + 2 Cl(g) x 2 Cl(g) Mg(g) ——> Mg+(g) + e¯ 5 Mg+(g) ——> Cl¯(g) x 2 Mg(s) + Cl 2(g) 1 Lattice Enthalpy of Mg. Cl 2 Mg 2+(g) + 2 Cl¯(g) ——> Mg. Cl 2(s) 7 Mg. Cl 2(s)

BORN-HABER CYCLES THE END KNOCKHARDY PUBLISHING
- Slides: 22