Bones SKELETAL SYSTEM Include all bones 206 in
Bones

SKELETAL SYSTEM: Include all bones (206) in the body associated cartilage and joint. Functions: 1. Support and movement 2. Protection 3. Mineral storage 4. Electrolyte balance 5. Formation of blood cell 6. Detoxification by absorbing heavy minerals

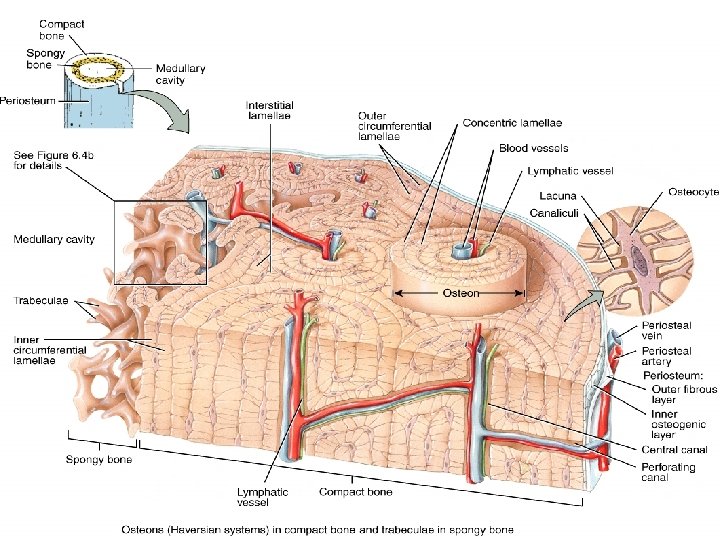
Composition of Bone compact and spongy bones are composed of bone cells and collagenous organic matrix (calcium and phosphate that deposited as a complex inorganic hydrated hydroxyapatites) which gives the strength of the bone. Type of bone cells: 1. Osteoclasts: Bone destroying cells “C” means chewing 2. Osteoblasts: Bone generating cells “B” means building 3. Osteocytes: Mature bone cells, spider shaped and maintain bone tissue
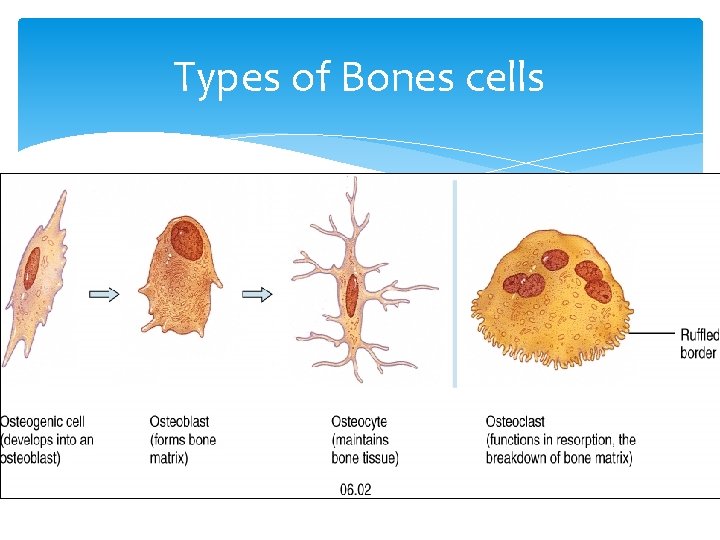
Types of Bones cells

Control of Bone Growth • Minerals, vitamins, and hormones influence growing of the bone. • The specific hormones which affect growth are growth hormone (GH), thyroid hormone (T 3 and T 4), and the sex steroids (estrogen and tesstosterone). • The growing of the bone increasing until adulthood then start remodeling constantly. • Each week we turn over about 5% of our bone mass.

Bone profile Bone Profile test which measures proteins, minerals and enzymes involved in bone turnover and can indicate problems with bone. Specimen Serum in gold-top vacutainer Serum: Calcium, Phosphate, ALP, Mg, Vitamin D. Plasma: PTH

Parathyroid hormon Parathyroid gland secrete PTH into the bloodstream in response to low blood calcium levels. Function: takes calcium from the body's bone stimulates the activation of vitamin D in the kidney Increase reabsorption of Ca and increase seretion of phosphate As calcium levels begin to increase in the blood, PTH normally decreases. Normal values are 10 - 55 (pg/m. L).

Excess PTH secretion may be due to • kidney failure and rickets • benign parathyroid tumor • vitamin D deficiency Low levels of PTH may be due to conditions causing • low levels of magnesium • abnormality in PTG

Vitamin D The main role of Vitamin D is to help regulate the absorption of calcium, phosphorus, and magnesium and helps form and maintain bones. These happen by active forms of vitamin D 1, 25 -dihydroxycholecalciferol (calcitriol) Hydroxylation of vitamin D by (25 - hydroxylase) in the liver and (1αhydroxylase) in the kidney. The normal range is 30. 0 to 74. 0 (ng/m. L).
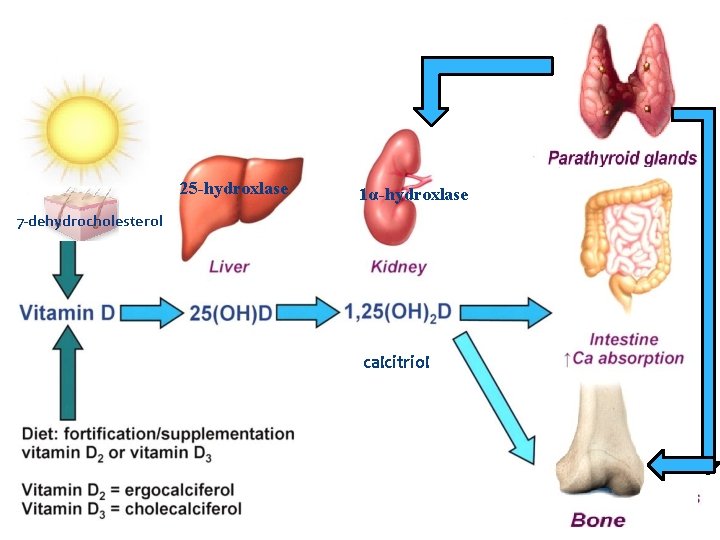
25 -hydroxlase 1α-hydroxlase 7 -dehydrocholesterol calcitriol
Vitamin D deficiency can result from inadequate intake inadequate sunlight exposure limit its absorption without it, bones will be soft, malformed, and unable to repair themselves normally, resulting in diseases called rickets in children and osteomalacia in adults. Increase amount of Vitamin D will be toxic when we take overdosing of supplements.

Calcium that is present in the bloodstream circulates in the free state(ion), bound to organic substances, and bound with albumin. Most of the physiological functions of calcium depend upon the ionized fraction, but for routine work only total calcium in serum is estimated. total calcium level is affected by the amount of protein present. Corrected calcium = [Ca]+ 0. 02*{40 -[alb]} if [alb]<40 = [Ca]+ 0. 02*{[alb]- 45} if [alb]>40

Function: (ionized form) muscular contraction, cardiac functioning, hormone secretion, cell division, and the transmission of nerve impulses, blood coagulation. Normal Results Total plasma calcium 9. 0 -10. 5 mg/dl Free calcium 3. 9 -4. 6 mg/dl

Hypercalcemia can be indicative of: Hypocalcemia can be indicative of: • Cancers of the lung, breast, thyroid, kidney, • hyperparathyroidism • Paget's disease of bone, • prolonged immobilization • Addison‘s disease. • hypoparathyroidism • renal failure • Vitamin D deficiencies • malabsorption Low ionized calcium can subsequently lead to coagulation and hemostasis problems

Principle: Serum + ammonium oxalate ppt calciumoxalate The precipitate is washed with ammonia to remove excess oxalate calcium oxalate + sulphuric acid oxalic acid The latter is titrated with standard potassium permanganate.

Procedure: 1. Measure 2 ml of serum, 2 ml of water and 1 ml of 4% ammonium oxalate in a centrifuge tube. 2. Mix and allow to stand for at least half an hour thin Mix again and centrifuge at 1, 500 r. p. m. for 15 minutes. 3. Pour off the supernatant fluid and drain the tube by keeping it inverted on a filter paperfor a few minutes. Wipe the mouth of the tube dry with a filer paper. 4. Add 3 ml of 2% ammonia, shake, centrifuge and drain as before.

5. Add 2 ml of 1 Nsulphuric acid and shake vigorously. 6. Keep the tube in a boiling water-bathshaking intermittently, until the precipitate completely dissolves. 7. titrate with 0. 01 N potassium permanganate until a pinkcolour develops and persists for at least one minute. 8. Note the volume of 0. 01 N potassium permanganate used. Suppose it is x ml. 9. For blank, take 2 ml of 1 N sulphuric acid and titrate it with 0. 01 N potassium permanganate exactlyas before, suppose the volume of potassium permanganate used in y ml. Calculation:

Phosphate phosphorus is found in bones and teeth and is combined with calcium. The rest of phosphorus is in the soft tissues. Function: Phosphorus in the blood necessary for the metabolism of glucose, fats, and proteins; and the storage and transfer of energy Normal Results • Adults 2. 7 -4. 5 mg/dl • Children 4. 5 -5. 5 mg/dl

hyperphosphatemia may be due to or associated with: Hypophosphatemia may be due to or associated with: • Diabetic ketoacidosis (when first seen) • Increased dietary intake • Hypoparathyroidism • Hypercalcemia, • Overuse of diuretics • Rickets and osteomalacia (due to Vitamin D deficiencies) • Kidney failure • Hyperparathyroidism • Malnutrition Phosphate levels are normally higher in children than in adults because their bones are actively growing.

Principle: Serum is deproteinized with trichloracetic acid. Protein-free is filtrate. acid molybdate + phosphate phosphomolybdic acid 1, 2, 4 -aminonaphtholsulphonic acid redduced phosphomolybdous acid (molybdenum blue) The intensity of the colour is measured colorimetrically.
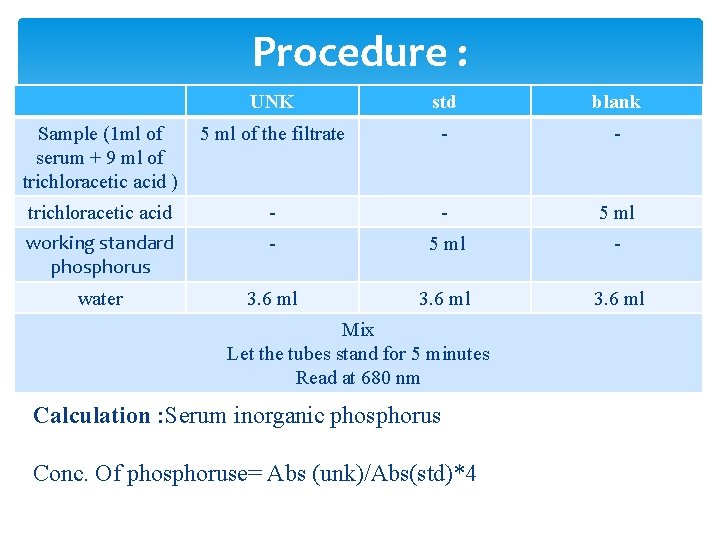
Procedure : UNK std blank Sample (1 ml of serum + 9 ml of trichloracetic acid ) 5 ml of the filtrate - - trichloracetic acid - - 5 ml working standard phosphorus - 5 ml - water 3. 6 ml Mix Let the tubes stand for 5 minutes Read at 680 nm Calculation : Serum inorganic phosphorus Conc. Of phosphoruse= Abs (unk)/Abs(std)*4

Magnesium Function: • muscular contraction • carbohydrate metabolism • protein synthesis It is usually filtered by the kidney through the glomerulus, and reabsorbed into the bloodstream by the renal tubule. Normal Results 1. 7 to 2. 2 mg/d. L

High magnesium levels may be due to : • • • Diabetic acidosis Oliguria Addison's disease Chronic renal failure Dehydration Low magnesium levels may be due to : • Alcoholism • Chronic diarrhea • Hemodialysis • Hepatic (liver) cirrhosis • Hyperaldosteronism • Hypoparathyroidism • Ulcerative colitis

Alkaline phosphatase ALP : enzyme that found in all body tissues. In the liver, it is found on the edges of cells that join to form bile ducts, in "osteoblasts“ of the bone cells. Normal Results Adult 17 -142 U/L Children 0 -12 yr 145 -530 U/L

High level of ALP may be due to: Low level of ALP may be due to: • Bone disease (Paget's disease, disease where • Some drugs (contraceptives ) bones become enlarged and deformed cancers spread to bone, Rickets, Osteomalacia) • Liver disease • Bile duct damage • Children & pregnant women (normal) *GGT test be done to differentiate between liver and bone disease. *If calcium and phosphorus measurements are abnormal, usually the coming from bone. ALP is

Principle : serum + buffer substrate phenol + 4 -animoantipyrine PH phenol alkaline medium red color Color is stable for at least an hour so, sodium hydroxide is added immediately after incubation to raise the p. H and stop the reaction.
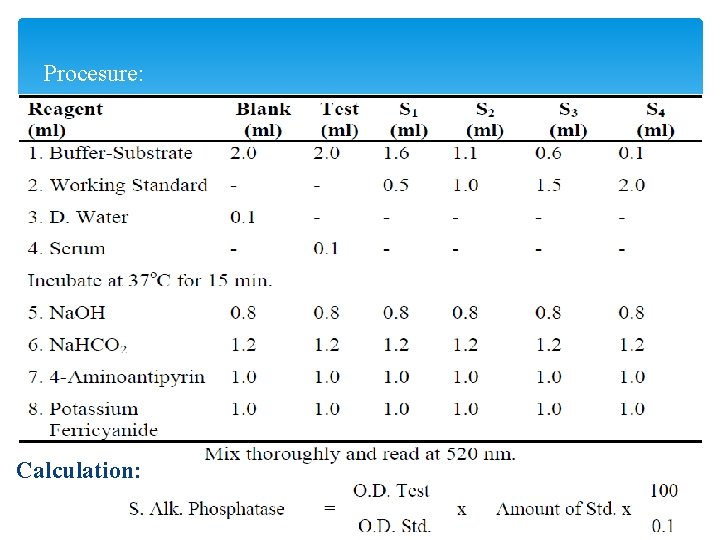
Procesure: Calculation:

Referance Marie A. Moisio, MA. (1993) Understanding Laboratory and Diagnostic Tests. Marshall, W. Bangert, K. (2008) clinical chemistry. 6 th ed. http: //www. westernsussexhospitals. nhs. uk/services-and-treatments/diagnosis-and -screening/blood/types/ http: //kidshealth. org/parent/system/index. html#cat 174 http: //labtestsonline. org/understanding/analytes/alp/tab/test http: //www. austincc. edu/search/results. php? cx=003770341465265277363%3 Ad 33 byhd 4 au 8&cof=FORID%3 A 11&ie=UTF-8&q=bone http: //courses. washington. edu/bonephys/opvit. D. html http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 1146022/ http: //www. nlm. nih. gov/medlineplus/ency/article/000344. htm
- Slides: 29