BONE RELATED NON COLLAGENOUS PROTEINS PERIODONTIUM The periodontium
BONE RELATED NON COLLAGENOUS PROTEINS

PERIODONTIUM The periodontium consists of the investing and supporting tissues of the tooth gingiva, periodontal ligament, cementum and alveolar bone. It has been divided into two parts. 1. The gingiva, the main function of which is protecting the underlying tissues, and 2. the attachment apparatus, composed of the periodontal ligament, cementum, and alveolar bone.
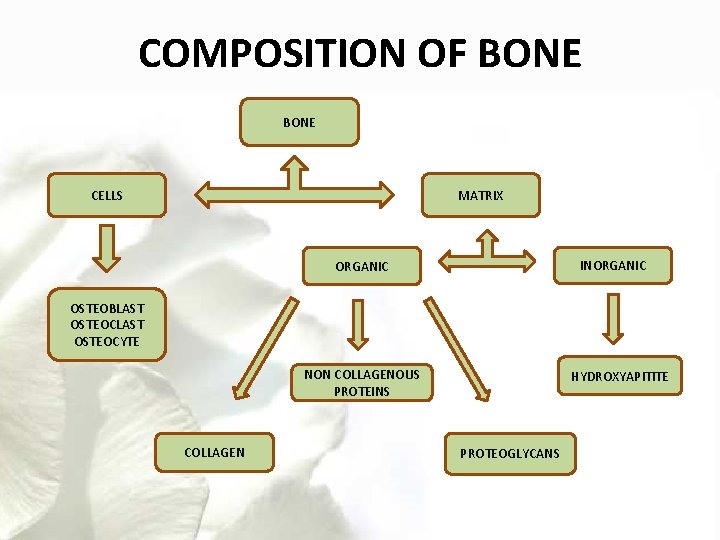
COMPOSITION OF BONE CELLS MATRIX INORGANIC OSTEOBLAST OSTEOCYTE NON COLLAGENOUS PROTEINS COLLAGEN HYDROXYAPITITE PROTEOGLYCANS

COLLAGEN Collagen is the major organic content of bone but collagen itself neither promotes nor inhibits the formation of hydroxyapatite (HA) (glimcher, 1989). Therefore the non-collagenous proteins of bone have been studied for possible crystal modulating activities.

Introduction Non-collagenous proteins of bone are a heterogeneous group which vary from entrapped serum protein to glycoproteins, which are unique to bone and which probably play a role in mineralization.

CLASSIFICATION OF NON COLLAGENOUS PROTEINS Basically classified according to the structure as I. Gla containing proteins 1. Bone gla protein or osteocalcin 2. Matrix gla protein II. SIBLING proteins 1. Osteopontin or BSP I 2. Bone sialoprotein or BSP II 3. Dentin matrix protein 1 (DMP 1) and 4. Dentin sialophosphoprotein (DSPP) III. Glycoproteins 1. Osteonectin IV. Proteoglycans Young et al 1992

NON-COLLAGENOUS PROTEINS: This seminar deals with four major noncollagenous bone proteins are 1, Osteopontin 2, Bone sialoprotein 3, Osteocalcin or bone Gla protein 4, Osteonectin

OSTEOPONTIN Secreted phosphoprotein 1 (SPP 1) Bone sialoprotein 1 (BSP 1) T-lymphocyte activation 1 (ETA) INTRODUCTION: Osteopontin is a glycosylated phosphoprotein with molecular mass of 41. 5 kd (bandt and guo 1993) Found primarily in bone as well as several other nonskeletal tissues. It is present in significant amounts in bone at mineralisation fronts and at cement lines where bone formation follows resorption
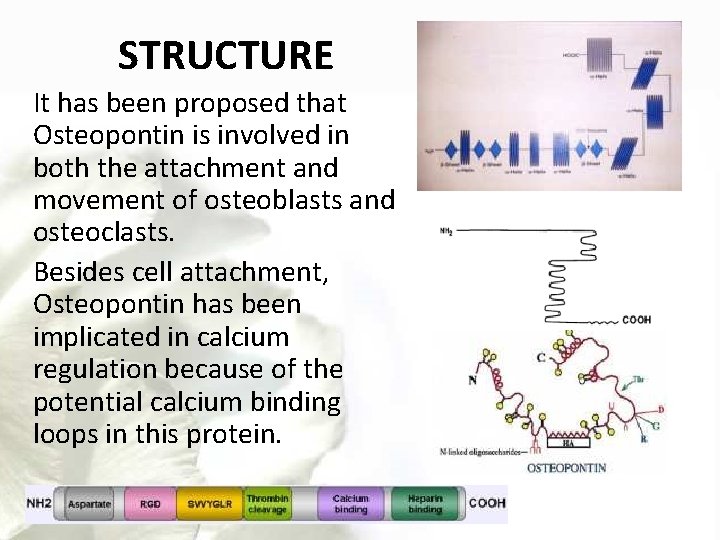
STRUCTURE It has been proposed that Osteopontin is involved in both the attachment and movement of osteoblasts and osteoclasts. Besides cell attachment, Osteopontin has been implicated in calcium regulation because of the potential calcium binding loops in this protein.

SYNTHESIS The human Osteopontin gene consists of seven exons, six of which contain the coding sequence and spans approximately 5 kd and localised on chromosome 4 q 13. In bone, the synthesis and release of osteopontin by osteoblasts under both endocrine and paracrine control. It is synthesised by many cell types including osteoblasts, osteocytes, smooth muscle cells and epithelial cells.

Cont; Its synthesis is up regulated by osteotropic hormones such as vitamin D 3 with high specificity VDRE. In the bone it is synthesised in two molecular forms as a 55 kd low phosphorylated species or as a 44 kd highly phosphorylated species (sodek et al 1995). Substances present in the site PDGF, TGF, EGF, BMP 7 and IL 2 up regulate its synthesis.

OSTEOPONTIN AND BONE MINERALISATION It plays a role in the attachment of osteoblasts and osteoclasts to bone matrix It act as a biologic matrix bonding agent. It plays a significant role in both mineralisation and resorption of bone (boskey 1992) Secreted Osteopontin is found in concentrated amount in areas of bone formation (mark et al 1998) It has been implicated in the recruitment and stimulation of macrophages and lymphocytes in response to nonspecific infections (mckee and nanci 1996).

ROLE IN BONE REMODELING Osteopontin has been implicated as an important factor in bone remodelling. It plays a role in anchoring osteoclasts to the mineral matrix of bones. Highest levels of Osteopontin expression are observed in preosteoblastic cells early in bone formation and mature osteoblasts at sites of bone remodelling. It plays a significant role in both mineralisation and resorption of bone (boskey 1992) It is a major ligand for osteoclasts thus it appears to be involved in bone remodelling (boskey 1992).

ROLE IN IMMUNE FUNCTIONS OPN binds to several integrin receptors expressed by leukocytes. These receptors have been well-established to function in cell adhesion, migration, and survival in these cells. Osteopontin (OPN) is expressed in a range of immune cells with varying kinetics. OPN is reported to act as an immune modulator. It has chemotactic properties, which promote cell recruitment to inflammatory sites. It also functions as an adhesion protein, involved in cell attachment and wound healing. OPN mediates cell activation and cytokine production, as well as promoting cell survival by regulating apoptosis.

SALIENT FEATURES Osteopontin is found in several non-mineralised tissues including kidney, arterial smooth muscle cells etc (denhardt and guo 1993). It covalently binds to fibronectin catalysed by the enzyme transglutaminase. It is produced in response to injury by macrophages, activated T lymphocytes and smooth muscle cells. It is also found in atherosclerotic plaques and it is also produced by transformed cells. It is found in many soft tissues suggesting that it may have a role in soft tissue organisation.
Cont; It is expressed earlier than bone sialoprotein. It is expressed during cementogenesis. Its significance in development may be related to its increased expression during mesenchymal cell migration. Osteopontin plays a key role in activating cell mediated immunity. It also appears to play a role in wound healing.

BONE SIALOPROTEIN Bone sialoprotein II Cell binding sialoprotein Integrin binding sialoprotein INTRODUCTION Bone sialoprotein (BSP) is a component of mineralized tissues such as bone, dentin, cementum and calcified cartilage. Similar to osteopontin, bone sialoprotein is another protein found primarily in the bone. It is specifically expressed by fully differentiated osteoblasts (Shapiro et al 1993; young et al 1992). It is a major structural protein of the bone matrix.
STRUCTURES It has a molecular mass of about 33 k. Da contains the ( Arg-Gly-Asp) RGD tripeptide sequence. Several well conserved domains have been noted in bone sialoprotein, that are potential sites for hydroxyapatite binding (Shapiro et al 1993). Bone sialoprotein appears to contain no cysteine (fisher et al 1990).

SYNTHESIS Human bone sialoprotein is coded by a single copy 15 -kb gene located on chromosome 4 q 28 q 31(fisher et al 1990; kerr et al 1993; kim et al 1994) The synthesis of bone sialoprotein by cultured osteoblasts. It is inhibited by calcitriol and stimulated by glucocorticoid hormones.

BONE SIALOPROTEIN AND OSTEOPONTIN They have considerable importance because they are implicated in both cellular and matrix events. They are part of a family of proteins(SIBLING). Both are found in mineralisation foci near the minaralisation front accumulate with in the spaces between the calcified collagen fibrils and are associated with cement lines. Both Inhibit mineral deposition when present in solution however when bound to a solid substrate they promotes mineral deposition. promote adhesion of selected cells to the newly forming root. BSP may be involved in promoting mineralisation whereas Osteopontin may regulate the extent of crystal growth.

DIFFERENCE BETWEEN OSTEOPONTIN AND BONE SIALOPROTEIN Both are phosphorylated sialoproteins with sulphated tyrosine Residues and regions of contiguous acidic aminoacids. BSP contains more sialic acid and less phosphate than Osteopontin (Franzen and Heinegard, 1985). In osteopontin the polycarboxylate sequences consist of aspartic acid (oldberg et al. , 1986) whereas in BSP they consist of glutamicacid (Oldberg etal. , 1988). BSP is specific to bone, although it has also been found in growthplate cartilage and the embryonic trophoblast layer (Fisheretal. , 1983; Biancoetal. , 1991). Osteopontin is expressed in a variety of tissues, but at higher levels in bone than elsewhere (Nomuraetal. , 1988; Brownetal. , 1992; Chen etal. , 1993).
BSP AND BONE MINERALISATION Bone sialoprotein is a promoter and osteocalcin is an inhibitor of bone mineralisation. Bone sialoprotein is thought to play an important role in osteoblast attachment to mineralised tissues. Providing an anchoring mechanism for bone cells to attach to mineralised tissues (boskey 1992). BSP is found in reversal lines of rapidly remodelling bone and it is produced by actively forming bone and produced by osteoblasts and odontoblasts (chen et al 1992). It is expressed during early formation of dentin and alveolar bone where it accumulates in peritubular dentin and bone matrix (amar et al).

Cont; It is associated with differentiated osteoblasts found on the surfaces of newly forming bone trabeculae. Nucleation of hydroxyapatite during bone formation and in the initial mineralisation of newly formed bone. BSP is not found in osteoid but is restricted to the mineralised bone matrix. High concentrations of BSP at the epiphyseal metaphyseal border during endochondral bone formation. It increases the osteoclastic resorption by promoting greater adhesion of the osteoclasts to bone matrix molecules. It promoting mineralisation in bone and cementum.

SALIENT FEATURES Bone sialoprotein also contains RGD sequences that involve integrin-mediated cell binding through the vitronectin receptor (oldberg et al 1988). It is also expressed by cementoblasts during cementogenesis ( chen et al 1992). BSP expression is up regulated by dexamethasone and suppressed by vitamin D (kim et al 1996; ogata et al 1995; yamauchi et al 1996). It can bind tightly to hydroxyapatite as well as to cells.

Cont; It has been proposed that the association of OCN and BSP with collagen fibrils creates locally high concentrations of calcium leading to precipitation of mineral. Not only BSP able to nucleate hydroxyapatite crystal formation, its temporospatial expression correlates with initial mineralisation and it localises to newly forming spherulite crystals. BSP found to be restricted to new cementum and bone particularly the periodontal ligament adjacent to new bone formation. Therefore it is an accurate marker of bone and cementum formation.

Cont; Immunostaining of periodontal ligament cells for bone sialoprotein, a marker of differentiated osteoblasts and cementoblasts, shows no staining reaction in periodontal ligament cells under physiological or wounding conditions. The absence of this late marker of osteoblast differentiation in repopulating periodontal ligament demonstrates that, while a significant portion of periodontal ligament cells may have osteogenic characteristics , these cells are blocked from differentiating into osteoblasts.

OSTEOCALCIN bone Gla protein (BGLAP) INTRODUCTION Osteocalcin, also known as bone gammacarboxyglutamic acid-containing protein (BGLAP), is a noncollagenous protein found in bone and dentin. In humans, the osteocalcin is encoded by the BGLAP gene. It is a member of a family of extracellular mineral binding proteins present in the bone (hauschka et al 1989).
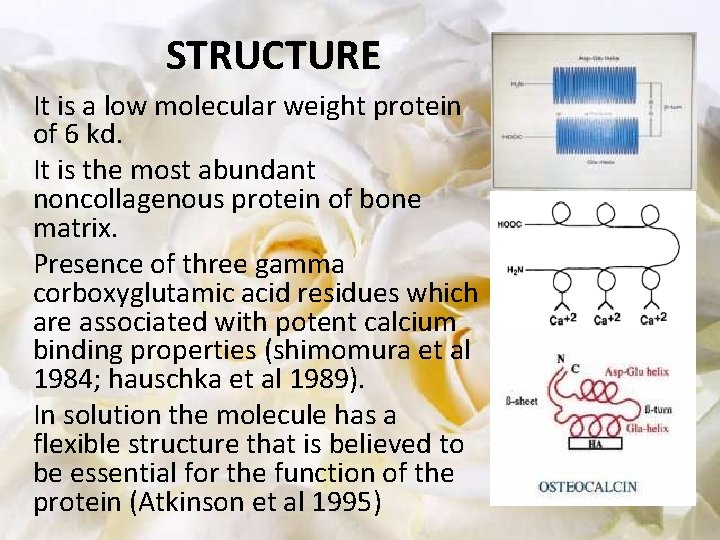
STRUCTURE It is a low molecular weight protein of 6 kd. It is the most abundant noncollagenous protein of bone matrix. Presence of three gamma corboxyglutamic acid residues which are associated with potent calcium binding properties (shimomura et al 1984; hauschka et al 1989). In solution the molecule has a flexible structure that is believed to be essential for the function of the protein (Atkinson et al 1995)

SYNTHESIS The osteocalcin gene is a 953 nucleotide sequence consisting of four exons and three introns and may be localised on chromosome 3 (lian et al 1989; desboiset al 1994) Osteocalcin is a small noncollagen protein synthesized principally by odontoblasts and osteoblasts. Only fully differentiated cells express this protein. It is inhibited by PTH and stimulated by calcitriol through VDRE and influences the movement and proliferation of osteoclasts. Osteocalcin is synthesized by the osteoblast as a 99 amino acid propeptide which is then cleaved to leave a 50 amino acid protein which is secreted.

FUNCTION Osteocalcin is secreted solely by osteoblasts and thought to play a role in the body's metabolic regulation and is pro-osteoblastic, or bonebuilding, by nature. It is also implicated in bone mineralization and calcium ion homeostasis. Osteocalcin acts as a hormone in the body, causing beta cells in the pancreas to release more insulin, and at the same time directing fat cells to release the hormone adiponectin, which increases sensitivity to insulin.

BIOCHEMICAL MARKER OF BONE FORMATION As osteocalcin is produced by osteoblasts, it is often used as a marker for the bone formation process. It has been observed that higher serumosteocalcin levels are relatively well correlated with increases in bone mineral density (BMD) during treatment with anabolic bone formation drugs for osteoporosis, such as Forteo. In many studies, osteocalcin is used as a preliminary biomarker on the effectiveness of a given drug on bone formation.

SALIENT FEATURES It is generally found in the bone matrix and specifically localizes to developing bone (bronkers et al 1987) Osteocalcin is highly conserved between species and is very specific for calcified tissues. The posttranslational vitamin K-dependent carboxylation of glutamic acid residues allows the osteocalcin molecule to bind calcium and hydroxyapatite molecules. As osteocalcin is one of the predominant noncollagen proteins in bone it has been suggested that osteocalcin is involved in the mineralization process.

Cont: Raised serum levels of osteocalcin have been reported in diseases which are associated with increased bone turnover such as Paget’s disease, renal osteodystrophy and primary hyperparathyroidism. Osteocalcin binds to osteopontin that interacts with osteoclasts; therefore it may be involved in recruiting osteoclasts to sites of newly formed bone and thus may function as a negative regulator (desbois and karsenty 1995)

OSTEONECTIN secreted protein and rich in cysteine (SPARC) BM 40, endothelial “culture shock” glycoprotein. INTRODUCTION: It is a calcium binding glycoprotein associated with the extracellular matrix of many tissues, especially bone. It is expressed by osteoprogenitor cells, osteoblasts, and newly formed osteocytes.
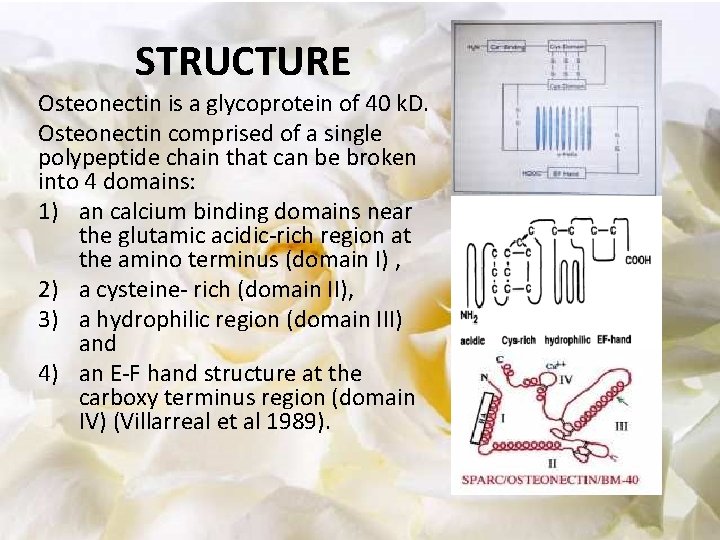
STRUCTURE Osteonectin is a glycoprotein of 40 k. D. Osteonectin comprised of a single polypeptide chain that can be broken into 4 domains: 1) an calcium binding domains near the glutamic acidic-rich region at the amino terminus (domain I) , 2) a cysteine- rich (domain II), 3) a hydrophilic region (domain III) and 4) an E-F hand structure at the carboxy terminus region (domain IV) (Villarreal et al 1989).

SYNTHESIS The human SPARC gene is 26. 5 kb long, and contains 10 exons and 9 introns and is located on chromosome 5 q 31 -q 33. Fibroblasts, including periodontal fibroblasts, synthesize osteonectin (wasi et al 1984). This protein is synthesized by macrophages at sites of wound repair and platelet degranulation. so it play a important role in wound healing. SPARC does not support cell attachment and like thrombospondin and tenascin, it antiadhesive and an inhibitor of cell spreading. It distrupt focal adhesion in fibroblasts. It regulates the proliferation of some cells especially the endothelial cells, and it mediate this effect through its ability to bind to cytokines and growth factors (young et al 1992). Osteonectin has also been found decrease DNA synthesis in cultured bone (lane and sage 1994).

SALIENT FEATURES The domains at the amino and carboxy terminus are calciumbinding regions. High levels of immunodetectable osteonectin are found in active osteoblasts and marrow progenitor cells, odontoblasts, pdl and gingival cells and some chondrocytes and hypertrophic chondrocytes. Osteonectin is also detectable in osteoid, bone matrix proper and dentin. Osteonectin localized in a variety of tissues, but generally speaking, it is found in greatest abundance in osseous tissue, tissues characterized by high turnover (such as intestinal epithelium), basement membranes and certain neoplasms.

cont; Osteonectin is expressed by a wide variety of cells and numerous neoplastic cell lines (such as Sa. OS-2 cells from human osteosarcoma). This molecule implicated in several biological functions, including mineralization of bone and cartilage, inhibiting mineralization, modulation of cell proliferation and promotion of cell attachment and spreading. The phosphate is bound to the protein backbone through serine or threonine amino acid residues. It binds collagen and hydroxyapatite through separate areas of its molecule, is found in relatively large amounts in immature bone and promotes mineralization of collagen. Thus it is possible that Osteonectin plays a crucial role in mineralization.

ACTION OF NON-COLLAGENOUS PROTEINS IN CEMENTUM AND PERIODONTAL LIGAMENT Non-collagen protein in the composition of the cementum, and periodontal tissue relationship and promote regeneration of periodontal ligament cell adhesion machine. recent research suggests that cementum contains some cementum, periodontal ligament attachment and periodontal ligament cells (PDLC) the synthesis of extracellular matrix collagen and other non-related proteins, including non-collagen sugar, non-collagen protein and proteoglycan. This “little protein content category, may be less than 1% of the total protein cementum, but they have a high biological activity, some in the periodontal tissue regeneration plays a key role.

Biochemical composition PERIODONTAL LIGAMENT CEMENTUM

1 cementum non-collagen protein (CNCPs) The composition of this compared with the enamel and dentin, cementum with the bone tissue has a more “multi-similarity. So cementum contains some of the bone tissue can be extracted to the noncollagenous proteins, such as Fibronectin, Osteopontin, vitronectin, osteocalcin, Osteonectin, Tenascin, bone sialoprotein – Ⅱ and so on. The structure and function of cementum and bone tissue have significant differences, so the noncollagen protein composition and function have certain differences.

2. CNCPs and periodontal tissue regeneration CNCPs can be induced in vitro experiments show that periodontal tissue regeneration. It has an important role in the differentiation of mesenchymal cells, promoting PDLC proliferation, mitosis, migration and adhesion in the root surface, and promote the formation of cementum and alveolar bone for dental bone regeneration to provide the necessary signals. CNCPs selective role in PDLC, significantly improved the biological activity of the cells.

most CNCPs (FN, BSP-Ⅱ, OPN, VN, p 55, etc. ) and integration by identifying factors that (integrin) receptor combination of RGD peptide sequence (arginine-glycine-aspartic acid sequence) and the surface of target cells with specific receptors, the transmembrane receptors play a role. synthetic peptide containing RGD sequence can be competitive inhibition CNCPs of this receptor – ligand effect, significantly inhibited cell adhesion CNCPs promote the role of fiber. cementum non-collagen protein found in the periodontal tissues as an important signaling molecule, is indeed able to regulate activity of periodontal ligament cells.

SUMMARY The non-collagenous proteins have the crystal modulating activity which helps in the formation of hydroxyapatite and mineralization of bone. Osteopontin, bone sialoprotein are necessary for the initiation of bone mineralisation where the osteocalcin or bone Gla protein, Osteonectin play a role in control the size and speed of crystal formation (roach HI. Cell, Boil. int. 1994, jun 18(6) pg 617 -28). Osteopontin is involved in both the attachment and movement of osteoblasts and osteoclasts in bone via integrin mediated cell binding. Osteopontin plays a significant role in both mineralisation and resorption of bone (boskey 1992) Osteopontin also called early T lymphocyte activation 1(eta) plays a key role in activating cell mediated immunity.

SUMMARY OPN and SPARC appear to play a role in wound healing. BSP and OPN inhibit mineral deposition when present in solution however when bound to a solid substrate they can act as a promoters of mineral deposition. BSP promote mineralisation in bone and cementum. So it is an accurate marker of bone and cementum formation. Osteopontin regulate the extent of crystal growth. OCN binds to OPN that interacts with osteoclasts. Osteocalcin function as a negative regulator of bone formation and is a biochemical marker of bone formation. Osteonectin and osteocalcin have been reported to inhibit the seeded growth and formation of HA. In the presence of fibrillar collagen, Osteonectin has been reported to promote the formation of HA (termine et al 1981)
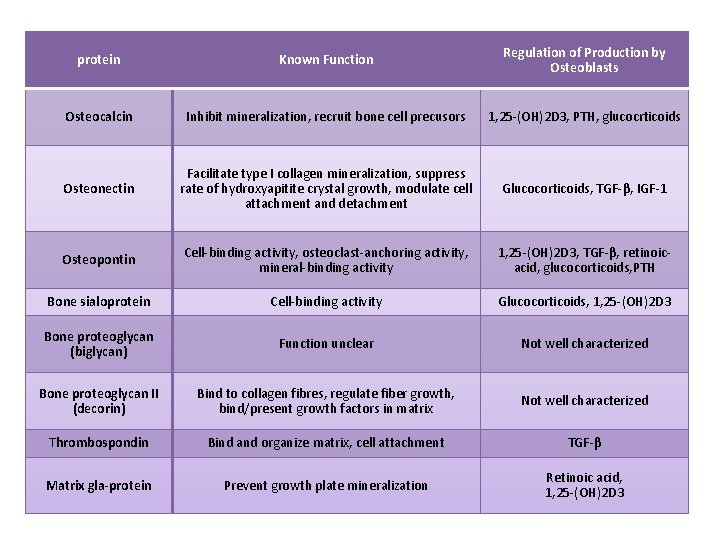
protein Known Function Regulation of Production by Osteoblasts Osteocalcin Inhibit mineralization, recruit bone cell precusors 1, 25 -(OH)2 D 3, PTH, glucocrticoids Osteonectin Facilitate type I collagen mineralization, suppress rate of hydroxyapitite crystal growth, modulate cell attachment and detachment Glucocorticoids, TGF-β, IGF-1 Osteopontin Cell-binding activity, osteoclast-anchoring activity, mineral-binding activity 1, 25 -(OH)2 D 3, TGF-β, retinoicacid, glucocorticoids, PTH Bone sialoprotein Cell-binding activity Glucocorticoids, 1, 25 -(OH)2 D 3 Bone proteoglycan (biglycan) Function unclear Not well characterized Bone proteoglycan II (decorin) Bind to collagen fibres, regulate fiber growth, bind/present growth factors in matrix Not well characterized Thrombospondin Bind and organize matrix, cell attachment TGF-β Matrix gla-protein Prevent growth plate mineralization Retinoic acid, 1, 25 -(OH)2 D 3
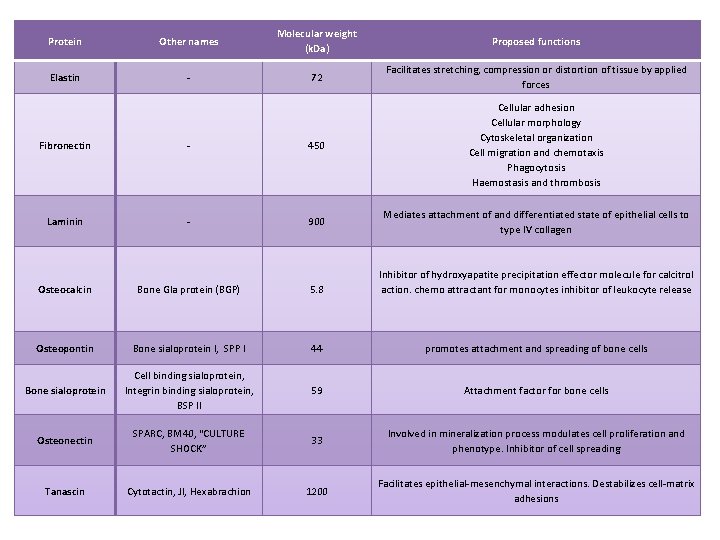
Protein Other names Molecular weight (k. Da) Proposed functions Elastin - 72 Facilitates stretching, compression or distortion of tissue by applied forces Fibronectin - 450 Cellular adhesion Cellular morphology Cytoskeletal organization Cell migration and chemotaxis Phagocytosis Haemostasis and thrombosis Laminin - 900 Mediates attachment of and differentiated state of epithelial cells to type IV collagen Osteocalcin Bone Gla protein (BGP) 5. 8 Inhibitor of hydroxyapatite precipitation effector molecule for calcitrol action. chemo attractant for monocytes inhibitor of leukocyte release Osteopontin Bone sialoprotein I, SPP I 44 promotes attachment and spreading of bone cells Bone sialoprotein Cell binding sialoprotein, Integrin binding sialoprotein, BSP II 59 Attachment factor for bone cells Osteonectin SPARC, BM 40, “CULTURE SHOCK” 33 Involved in mineralization process modulates cell proliferation and phenotype. Inhibitor of cell spreading Tanascin Cytotactin, JI, Hexabrachion 1200 Facilitates epithelial-mesenchymal interactions. Destabilizes cell-matrix adhesions

OTHER NON-COLLAGENOUS PROTEINS ARE: Matrix Gla Protein As much as 50 percent of the total glutamic acid containing proteins of bone are distinct from osteocalcin. The best characterized of these is matrix Gla protein (MGP), a 10, 000 Da protein found in association with bone morphogenetic protein. Proteoglycans Bone proteoglycan has a small protein core with up to two chondroitin sulphate chains attached. They constitute approximately 10 percent of the non-collagenous proteins of bone. Serum proteins These constitute the largest number of non-collagenous proteins in bone. They include serum albumin and some immunoglobulins. They constitute approximately a quarter of the total non-collagenous protein


REFERANCES BIOLOGY OF THE PERIODONTAL CONNECTIVE TISSUES - DR. BARTOLD. FUNDAMENTALS OF PERIODONTICS – WILLSON/KORNMAN. MOLECULAR BIOLOGY - DR. K. V. ARUN. PERIO 2000 VOL -3, VOL-24. JOURNAL OF STRUCTURAL BIOLOGY 1999. PUBMED AND OTHER MEDICAL WEBSITE.
- Slides: 50