Bone and Lung Cancer Dr Peter Harper Leaders

Bone and Lung Cancer Dr Peter Harper Leaders in Oncology Care London

WHAT UNITES TORINO AND LIVERPOOL?

WHAT UNITES TORINO AND LIVERPOOL? Prof Giorgio Vittorio Scagliotti & Prof Pieter Edsge Postmus

WHAT UNITES TORINO AND MANCHESTER?

TORINO 4 th MAY 1949 The Superga air disaster occurred on 4 May 1949 when the Fiat G. 212 of Avio Linee Italiane (Italian Airlines), carrying the Entire Torino football team crashed into the retaining wall at the back of the Basilica of Superga, which stands on the hill of Turin. 31 people died in the crash.

After refuelling at Barcelona, the plane carrying the team flew into a thunderstorm on the approach to Turin and encountered conditions of low cloud and poor visibility. They were forced to descend to be able to fly visually. . The emotional impact of the crash on Italian sports fans was profound, as it claimed the lives of the players of a team which had won five successive Serie A titles, tying the all-time record, and seriously weakened the Italian national team, which had included up to 10 Torino players. Torino itself would not claim another title until 1976.

Manchester United February 6 th 1958 On that day in 1958, the darkest day in United's history, 23 people - including eight players and three members of the club's staff - suffered fatal injuries in the Munich air crash. Flying back from a European Cup tie against Red Star Belgrade, the team plane stopped in Germany to refuel. The first two attempts to take off from Munich airport were aborted; following a third attempt, the plane crashed. Twenty-one of the people on board died instantly. Aeroplane captain Kenneth Rayment died a few weeks later from the injuries he sustained while Duncan Edwards - one of the eight victims from the team - passed away 15 days after the crash. The tragedy is an indelible part of United's history, as is Sir Matt Busby overcoming his injuries to build another great team which won the European Cup 10 years later.

Bone Metastases Have Debilitating Consequences ‘Skeletal Related Events’ (SREs) Disease Skeletal-related events Loss of autonomy Fracture Radiation to bone Bone mets Consequences Spinal cord compression Hypercalcemia Surgery to bone Kinnane N. Eur J Oncol Nurs. 2007; 11(suppl): S 28 -S 31. Significant morbidity Bone pain Increased healthcare costs and resources

Bone Metastases Have Debilitating Consequences Disease Skeletal-related events Loss of autonomy Fracture Radiation to bone Bone metastases Consequences Ultimate consequence Spinal cord compression Hypercalcemia Surgery to bone Kinnane N. Eur J Oncol Nurs. 2007; 11(suppl): S 28 -S 31. Significant morbidity Bone pain Increased healthcare costs and resources Decreased survival
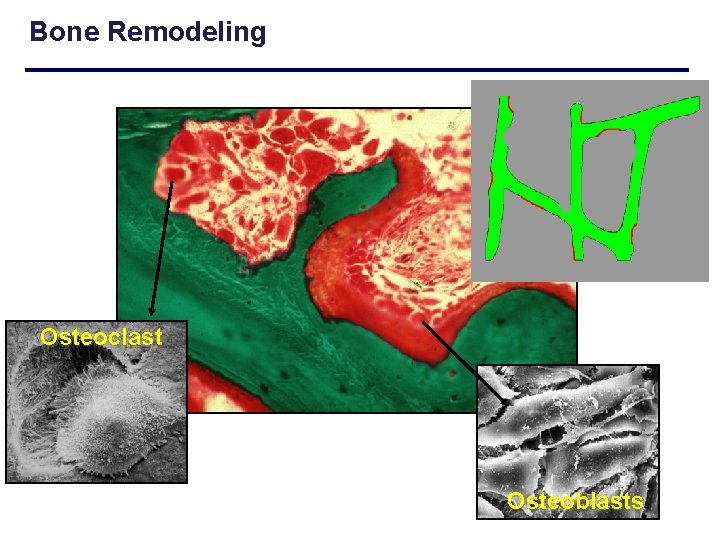
Bone Remodeling Osteoclast Osteoblasts
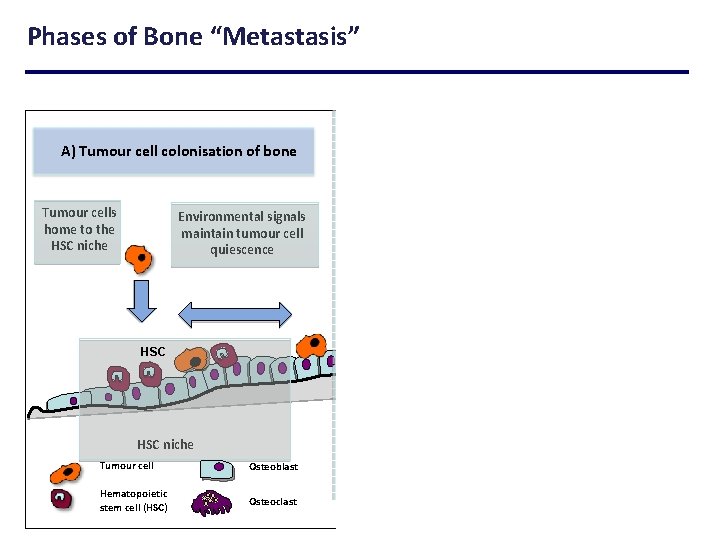
Phases of Bone “Metastasis” A) Tumour cell colonisation of bone Tumour cells home to the HSC niche Environmental signals maintain tumour cell quiescence B) Tumour cell proliferation and bone metastasis progression Escape from quiescence Tumour cell proliferation Re-circulation to other metastatic sites HSC Stimulation of bone resorption HSC niche Tumour cell Osteoblast Hematopoietic stem cell (HSC) Osteoclast Development of bone lesions
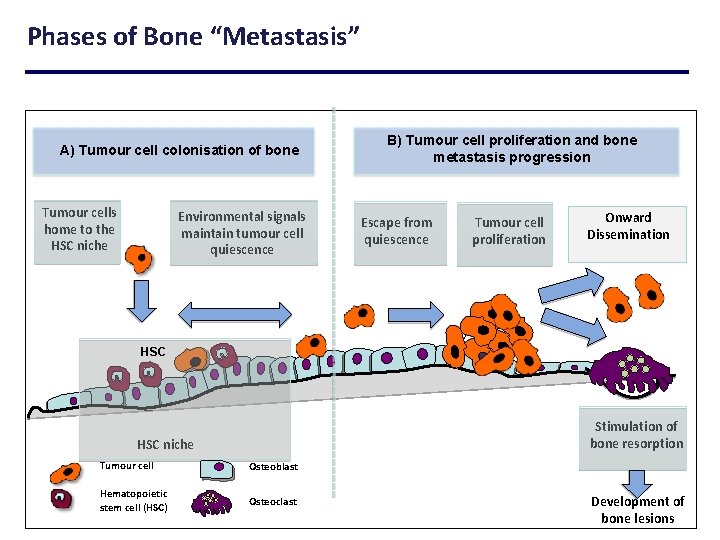
Phases of Bone “Metastasis” A) Tumour cell colonisation of bone Tumour cells home to the HSC niche Environmental signals maintain tumour cell quiescence B) Tumour cell proliferation and bone metastasis progression Escape from quiescence Tumour cell proliferation Onward Dissemination HSC Stimulation of bone resorption HSC niche Tumour cell Osteoblast Hematopoietic stem cell (HSC) Osteoclast Development of bone lesions
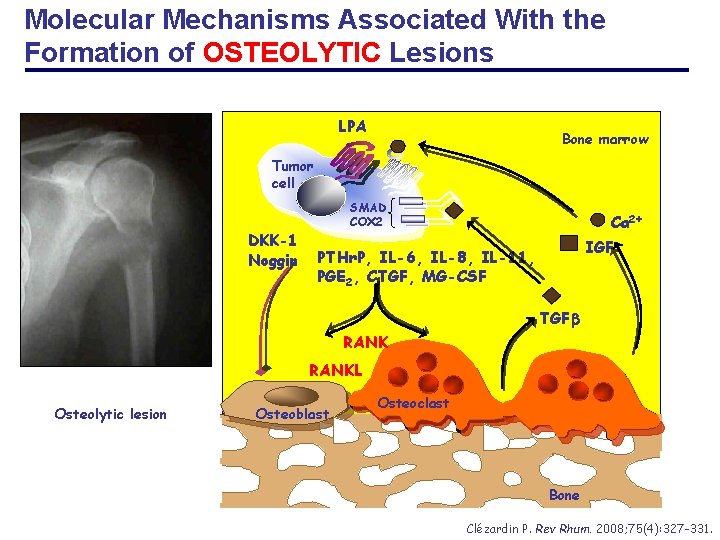
Molecular Mechanisms Associated With the Formation of OSTEOLYTIC Lesions LPA Bone marrow Tumor cell DKK-1 Noggin SMAD COX 2 Ca 2+ IGF PTHr. P, IL-6, IL-8, IL-11, PGE 2, CTGF, MG-CSF TGF RANKL Osteolytic lesion Osteoblast Osteoclast Bone Clézardin P. Rev Rhum. 2008; 75(4): 327 -331.
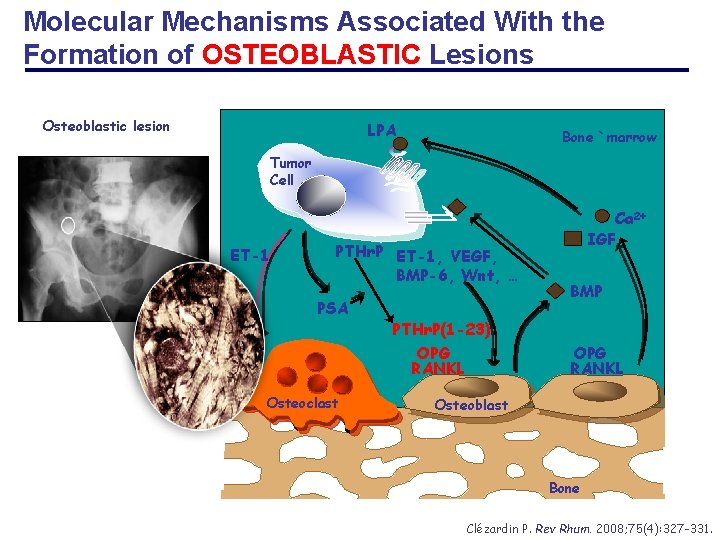
Molecular Mechanisms Associated With the Formation of OSTEOBLASTIC Lesions Osteoblastic lesion LPA Bone `marrow Tumor Cell ET-1 PTHr. P ET-1, VEGF, BMP-6, Wnt, … PSA Osteoclast PTHr. P(1 -23) OPG RANKL Ca 2+ IGF BMP OPG RANKL Osteoblast Bone Clézardin P. Rev Rhum. 2008; 75(4): 327 -331.
Clinical Priorities Across the Spectrum of Bone Disease Prevention of Treatment Induced Bone Loss Metastasis Prevention of Skeletal Morbidity
Clinical Priorities Across the Spectrum of Bone Disease Prevention of Treatment Induced Bone Loss Metastasis Prevention (ie ‘do something’) Prevention of Skeletal Morbidity (ie ‘do something’)
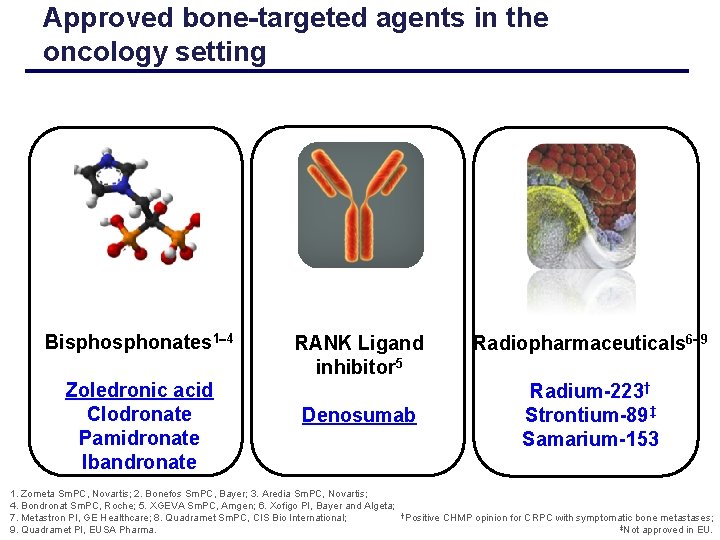
Approved bone-targeted agents in the oncology setting Bisphonates 1 4 Zoledronic acid Clodronate Pamidronate Ibandronate RANK Ligand inhibitor 5 Denosumab Radiopharmaceuticals 6 9 Radium-223† Strontium-89‡ Samarium-153 1. Zometa Sm. PC, Novartis; 2. Bonefos Sm. PC, Bayer; 3. Aredia Sm. PC, Novartis; 4. Bondronat Sm. PC, Roche; 5. XGEVA Sm. PC, Amgen; 6. Xofigo PI, Bayer and Algeta; †Positive CHMP opinion for CRPC with symptomatic bone metastases; 7. Metastron PI, GE Healthcare; 8. Quadramet Sm. PC, CIS Bio International; ‡Not approved in EU. 9. Quadramet PI, EUSA Pharma.

Bisphonates • High affinity for bone 1 • Rapidly absorbed onto bone surface 1 • Induce apoptosis of activated osteoclasts 1 • Long half-life in bone (1 10 years)2 • Excreted unchanged by the kidneys 2 1. Green JR. Oncologist 2004; 9(Suppl 4): 3– 13; 2. Lin JH. Bone 1996; 18: 75 85.
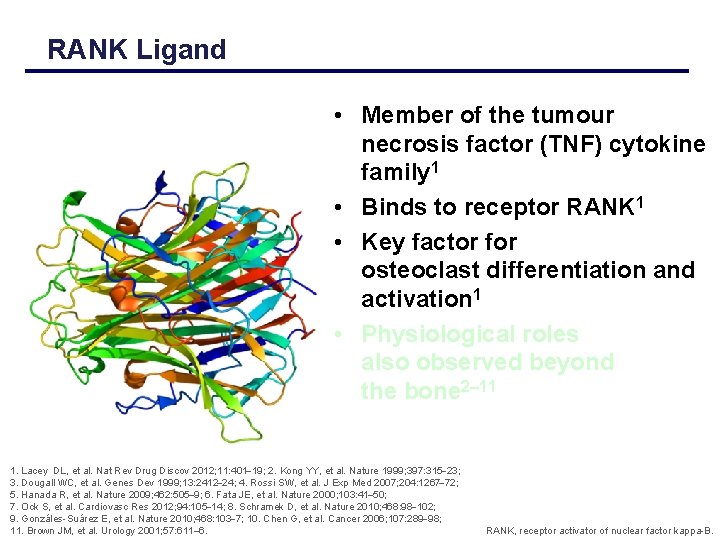
RANK Ligand • Member of the tumour necrosis factor (TNF) cytokine family 1 • Binds to receptor RANK 1 • Key factor for osteoclast differentiation and activation 1 • Physiological roles also observed beyond the bone 2 11 1. Lacey DL, et al. Nat Rev Drug Discov 2012; 11: 401 19; 2. Kong YY, et al. Nature 1999; 397: 315 23; 3. Dougall WC, et al. Genes Dev 1999; 13: 2412 24; 4. Rossi SW, et al. J Exp Med 2007; 204: 1267 72; 5. Hanada R, et al. Nature 2009; 462: 505 9; 6. Fata JE, et al. Nature 2000; 103: 41 50; 7. Ock S, et al. Cardiovasc Res 2012; 94: 105 14; 8. Schramek D, et al. Nature 2010; 468: 98 102; 9. Gonzáles-Suárez E, et al. Nature 2010; 468: 103 7; 10. Chen G, et al. Cancer 2006; 107: 289 98; 11. Brown JM, et al. Urology 2001; 57: 611– 6. RANK, receptor activator of nuclear factor kappa-B.
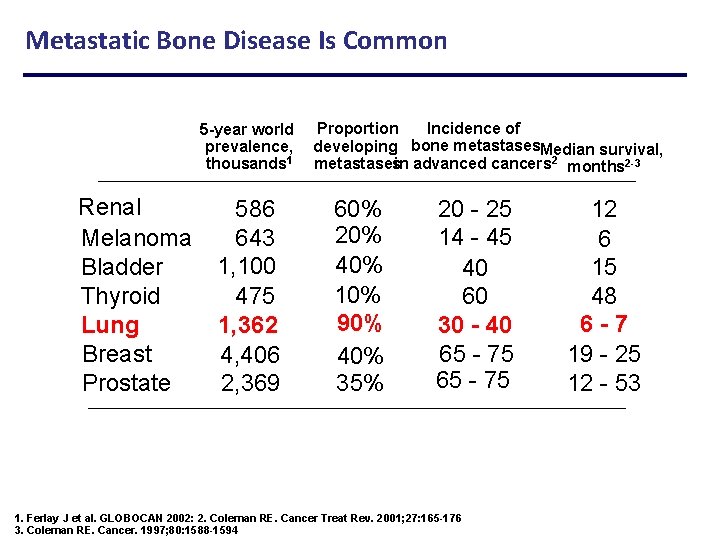
Metastatic Bone Disease Is Common 5 -year world prevalence, thousands 1 Renal 586 Melanoma 643 1, 100 Bladder Thyroid 475 Lung 1, 362 Breast 4, 406 Prostate 2, 369 Incidence of Proportion developing bone metastases. Median survival, metastasesin advanced cancers 2 months 2 -3 60% 20% 40% 10% 90% 40% 35% 20 - 25 14 - 45 40 60 30 - 40 65 - 75 1. Ferlay J et al. GLOBOCAN 2002: 2. Coleman RE. Cancer Treat Rev. 2001; 27: 165 -176 3. Coleman RE. Cancer. 1997; 80: 1588 -1594 12 6 15 48 6 -7 19 - 25 12 - 53
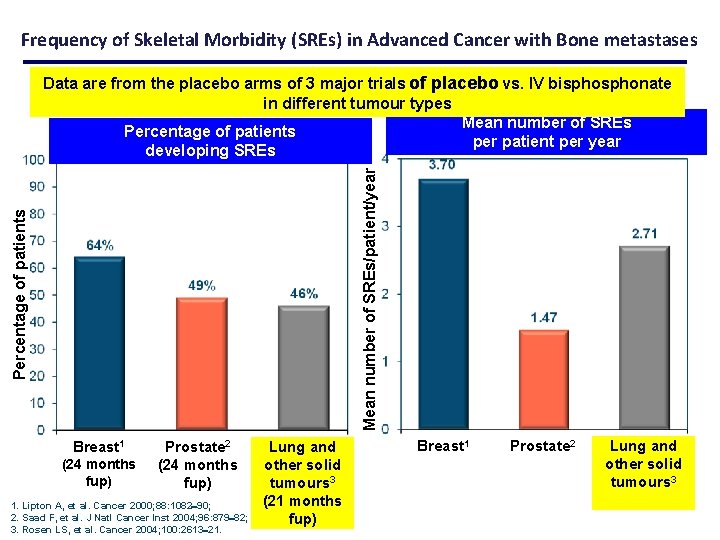
Frequency of Skeletal Morbidity (SREs) in Advanced Cancer with Bone metastases Percentage of patients Mean number of SREs/patient/year Data are from the placebo arms of 3 major trials of placebo vs. IV bisphonate in different tumour types Mean number of SREs Percentage of patients per patient per year developing SREs Breast 1 (24 months fup) Prostate 2 (24 months fup) 1. Lipton A, et al. Cancer 2000; 88: 1082 90; 2. Saad F, et al. J Natl Cancer Inst 2004; 96: 879 82; 3. Rosen LS, et al. Cancer 2004; 100: 2613 21. Lung and other solid tumours 3 (21 months fup) Breast 1 Prostate 2 Lung and other solid tumours 3
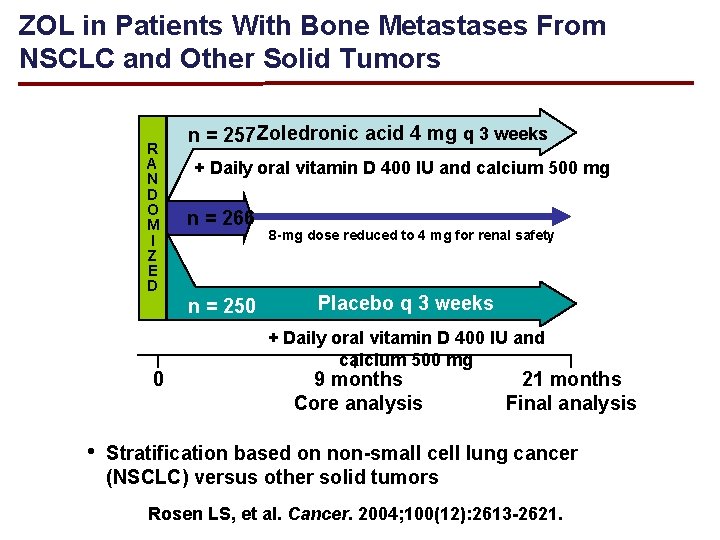
ZOL in Patients With Bone Metastases From NSCLC and Other Solid Tumors R A N D O M I Z E D n = 257 Zoledronic acid 4 mg q 3 weeks + Daily oral vitamin D 400 IU and calcium 500 mg n = 266 n = 250 0 • 8 -mg dose reduced to 4 mg for renal safety Placebo q 3 weeks + Daily oral vitamin D 400 IU and calcium 500 mg 9 months Core analysis 21 months Final analysis Stratification based on non-small cell lung cancer (NSCLC) versus other solid tumors Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621.
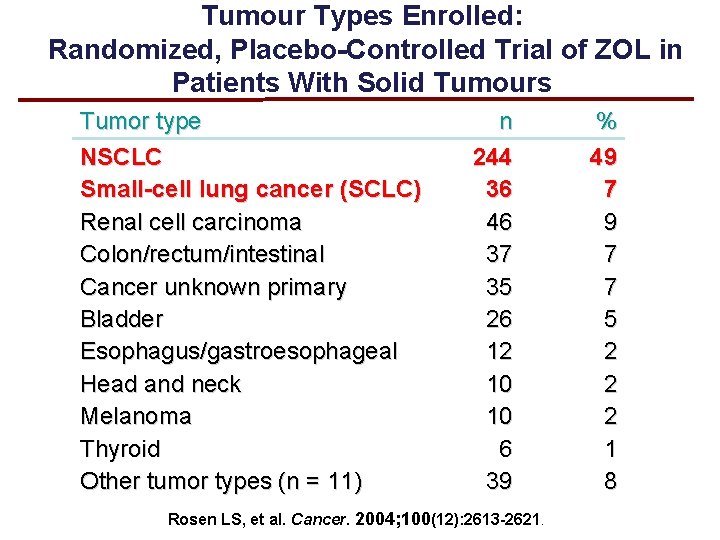
Tumour Types Enrolled: Randomized, Placebo-Controlled Trial of ZOL in Patients With Solid Tumours Tumor type NSCLC Small-cell lung cancer (SCLC) Renal cell carcinoma Colon/rectum/intestinal Cancer unknown primary Bladder Esophagus/gastroesophageal Head and neck Melanoma Thyroid Other tumor types (n = 11) n 244 36 46 37 35 26 12 10 10 6 39 Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621. % 49 7 7 5 2 2 2 1 8
![Disease Progression and Survival in the Overall Trial Population Median time to, [days] Bone Disease Progression and Survival in the Overall Trial Population Median time to, [days] Bone](http://slidetodoc.com/presentation_image_h/8b3c50efbc6f7e674494d51fa9d135a7/image-24.jpg)
Disease Progression and Survival in the Overall Trial Population Median time to, [days] Bone lesion progression ZOL 4 mg (n = 257) Placebo (n = 250) 145 days 109 P =. 415 Overall disease progression 89 days 84 P =. 089 Death 203 days P =. 929 Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621. 183

Bone Metastases are all bone metastases the same? ?
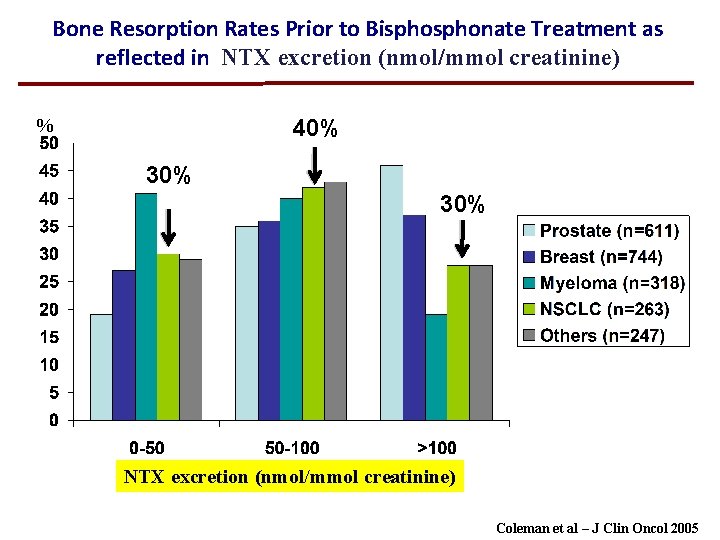
Bone Resorption Rates Prior to Bisphonate Treatment as reflected in NTX excretion (nmol/mmol creatinine) % 40% 30% NTX excretion (nmol/mmol creatinine) Coleman et al – J Clin Oncol 2005
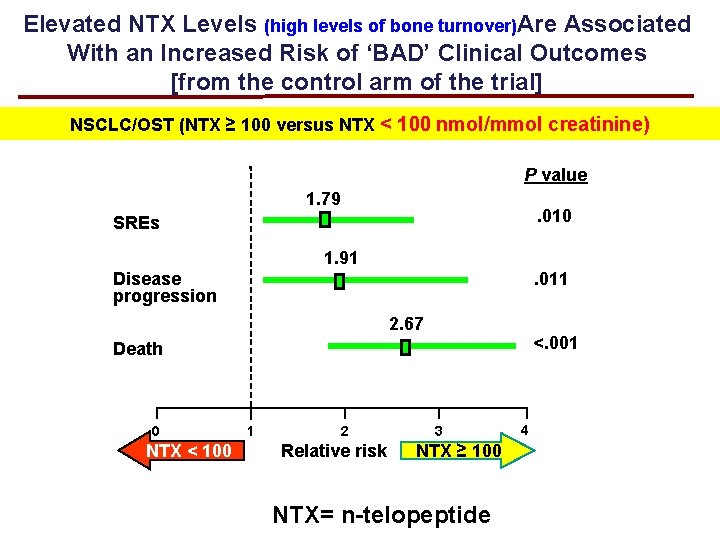
Elevated NTX Levels (high levels of bone turnover)Are Associated With an Increased Risk of ‘BAD’ Clinical Outcomes [from the control arm of the trial] NSCLC/OST (NTX ≥ 100 versus NTX < 100 nmol/mmol creatinine) P value 1. 79 . 010 SREs 1. 91 . 011 Disease progression 2. 67 <. 001 Death 0 NTX < 100 1 2 Relative risk 3 NTX ≥ 100 NTX= n-telopeptide 4
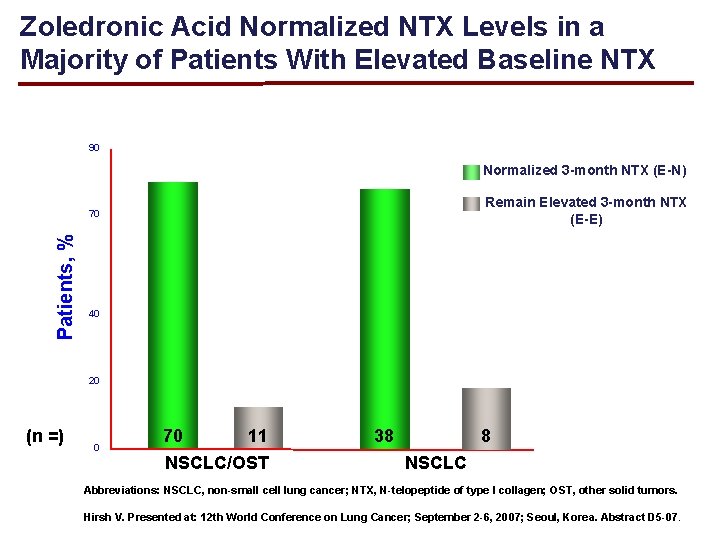
Zoledronic Acid Normalized NTX Levels in a Majority of Patients With Elevated Baseline NTX 90 Normalized 3 -month NTX (E-N) 80 Remain Elevated 3 -month NTX (E-E) Patients, % 70 60 50 40 30 20 10 (n =) 0 70 11 NSCLC/OST 38 8 NSCLC Abbreviations: NSCLC, non-small cell lung cancer; NTX, N-telopeptide of type l collagen; OST, other solid tumors. Hirsh V. Presented at: 12 th World Conference on Lung Cancer; September 2 -6, 2007; Seoul, Korea. Abstract D 5 -07.
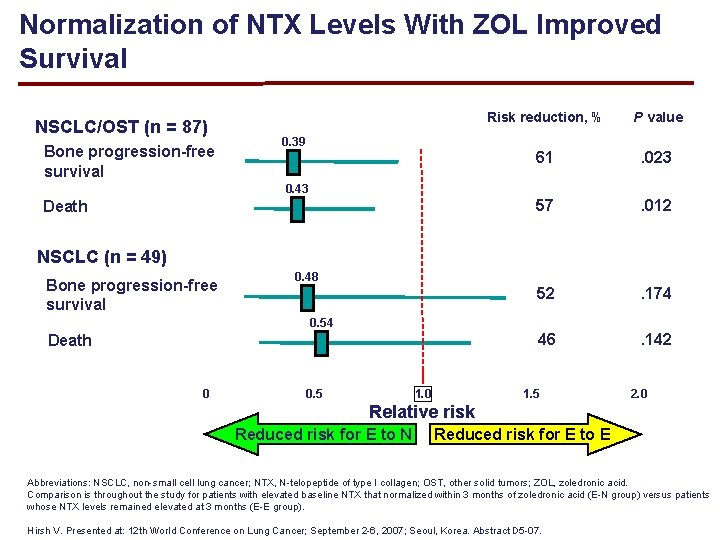
Normalization of NTX Levels With ZOL Improved Survival NSCLC/OST (n = 87) Bone progression-free survival Risk reduction, % P value 61 . 023 57 . 012 52 . 174 46 . 142 0. 39 0. 43 Death NSCLC (n = 49) Bone progression-free survival 0. 48 0. 54 Death 0 0. 5 1. 0 1. 5 2. 0 Relative risk Reduced risk for E to N Reduced risk for E to E Abbreviations: NSCLC, non-small cell lung cancer; NTX, N-telopeptide of type I collagen; OST, other solid tumors; ZOL, zoledronic acid. Comparison is throughout the study for patients with elevated baseline NTX that normalized within 3 months of zoledronic acid (E-N group) versus patients whose NTX levels remained elevated at 3 months (E-E group). Hirsh V. Presented at: 12 th World Conference on Lung Cancer; September 2 -6, 2007; Seoul, Korea. Abstract D 5 -07.
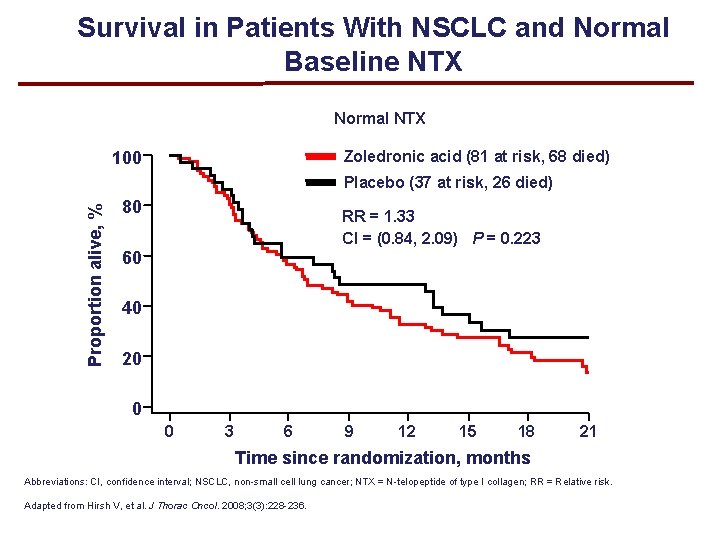
Survival in Patients With NSCLC and Normal Baseline NTX Normal NTX 100 Zoledronic acid (81 at risk, 68 died) Proportion alive, % Placebo (37 at risk, 26 died) 80 RR = 1. 33 CI = (0. 84, 2. 09) P = 0. 223 60 40 20 0 0 3 6 9 12 15 18 21 Time since randomization, months Abbreviations: CI, confidence interval; NSCLC, non-small cell lung cancer; NTX = N-telopeptide of type I collagen; RR = Relative risk. Adapted from Hirsh V, et al. J Thorac Oncol. 2008; 3(3): 228 -236.
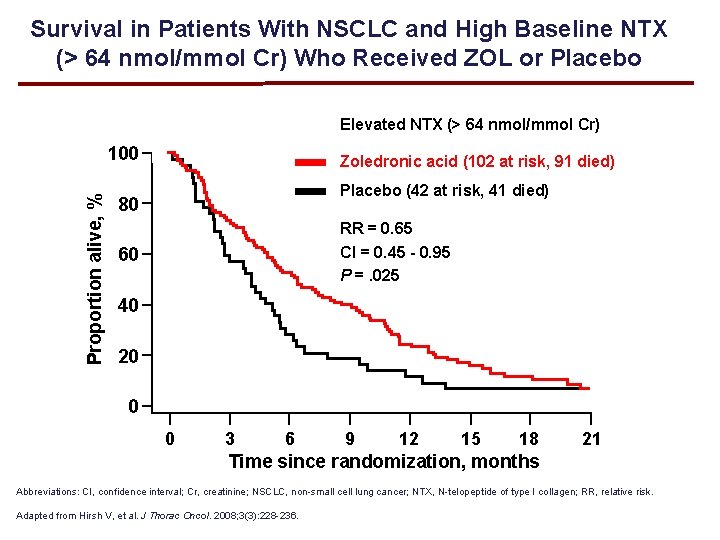
Survival in Patients With NSCLC and High Baseline NTX (> 64 nmol/mmol Cr) Who Received ZOL or Placebo Elevated NTX (> 64 nmol/mmol Cr) Proportion alive, % 100 Zoledronic acid (102 at risk, 91 died) Placebo (42 at risk, 41 died) 80 RR = 0. 65 CI = 0. 45 - 0. 95 P =. 025 60 40 20 0 0 3 6 9 12 15 18 21 Time since randomization, months Abbreviations: CI, confidence interval; Cr, creatinine; NSCLC, non-small cell lung cancer; NTX, N-telopeptide of type I collagen; RR, relative risk. Adapted from Hirsh V, et al. J Thorac Oncol. 2008; 3(3): 228 -236.
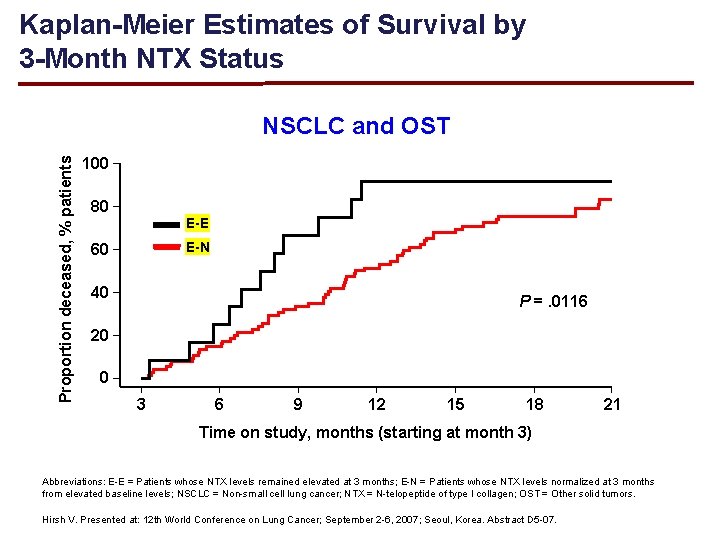
Kaplan-Meier Estimates of Survival by 3 -Month NTX Status Proportion deceased, % patients NSCLC and OST 100 80 E-E E-N 60 40 P =. 0116 20 0 3 6 9 12 15 18 21 Time on study, months (starting at month 3) Abbreviations: E-E = Patients whose NTX levels remained elevated at 3 months; E-N = Patients whose NTX levels normalized at 3 months from elevated baseline levels; NSCLC = Non-small cell lung cancer; NTX = N-telopeptide of type I collagen; OST = Other solid tumors. Hirsh V. Presented at: 12 th World Conference on Lung Cancer; September 2 -6, 2007; Seoul, Korea. Abstract D 5 -07.
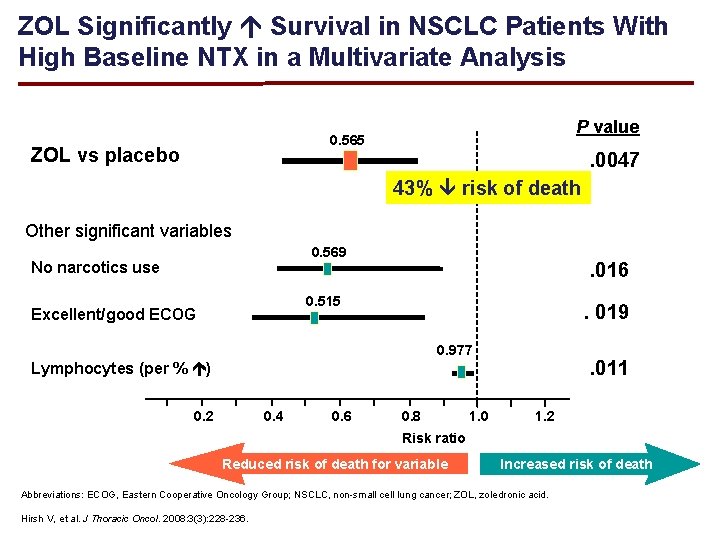
ZOL Significantly Survival in NSCLC Patients With High Baseline NTX in a Multivariate Analysis P value 0. 565 ZOL vs placebo . 0047 43% risk of death Other significant variables 0. 569 No narcotics use . 016 0. 515 Excellent/good ECOG . 019 0. 977 . 011 Lymphocytes (per % ) 0. 2 0. 4 0. 6 0. 8 1. 0 1. 2 Risk ratio Reduced risk of death for variable Increased risk of death Abbreviations: ECOG, Eastern Cooperative Oncology Group; NSCLC, non-small cell lung cancer; ZOL, zoledronic acid. Hirsh V, et al. J Thoracic Oncol. 2008: 3(3): 228 -236.

Potential Survival Benefits With Zoledronic Acid in Metastatic Bone Disease; META-ANALYSIS Coleman et al. J Bone Oncology 2013 : 2, 70 -76. QUESTION was; ‘Is survival improved by bisphonates in patients with a high rate of bone turnover? ’

Potential Survival Benefits With Zoledronic Acid in Metastatic Bone Disease; META-ANALYSIS Coleman et al. J Bone Oncology 2013 : 2, 70 -76. QUESTION was; ‘Is survival improved by bisphonates in patients with a high rate of bone turnover? ’ Independent individual patient level meta-analysis of placebo controlled trials of zoledronic acid 1642 patients – Prostate cancer – 643 patients – Breast cancer – 227 patients – Lung and other solid tumours – 772 patients – Overall survival RR, 0. 939; 95% CI, 0. 828 to 1. 064,
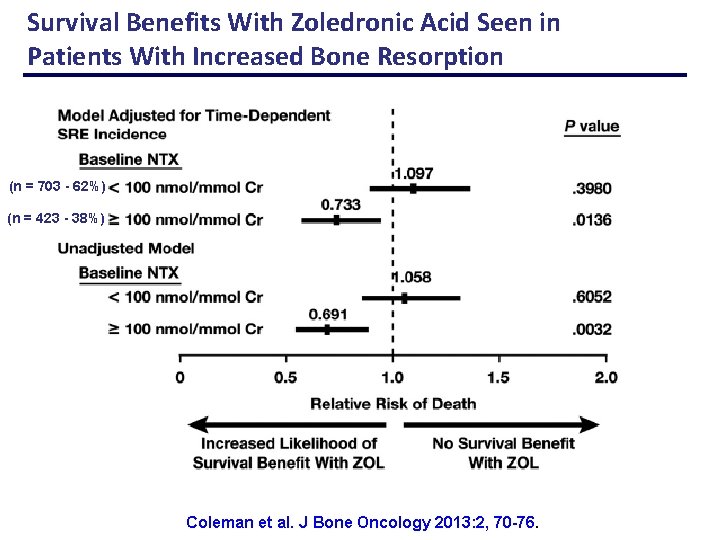
Survival Benefits With Zoledronic Acid Seen in Patients With Increased Bone Resorption (n = 703 - 62%) (n = 423 - 38%) Coleman et al. J Bone Oncology 2013: 2, 70 -76.
RANK Ligand • Member of the tumour necrosis factor (TNF) cytokine family 1 • Binds to receptor RANK 1 • Key factor for osteoclast differentiation and activation 1 • Physiological roles also observed beyond the bone 2 11 1. Lacey DL, et al. Nat Rev Drug Discov 2012; 11: 401 19; 2. Kong YY, et al. Nature 1999; 397: 315 23; 3. Dougall WC, et al. Genes Dev 1999; 13: 2412 24; 4. Rossi SW, et al. J Exp Med 2007; 204: 1267 72; 5. Hanada R, et al. Nature 2009; 462: 505 9; 6. Fata JE, et al. Nature 2000; 103: 41 50; 7. Ock S, et al. Cardiovasc Res 2012; 94: 105 14; 8. Schramek D, et al. Nature 2010; 468: 98 102; 9. Gonzáles-Suárez E, et al. Nature 2010; 468: 103 7; 10. Chen G, et al. Cancer 2006; 107: 289 98; 11. Brown JM, et al. Urology 2001; 57: 611– 6. RANK, receptor activator of nuclear factor kappa-B.

Three Identical Randomized Trials ; Zoledronic Acid vs Denosumab ; (each ‘arm’ Placebo § Adults with breast, prostate, or other solid tumors and bone metastases, or multiple myeloma § No current or previous IV bisphonate administration for treatment of bone metastases (N = 5723) Controlled) Denosumab 120 mg SC + Placebo IV* q 4 w (n = 2862) Supplemental calcium and vitamin D recommended Zoledronic Acid 4 mg IV* + Placebo SC q 4 w (n = 2861) 1° Endpoint § Time to first on-study SRE (noninferiority) 2° Endpoints § Time to first on-study SRE (superiority) § Time to first and subsequent on-study SRE (superiority) *Per protocol and zoledronic acid label, IV product dose adjusted for baseline creatinine. Lipton A, et al. ESMO 2010. Abstract 1249 P.
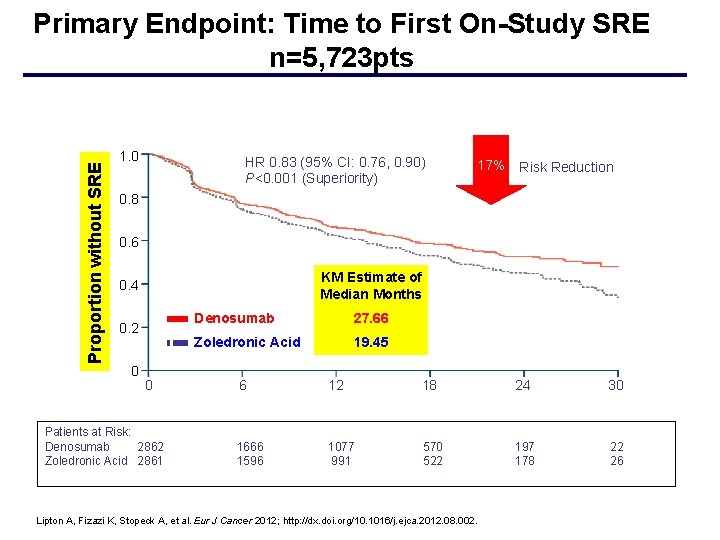
Proportion without SRE Primary Endpoint: Time to First On-Study SRE n=5, 723 pts 1. 0 HR 0. 83 (95% CI: 0. 76, 0. 90) P<0. 001 (Superiority) 17% Risk Reduction 0. 8 0. 6 KM Estimate of Median Months 0. 4 0. 2 0 0 Denosumab 27. 66 Zoledronic Acid 19. 45 6 12 18 24 30 570 522 197 178 22 26 Month Patients at Risk: Denosumab 2862 Zoledronic Acid 2861 1666 1596 1077 991 Lipton A, Fizazi K, Stopeck A, et al. Eur J Cancer 2012; http: //dx. doi. org/10. 1016/j. ejca. 2012. 08. 002.
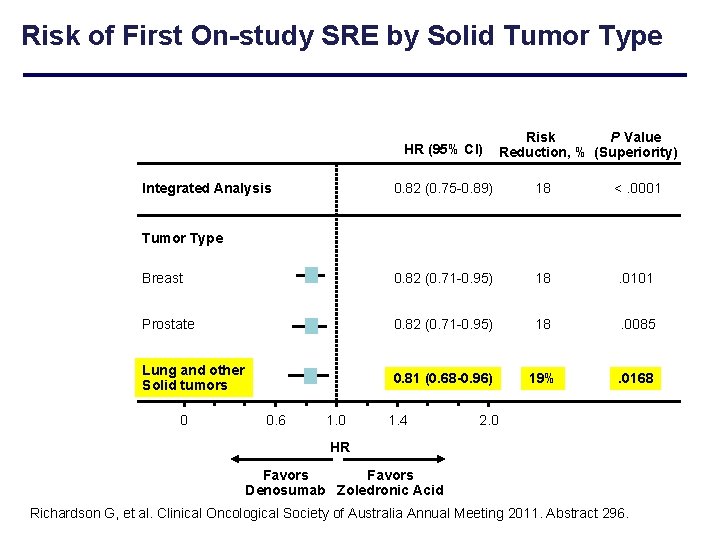
Risk of First On-study SRE by Solid Tumor Type HR (95% CI) Integrated Analysis Risk P Value Reduction, % (Superiority) 0. 82 (0. 75 -0. 89) 18 <. 0001 Breast 0. 82 (0. 71 -0. 95) 18 . 0101 Prostate 0. 82 (0. 71 -0. 95) 18 . 0085 Lung and other Solid tumors 0. 81 (0. 68 -0. 96) 19% . 0168 Tumor Type 0 0. 6 1. 0 1. 4 2. 0 HR Favors Denosumab Zoledronic Acid Richardson G, et al. Clinical Oncological Society of Australia Annual Meeting 2011. Abstract 296.
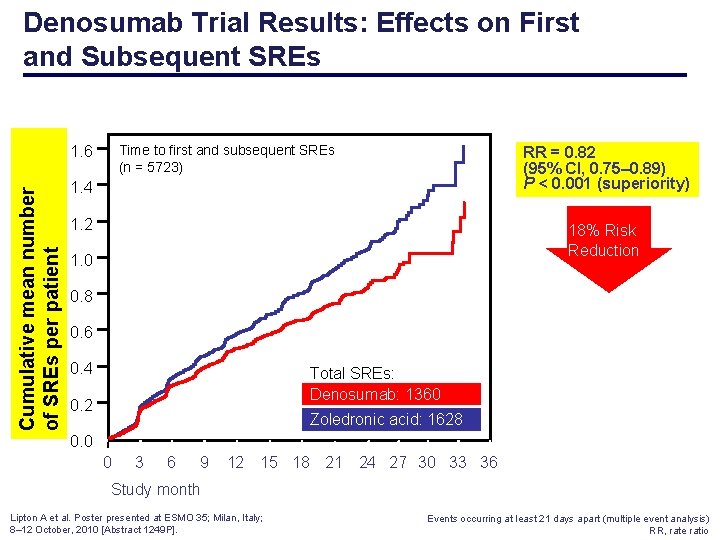
Denosumab Trial Results: Effects on First and Subsequent SREs Time to first and subsequent SREs (n = 5723) Cumulative mean number of SREs per patient 1. 6 RR = 0. 82 (95% CI, 0. 75– 0. 89) P < 0. 001 (superiority) 1. 4 1. 2 18% Risk Reduction 1. 0 0. 8 0. 6 0. 4 Total SREs: Denosumab: 1360 0. 2 Zoledronic acid: 1628 0. 0 0 3 6 9 12 15 18 21 24 27 30 33 36 Study month Lipton A et al. Poster presented at ESMO 35; Milan, Italy; 8– 12 October, 2010 [Abstract 1249 P]. Events occurring at least 21 days apart (multiple event analysis) RR, rate ratio
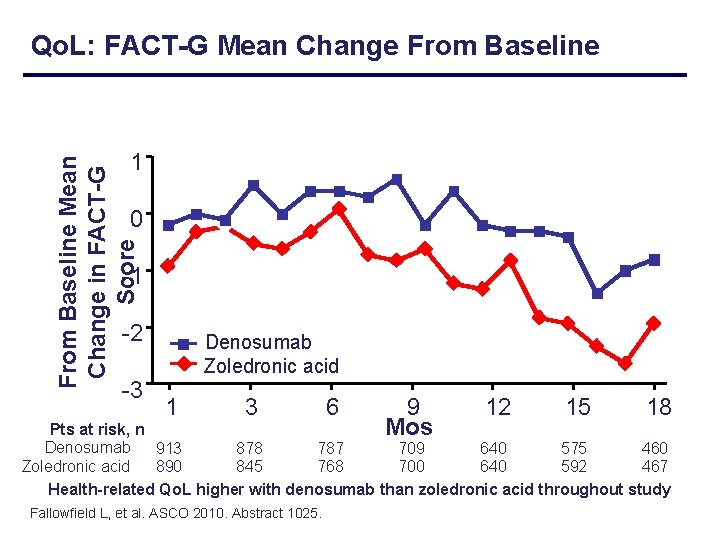
Qo. L: FACT-G Mean Change From Baseline Mean Change in FACT-G Score 1 0 -1 -2 -3 Denosumab Zoledronic acid 1 3 6 9 Mos 12 15 18 Pts at risk, n Denosumab 913 878 787 709 640 575 460 Zoledronic acid 890 845 768 700 640 592 467 Health-related Qo. L higher with denosumab than zoledronic acid throughout study Fallowfield L, et al. ASCO 2010. Abstract 1025.
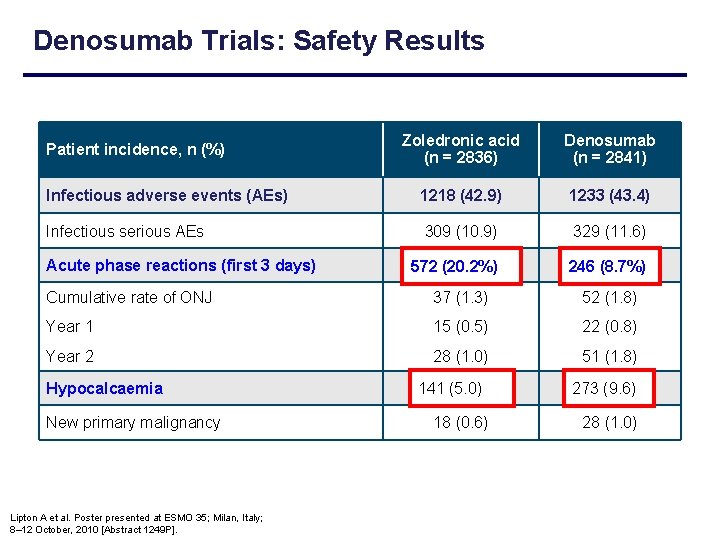
Denosumab Trials: Safety Results Zoledronic acid (n = 2836) Denosumab (n = 2841) Infectious adverse events (AEs) 1218 (42. 9) 1233 (43. 4) Infectious serious AEs 309 (10. 9) 329 (11. 6) 572 (20. 2%) 246(8. 7%) (8. 7) 246 Patient incidence, n (%) Acute phase reactions (first 3 days) Cumulative rate of ONJ 37 (1. 3) 52 (1. 8) Year 1 15 (0. 5) 22 (0. 8) Year 2 28 (1. 0) 51 (1. 8) 141(5. 0) 141 273(9. 6) 273 18 (0. 6) 28 (1. 0) Hypocalcaemia New primary malignancy Lipton A et al. Poster presented at ESMO 35; Milan, Italy; 8– 12 October, 2010 [Abstract 1249 P].
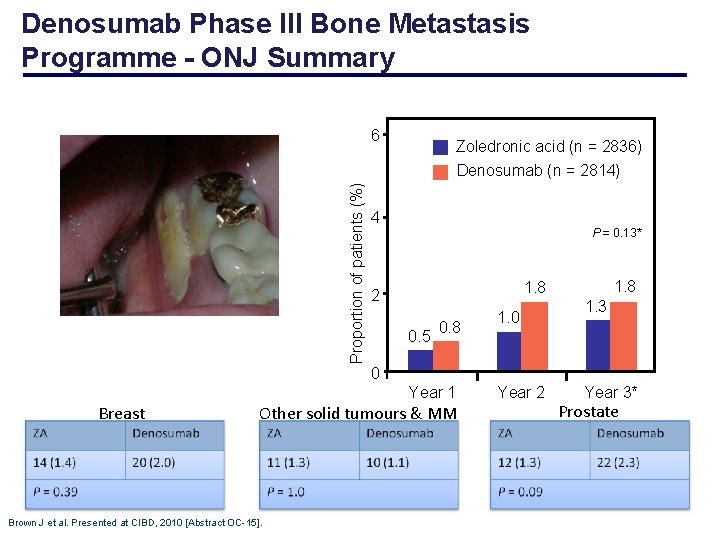
Denosumab Phase III Bone Metastasis Programme - ONJ Summary 6 Zoledronic acid (n = 2836) Proportion of patients (%) Denosumab (n = 2814) 4 P = 0. 13* 1. 8 2 0. 5 0. 8 1. 0 1. 3 0 Year 1 Breast Other solid tumours & MM Brown J et al. Presented at CIBD, 2010 [Abstract OC-15]. Year 2 Year 3* Prostate Proportions are % of all patients treated with zoledronic acid or denosumab
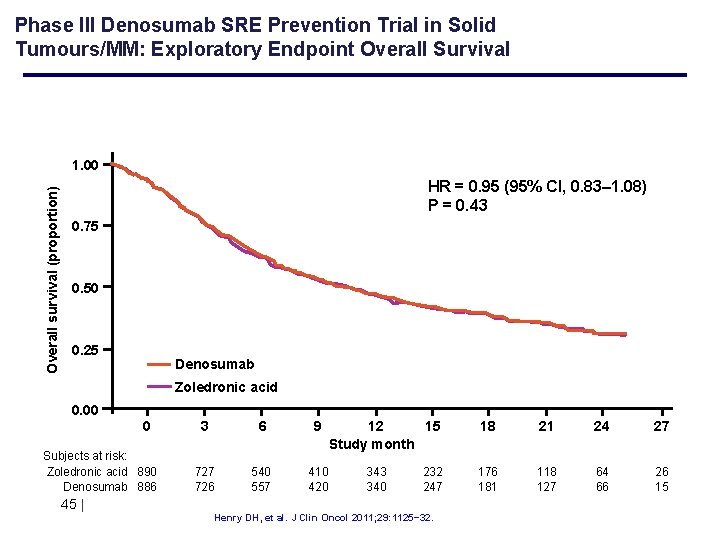
Phase III Denosumab SRE Prevention Trial in Solid Tumours/MM: Exploratory Endpoint Overall Survival Overall survival (proportion) 1. 00 HR = 0. 95 (95% CI, 0. 83– 1. 08) P = 0. 43 0. 75 0. 50 0. 25 Denosumab Zoledronic acid 0. 00 0 Subjects at risk: Zoledronic acid 890 Denosumab 886 45 | 3 6 9 727 726 540 557 410 420 12 15 Study month 343 340 232 247 Henry DH, et al. J Clin Oncol 2011; 29: 1125− 32. 18 21 24 27 176 181 118 127 64 66 26 15
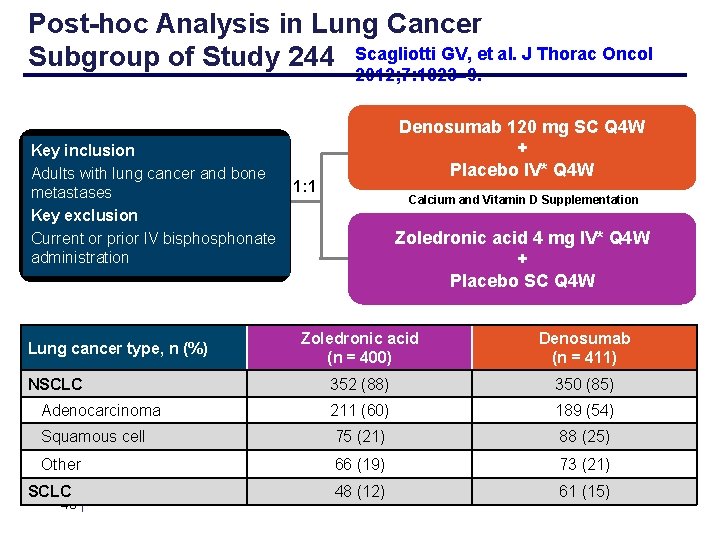
Post-hoc Analysis in Lung Cancer Subgroup of Study 244 Scagliotti GV, et al. J Thorac Oncol 2012; 7: 1823 9. Key inclusion Adults with lung cancer and bone metastases Key exclusion Current or prior IV bisphonate administration Denosumab 120 mg SC Q 4 W + Placebo IV* Q 4 W 1: 1 Calcium and Vitamin D Supplementation Zoledronic acid 4 mg IV* Q 4 W + Placebo SC Q 4 W Zoledronic acid (n = 400) Denosumab (n = 411) 352 (88) 350 (85) Adenocarcinoma 211 (60) 189 (54) Squamous cell 75 (21) 88 (25) Other 66 (19) 73 (21) 48 (12) 61 (15) Lung cancer type, n (%) NSCLC 46 |
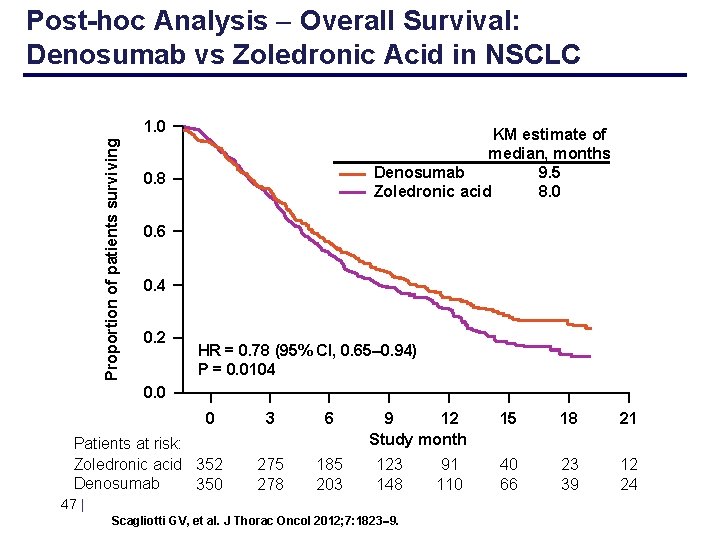
Post-hoc Analysis Overall Survival: Denosumab vs Zoledronic Acid in NSCLC Proportion of patients surviving 1. 0 KM estimate of median, months Denosumab 9. 5 Zoledronic acid 8. 0 0. 8 0. 6 0. 4 0. 2 HR = 0. 78 (95% CI, 0. 65– 0. 94) P = 0. 0104 0. 0 0 Patients at risk: Zoledronic acid 352 Denosumab 350 3 6 275 278 185 203 9 12 Study month 123 148 47 | Scagliotti GV, et al. J Thorac Oncol 2012; 7: 1823 9. 91 110 15 18 21 40 66 23 39 12 24

What is the Mechanism of the Observed Survival Benefit in the Denosumab Lung Cancer Subgroup? • Mechanism currently unknown • Possible explanations: Direct or indirect anticancer effect of denosumab NFk. B modulation through RANK in cancer cells Impact on micro-environment? Secondary consequence of SRE reduction Spurious result

SPLENDOUR A Randomised Phase III Trial Evaluating the Addition of Denosumab to Standard First-line Anticancer Treatment in Advanced NSCLC • Sponsor: European Thoracic Oncology Platform (ETOP) • Trial Coordinators: European Organization for Research and Treatment of Cancer (EORTC) Central European Cooperative Oncology Group (CECOG) • Pharma Partner: Amgen • PI: Solange Peters Survival im. Provement in Lung canc. Er i. Nduced by Den. Os. Umab the. Rapy (SPLENDOUR)
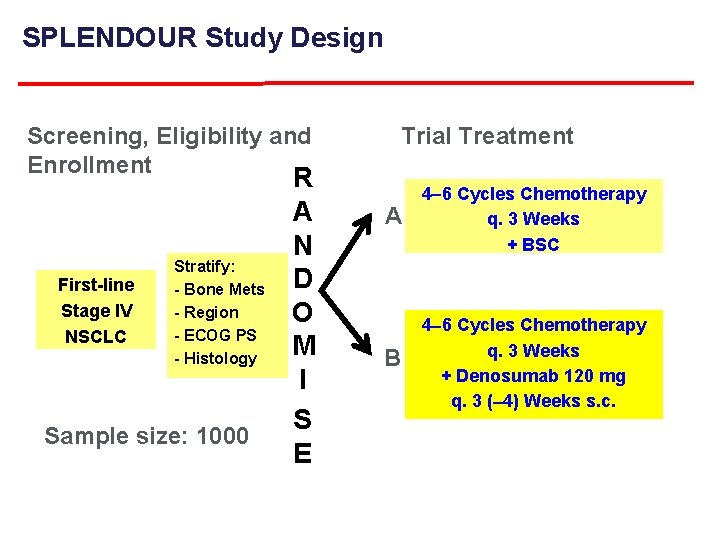
SPLENDOUR Study Design Screening, Eligibility and Enrollment First-line Stage IV NSCLC Stratify: - Bone Mets - Region - ECOG PS - Histology Sample size: 1000 R A N D O M I S E Trial Treatment A 4 6 Cycles Chemotherapy q. 3 Weeks + BSC B 4 6 Cycles Chemotherapy q. 3 Weeks + Denosumab 120 mg q. 3 ( 4) Weeks s. c.

Objectives and Endpoints Primary Objective Overall survival Secondary Objectives Progression free survival (PFS) (RECIST 1. 1) Toxicity profile of denosumab (CTCAE v 4) Determination of biomarkers for translational research

Translational Mandatory FFPE tissue (slides or block), serum and urine samples will be collected at baseline (prior to the start of chemotherapy), on day 1 of cycle 3 and at progression Serum analyses by ELISA include: Osteopontin (OPN); bone sialoprotein (BSP); RANK Ligand by ELISA kit designed for the quantitative determination of total (free RANK Ligand complexed to osteoprotegerin [OPG]) soluble RANK Ligand in serum and OPG levels Urine samples: Will be analysed for NTX • FFPE tumour samples: • Will be accessed for correlative research whenever possible. Evaluations will include: IHC for RANK Ligand RANK; NFk. B pathway components, and potentially BSP and OPN levels in primary tumour may correlate with tumour aggressiveness
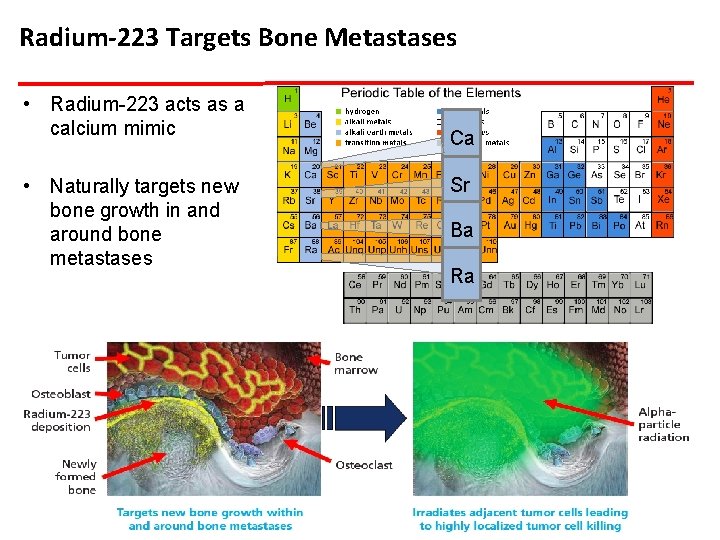
Radium-223 Targets Bone Metastases • Radium-223 acts as a calcium mimic • Naturally targets new bone growth in and around bone metastases Ca Sr Ba Ra
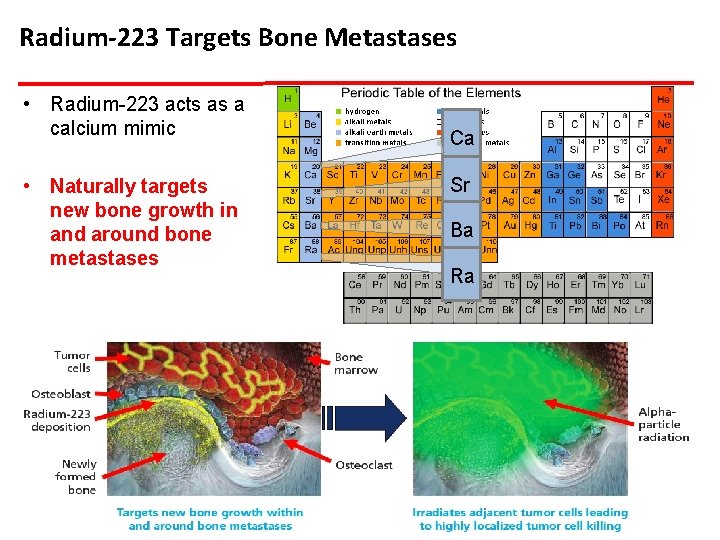
Radium-223 Targets Bone Metastases • Radium-223 acts as a calcium mimic • Naturally targets new bone growth in and around bone metastases Ca Sr Ba Ra
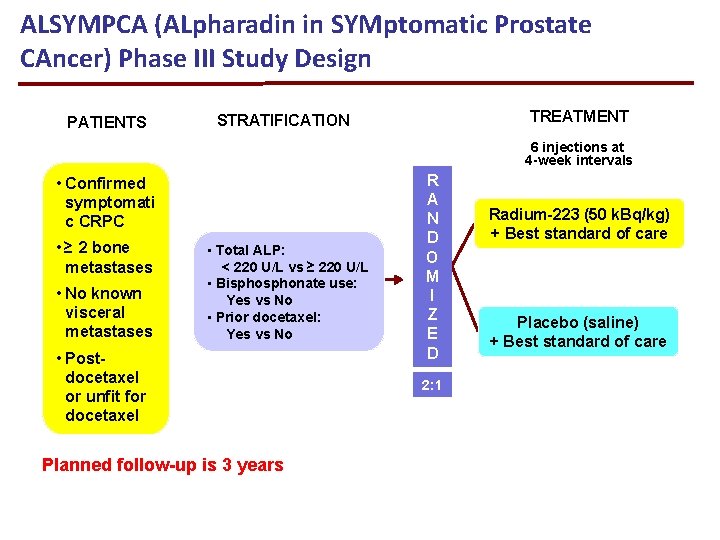
ALSYMPCA (ALpharadin in SYMptomatic Prostate CAncer) Phase III Study Design PATIENTS TREATMENT STRATIFICATION 6 injections at 4 -week intervals • Confirmed symptomati c CRPC • ≥ 2 bone metastases • No known visceral metastases • Total ALP: < 220 U/L vs ≥ 220 U/L • Bisphonate use: Yes vs No • Prior docetaxel: Yes vs No • Postdocetaxel or unfit for docetaxel Planned follow-up is 3 years R A N D O M I Z E D 2: 1 N = 921 Radium-223 (50 k. Bq/kg) + Best standard of care Placebo (saline) + Best standard of care
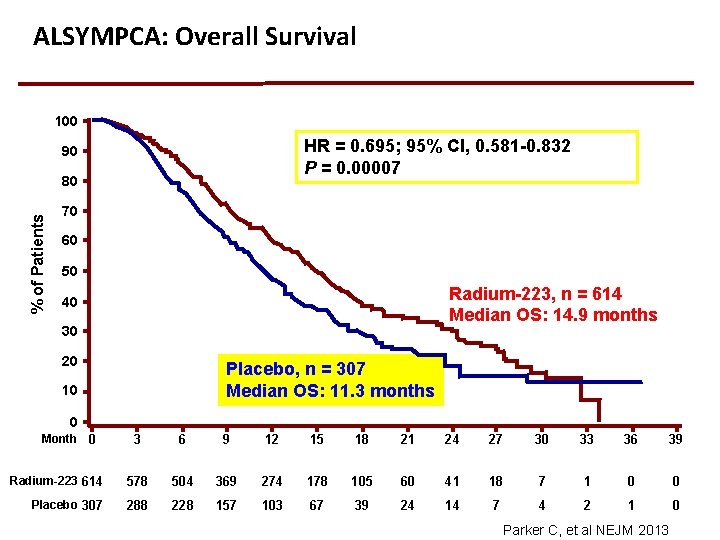
ALSYMPCA: Overall Survival 100 HR = 0. 695; 95% CI, 0. 581 -0. 832 P = 0. 00007 90 % of Patients 80 70 60 50 Radium-223, n = 614 Median OS: 14. 9 months 40 30 20 Placebo, n = 307 Median OS: 11. 3 months 10 0 Month 0 3 6 9 12 15 18 21 24 27 30 33 36 39 Radium-223 614 578 504 369 274 178 105 60 41 18 7 1 0 0 Placebo 307 288 228 157 103 67 39 24 14 7 4 2 1 0 Parker C, et al NEJM 2013

Bone Metastases Have Debilitating Consequences Disease Skeletal-related events Loss of autonomy Fracture Radiation to bone Bone metastases Consequences Ultimate consequence Spinal cord compression Hypercalcemia Surgery to bone Kinnane N. Eur J Oncol Nurs. 2007; 11(suppl): S 28 -S 31. Significant morbidity Bone pain Increased healthcare costs and resources Decreased survival

- Slides: 58