BONDS Chemical bonding Joining of atoms to form

BONDS

Chemical bonding • Joining of atoms to form new substances • A force of attraction that holds atoms together • Atoms bond to each other based on their valence electrons (electrons in outer energy level) • Atoms bond by gaining, losing, or sharing electrons to fill outer energy level with 8 electrons. (except H and He which need 2)

Three Types of Bonds • Ionic • Covalent • Metallic

Ionic bonds – loss and gain • Ion- charged particles, a charged atom • Ionic bonds are a force of attraction between ions • In ionic bonding a transfer of electrons occurs, an atom gains electrons while the other loses electrons • Bonds between metals and nonmetals

Example of ionic bonds • Static cling, casts, and seashells • Salt (Na. Cl)- Sodium(Na) is in group 1, chloride(Cl) is in group 7 • Losing an electron makes the atom a positive ion, gaining turns it into a negative ion. But combined the compound is neutral. • Usually metal to nonmetal : the metal loses the electron and the nonmetal gains it

Covalent Bonds - share • Co means to share • Electron dot diagram is used to represent shared electrons • These bonds are called molecules • Shared electrons are attracted to the nuclei of all atoms involved • Ex. Water (H 2 O), sugar, carbon dioxide (CO 2) • Nonmetal to nonmetal

Metallic - Pool • Form a pool of electrons which gives metallic bonds unique characteristics • These bonds are malleable (can be hammered into thin sheets) and ductile (can be drawn into a wire) • The pooling (free moving )allows electrons to flow easily so they don’t break and carry electricity well • Ex. Copper, aluminum, gold • Metal to metal

Electron Dot Diagram • Shows an atom and the electrons in its outer energy level • Shows symbol of atom and dots around it to represent electrons • Remember, group/family # tells us how many valence electrons (which tells us what type of bond will form)

How to write electron dot diagram • Start by putting one dot at the top of the symbol • Then add dots around the symbol in the shape of a box (one the right, bottom, left) • Add fifth dot to top to make a pair • * you should never have more than eight dots around the symbol
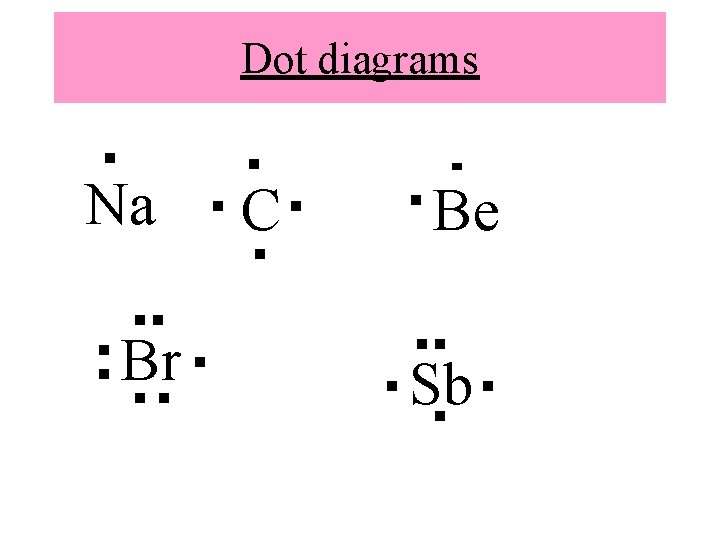
Dot diagrams Na Br C Be Sb
- Slides: 10