Bonds Atoms Protons positively charged Neutrons neutrally charged

Bonds!

Atoms • Protons- positively charged • Neutrons- neutrally charged • Electrons- negatively charged • Where does an atom’s mass come from?
Elements • Element- substance that is made up of only one type of atom • Four most common elements in nature include oxygen, nitrogen, carbon, and hydrogen
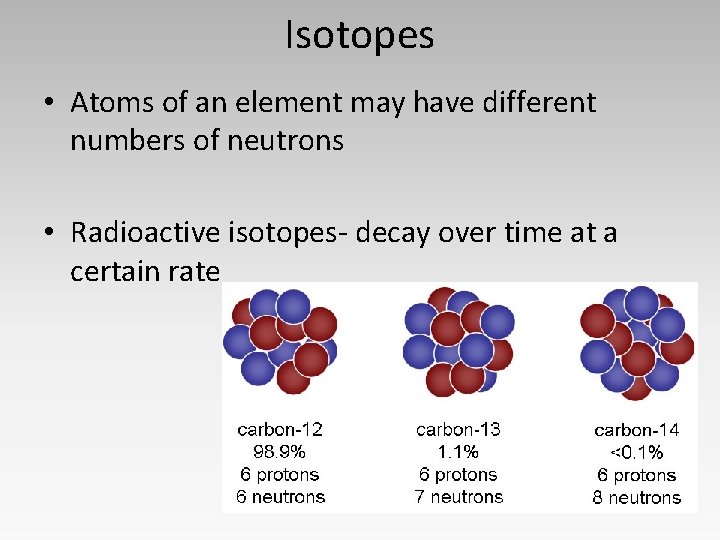
Isotopes • Atoms of an element may have different numbers of neutrons • Radioactive isotopes- decay over time at a certain rate
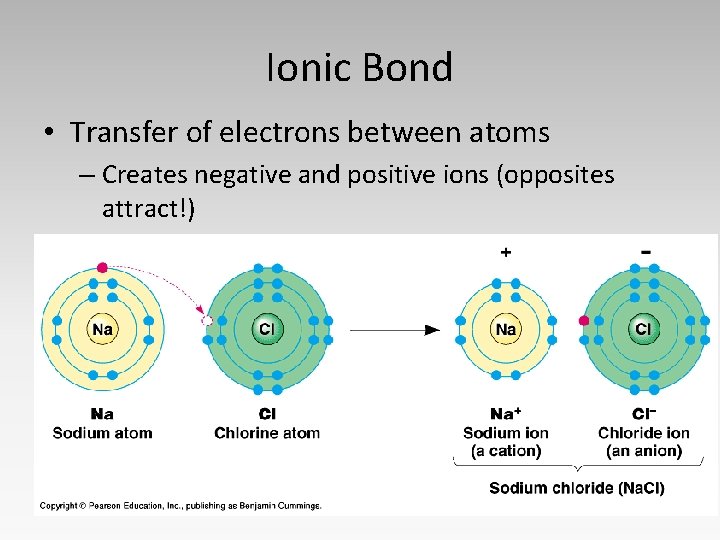
Ionic Bond • Transfer of electrons between atoms – Creates negative and positive ions (opposites attract!)
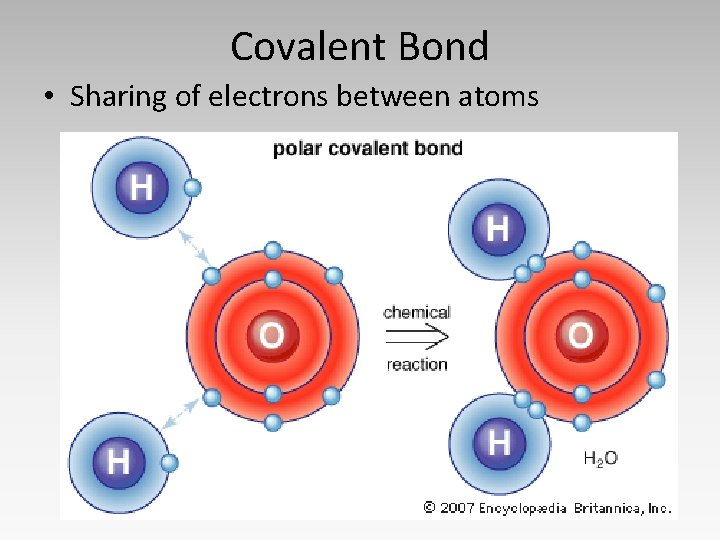
Covalent Bond • Sharing of electrons between atoms
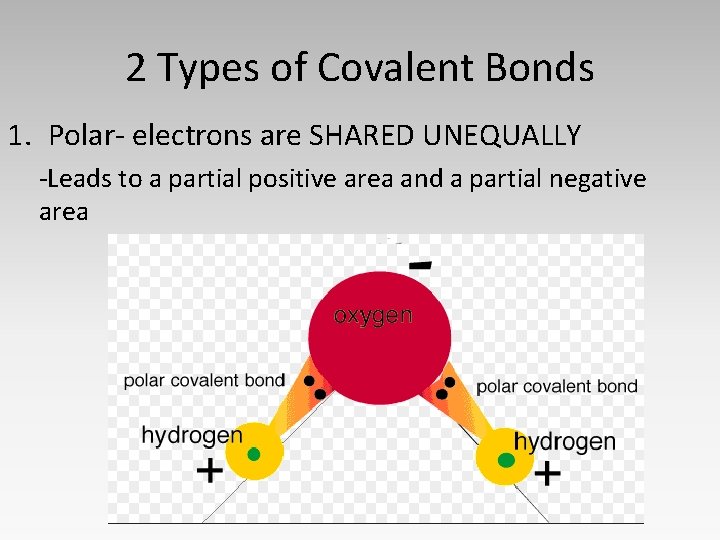
2 Types of Covalent Bonds 1. Polar- electrons are SHARED UNEQUALLY -Leads to a partial positive area and a partial negative area
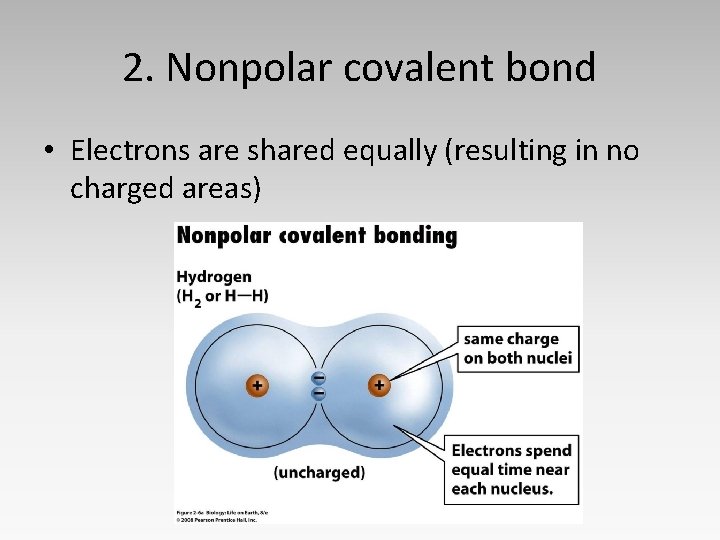
2. Nonpolar covalent bond • Electrons are shared equally (resulting in no charged areas)

Van der Waals Forces • Electrons are unevenly distributed around an atom. Causes positive and negative areas. – 2 atoms can bond temporarily because opposites attract!

Read pg. 39 • Connection to Real World • What are some situations in which adhesive bandages don’t stay on very well? • Why don’t bandages stay on well in these situations? • What do doctors typically use to hold together tissues after surgery? • Why might bandages be a better alternative?
- Slides: 10