Bonding Theories Hybrid Orbitals VSEPR does not describe

Bonding Theories

Hybrid Orbitals • VSEPR does not describe type of bonds • ORBITAL HYBRIDIZATION – provides information about molecular bonding and molecular shape • HYBRIDIZATION – several atomic orbitals mix to form the same total number of equivalent hybrid orbitals
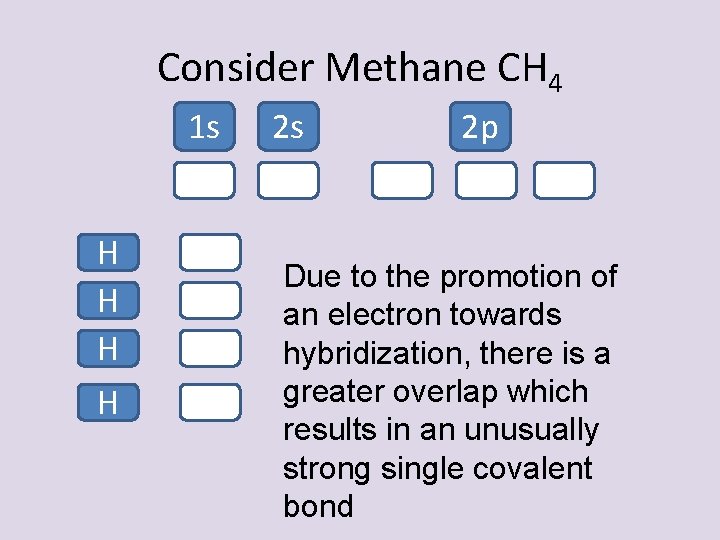
Consider Methane CH 4 1 s H H 2 s 2 p Due to the promotion of an electron towards hybridization, there is a greater overlap which results in an unusually strong single covalent bond

Bond Types • Covalent bonds differ in terms of how the bonded atoms share the electrons. • The character of the bonds in a given molecule depends on the kind and number of atoms joined together.

Three types of Bonds • Nonpolar covalent • Polar covalent • Ionic

Nonpolar Covalent Bonds • The bonding electrons are shared equally between the nuclei of the atoms sharing electrons • Examples: diatomic molecules • The electronegativity difference between the atoms is very small

Polar Covalent Bonds (Polar Bonds) • A covalent bond between atoms in which the e- are shared unequally • The more EN atom attracts more strongly and gains a slight (less than 1) negative charge.

• The less EN atom has a slight positive charge. • Example: Water – Oxygen has a much higher EN value than hydrogen – Oxygen will have a slight negative charge, hydrogen will have a slight positive one

THE REAL STORY • Three basic bond types are defined by the DIFFERENCE in electronegativity (ΔEN) between the atoms – Ionic ≥ 1. 7 – Polar Covalent 0. 6 – 1. 7 (unequal sharing) – Non-polar Covalent ≤ 0. 6
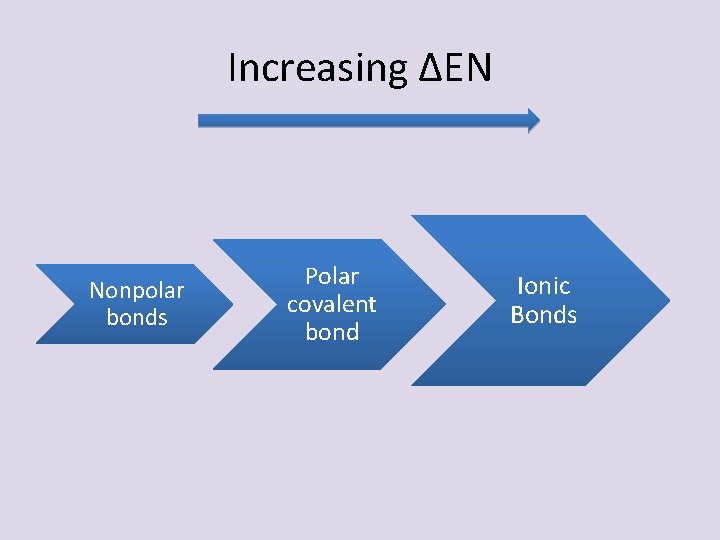
Increasing ΔEN Nonpolar bonds Polar covalent bond Ionic Bonds

• To find the DIFFERENCE… subtract the lower value from the higher value. • ΔEN = (larger EN)- (smaller EN)

Examples: Compound F 2 HF Li. F ΔEN 4. 0 - 4. 0 = 0 4. 0 - 2. 1 = 1. 9 4. 0 - 1. 0 = 3. 0 Type of Bond Nonpolar covalent Polar covalent Ionic (noncovalent)


• Generally, the element closest to fluorine will be relatively negative, and the element furthest from fluorine will be relatively positive • Polarity is indicated by •

• Indicate the positive and negative ends of each of the following bonds by using the symbols: • a. S-O • b. C-N • c. S-P • d. C-F • e. H-Br

• Geometry or shape of a molecule can be extended to larger molecules • Helpful in determining properties of compound

• Valence Shell Electron Pair VSEPR THEORY Repulsion. Theory Check out the site below to • Repulsion between find out more about electron pairs causes molecular geometry. http: //intro. chem. okstate. molecular shapes to adjust edu/1314 F 00/Lecture/Cha so that valence electron pter 10/VSEPR. html pairs stay as far apart as possible Remember: predicted shapes may be different • Unshared pairs of e- are from actual shapes. also important in predicting Draw Lewis Structures First shapes of molecules

Polar Molecules • One end of the molecule is slightly negative, the other end is slightly positive. • A molecule with two poles is called a dipolar molecule. • The effect of polar bonds on the polarity of an entire molecule depends on the shape of the molecule and the orientation of the polar bonds. Lets see why on the next page

Four General Rules for molecular polarity • 1. a molecule that is symmetrical is nonpolar, it does not matter how polar the individual bonds are (if all outer atoms are the same) • 2. a nonsymmetrical molecule is polar if the bonds are polar

a molecule with more that one type of atom attached to the central atom is often nonsymmetrical and therefore polar • 4. A central atom with nonbonding electron pairs is often nonsymmetrical and polar • 3.

Polar Molecules continued • Carbon dioxide has two polar bonds • It is linear • Bond polarities are on the same axis in opposite directions and will cancel…therefore it is nonpolar • Water has two polar bonds • It is bent • Bond polarities do not cancel, making this a polar molecule Remember: we are referring to the polarity of the entire molecule here, not just the individual bonds within the molecule

• Ex: construct the Lewis structure and predict the polarity of each of the following • A. CH 3 Cl • B. CS 2 • C. PH 3 • D. Si. F 4 • E. CO 32 -

• • • A. Polar with Cl as the negative end B. Nonpolar C. Polar with P at the negative end D. Nonpolar E. Nonpolar because of resonance, charge on the ion is distributed evenly over the ion – charged but not polar

Do Now • 1. If two atoms have a large difference in electronegativity, is the bond between them polar, nonpolar or ionic?
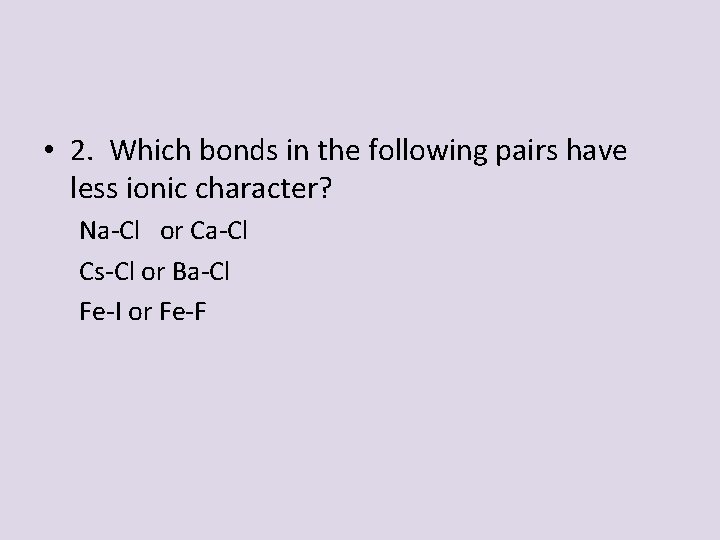
• 2. Which bonds in the following pairs have less ionic character? Na-Cl or Ca-Cl Cs-Cl or Ba-Cl Fe-I or Fe-F

• 3. For each of the following bonds, draw a figure indicating the direction of the bond dipole, including which end is positive and which is negative • H-C • N-O • N-S

Intermolecular attractions • intermolecular attractions are weaker than either ionic or covalent bonds. • They are, however, still important and can determine things like whether a molecular compound is a solid, liquid, or gas.
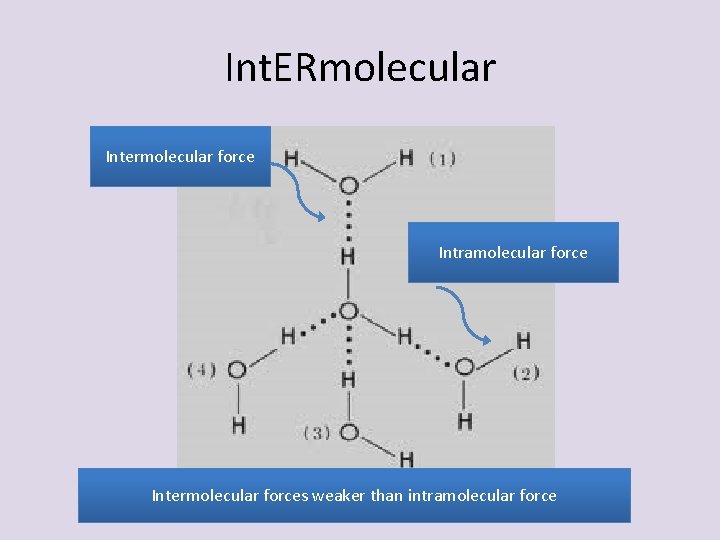
Int. ERmolecular Intermolecular force Intramolecular force Intermolecular forces weaker than intramolecular force

• Halogens change from gas, to liquid, to solid because of these forces • Responsible for physical and chemical properties of a substance – freezing point and boiling point

• use the force. . .
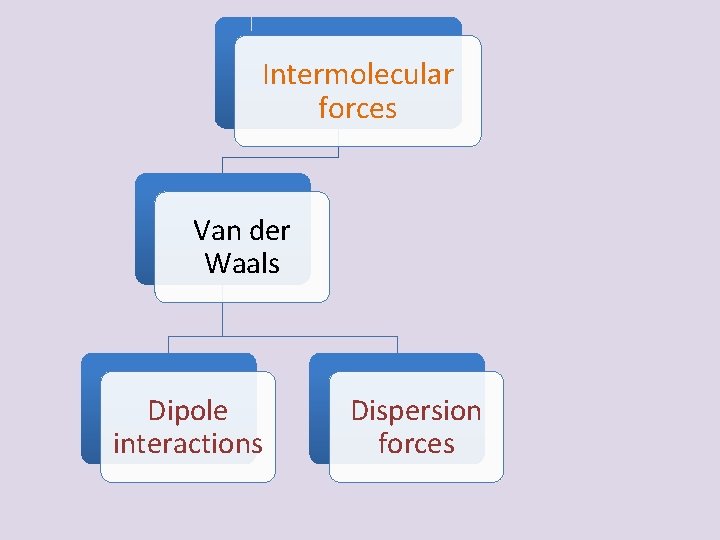
Intermolecular forces Van der Waals Dipole interactions Dispersion forces

Van der Waals Forces • Types of Van der Waals forces – Dipole interactions - occur when polar molecules are attracted to one another They are similar to but weaker than ionic bonds – Dispersion forces – weakest of the molecular interactions

Dipole interactions • Ion-Dipole- attractive forces between an ion and a polar molecule
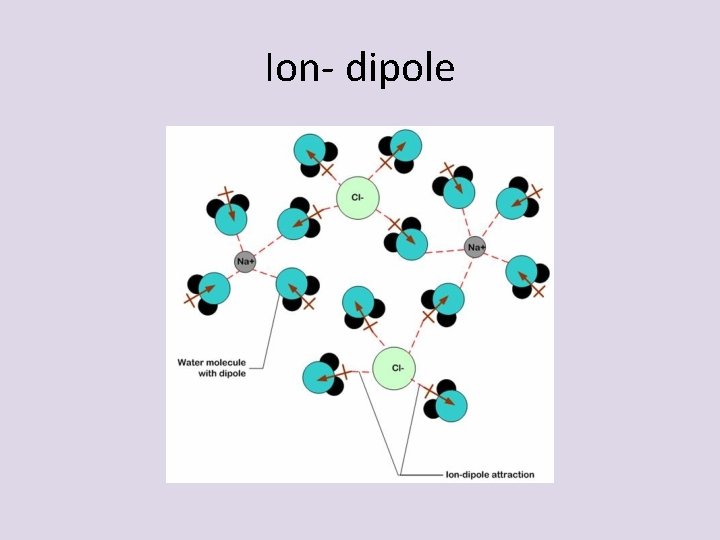
Ion- dipole

Dipole- Dipole • Dipole-Dipole interactions- attractive forces between the negative end of one POLAR molecule and the positive end of another POLAR molecule
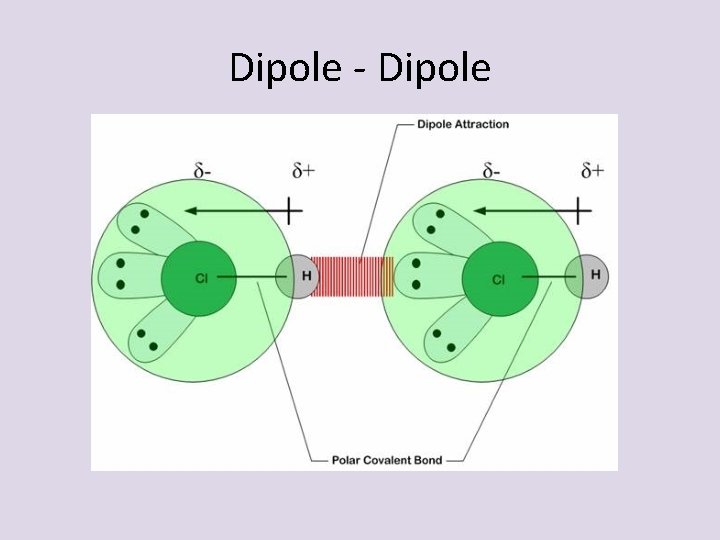
Dipole - Dipole

Hydrogen Bonds-type of dipole - dipole • Attractive forces in which a hydrogen covalently bonded to a very electronegative atom (F, O, N) is also weakly bonded to an unshared electron pair of another electronegative atom • 5 x stronger than average dipole-dipole

Hydrogen bond

Hydrogen bonding – Strongest intermolecular forces- why? - Always involves hydrogen- 1 e- and 1 p – Hydrogen’s valence e- are not shielded from the nucleus by a layer of underlying e– F, O, N have large electronegativity – lone pairs on these increase negative charge
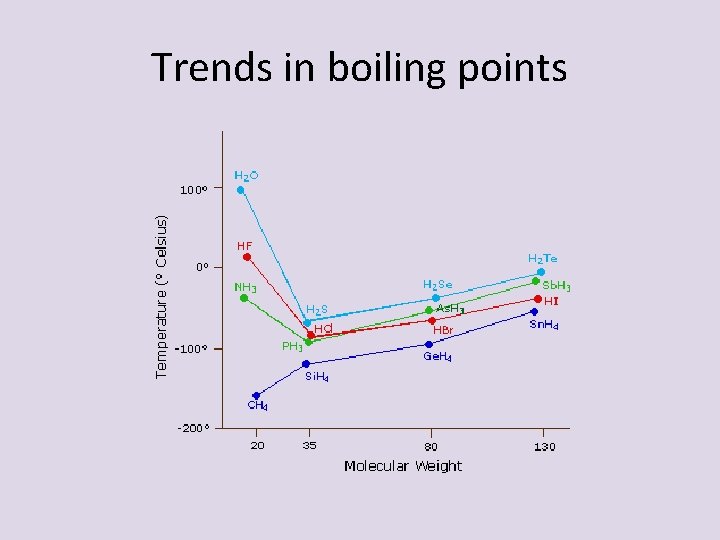
Trends in boiling points

Trends in boiling points • As you move down a group the molar mass increases – so does boiling point – Takes more energy to push a large atom into the gas phase – water and ammonia- have high bp because only molecules on the graph that have hydrogen bonding – More energy to break H-bonds and send into gas phase

Ion induced dipole • An ion can “induce” or “create” a dipole in a nonpolar species – Electrons are attracted or repel the nearby ion – Causes temporary increase in electron density on one side of the molecule – Then electrons re-align – Attraction constantly being turned on and off
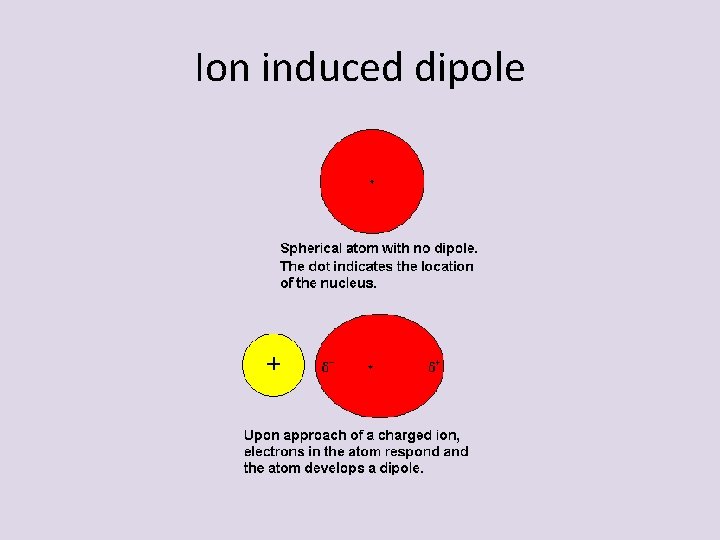
Ion induced dipole

Hemoglobin Globular protein 4 segments in tetrahedral – like arrangement 4 segments of protein structures - fold to form pocket contain non protein heme group Hydrogen bonds stabilize the helical sections inside this protein, causing attractions within the molecule, folding each polypeptide chain into a specific shape
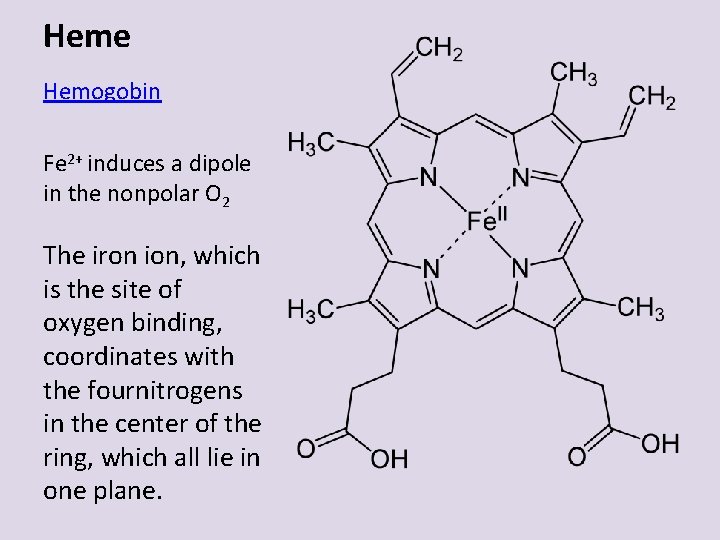
Heme Hemogobin Fe 2+ induces a dipole in the nonpolar O 2 The iron ion, which is the site of oxygen binding, coordinates with the fournitrogens in the center of the ring, which all lie in one plane.
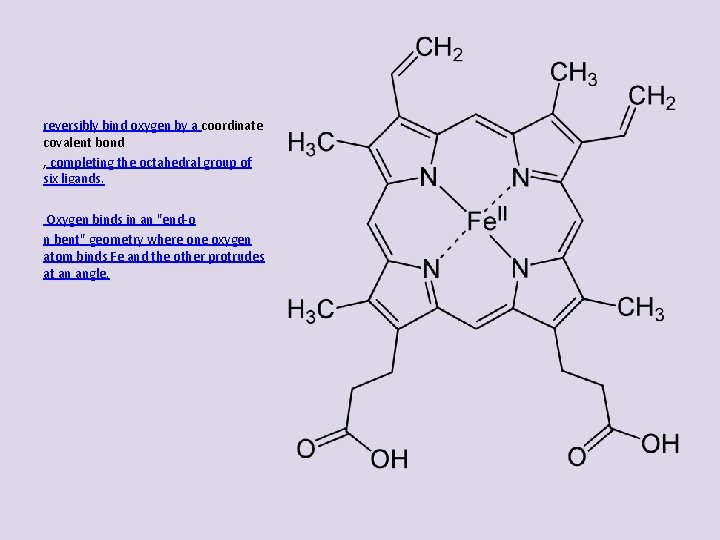
reversibly bind oxygen by a coordinate covalent bond , completing the octahedral group of six ligands. Oxygen binds in an "end-o n bent" geometry where one oxygen atom binds Fe and the other protrudes at an angle.

reversibly bind oxygen by a coordinate covalent bond completing the octahedral group of six ligands. Oxygen binds in an "end-on bent" geometry where one oxygen atom binds Fe and the other protrudes at an angle. octahedron.

• Dispersion Forces are the weakest of all molecular interactions. • As electrons move, they repel other electrons and cause them to move (a chain reaction of sorts) • The force increases as the # of eincreases

• www. youtube. com/watch? v=LGwy. Beu. Vjh. U&f eature=relatedhttp: //www. youtube. com/wat ch? v=LGwy. Beu. Vjh. U&feature=related • dispersion forces
• http: //www. youtube. com/watch? v=o. Swl. Z 4 Sio 9 Q&feature=related • Water video

Molecular Orbitals When two atoms combine, their atomic orbitals overlap and form molecular orbitals • Sigma Bonds σ • Pi Bonds π • When two atomic orbitals overlap side by • Orbitals overlap side by side to form a molecular • Electron density above orbital a sigma bond is and below the nuclei formed • Can be overlap of two s orbitals or one s and one p

• Single bond: sigma bond • Hybridization • Double bond: one sigma and one pi bond • Triple bond: one sigma and two pi

Hybrid orbital model Several atomic orbitals mix to form the same number of equivalent hybrid orbitals • hybridization makes the VSEPR work mathematically • orbital formation • sp, sp 2, and sp 3 • hybrid orbital- a set of orbitals formed from the combination of two or more atomic orbitals with different energies

The End
- Slides: 55