Bonding Naming Compounds http www skanschools orgwebpagesrallenatomictheory cfm

Bonding & Naming Compounds http: //www. skanschools. org/webpages/rallen/atomic_theory_. cfm? subpage=1179356

Compound • A substance that has two or more atoms different elements chemically bonded together

Bond • Forces of attraction between the protons (nucleus) of one atom and the electrons of another atom • Only valence electrons participate in bonding • Bonds are formed as a result of a chemical reaction

Law of Conservation • Conservation of energy/mass/charge • During a chemical reaction, energy, mass, and charge can not be created or destroyed (that means all three are conserved)

Bond • Octet rule: atoms bond together to get 8 valence electrons around them • Noble gases already have 8 valence electrons not reactive, inert

Energy & Bonding • Energy associated with bonding • Spontaneous bond formation= energy is released • Go from high energy to low energy • Creating a bond creates stability • Exothermic when energy is released as a product • A + B C + energy
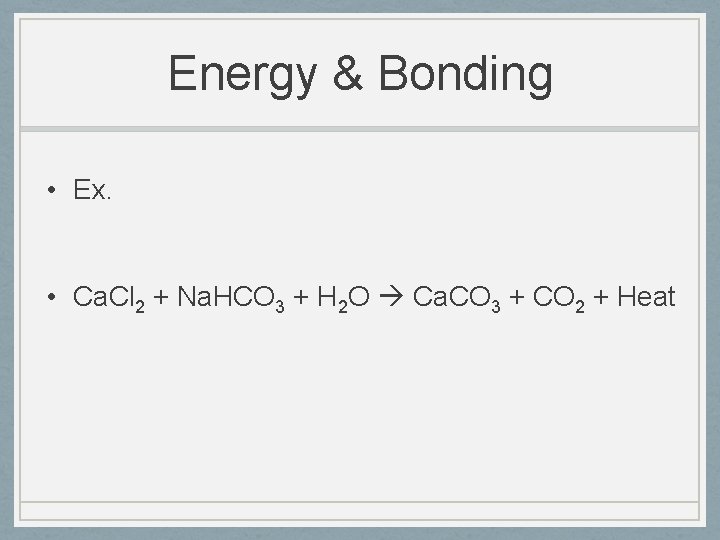
Energy & Bonding • Ex. • Ca. Cl 2 + Na. HCO 3 + H 2 O Ca. CO 3 + CO 2 + Heat

Energy & Bonding • Breaking Bonds (not spontaneous) • Energy is consumed • Go from low energy to higher energy • Breaking two atoms apart requires energy • Endothermic when energy is consumed or needed as an ingredient to fuel the process • A + energy B + C

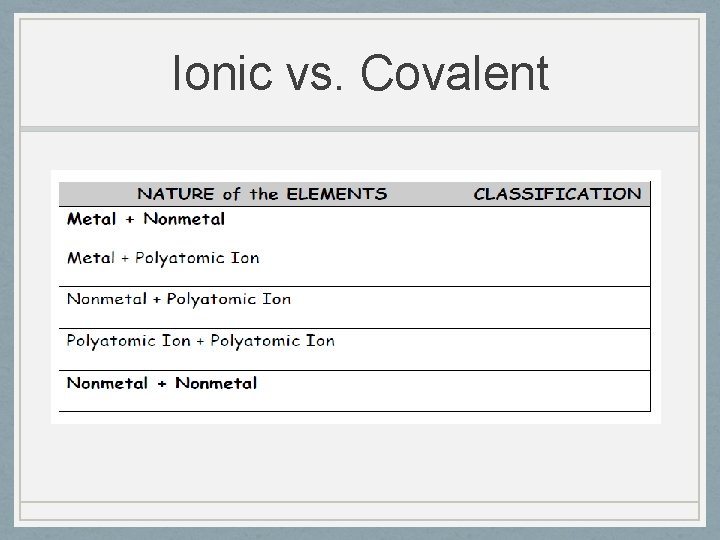
Types of Compounds • Ionic compounds formed by the transfer of electrons from one atom to another • A. Metal loses e- to a nonmetal
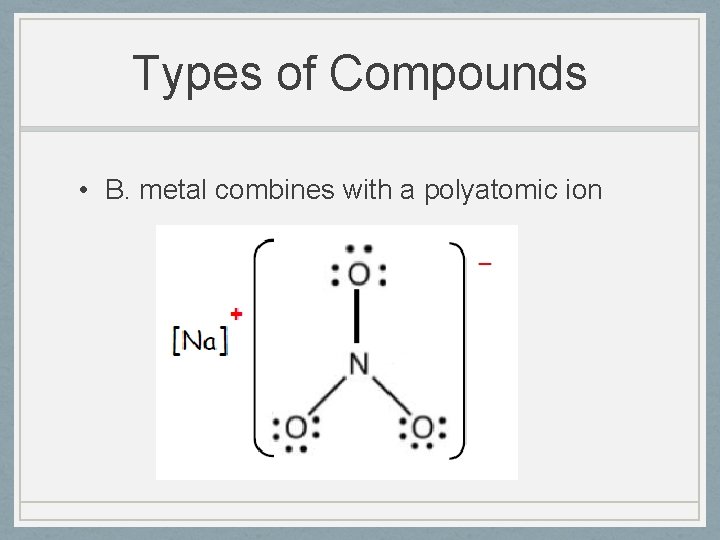
Types of Compounds • B. metal combines with a polyatomic ion
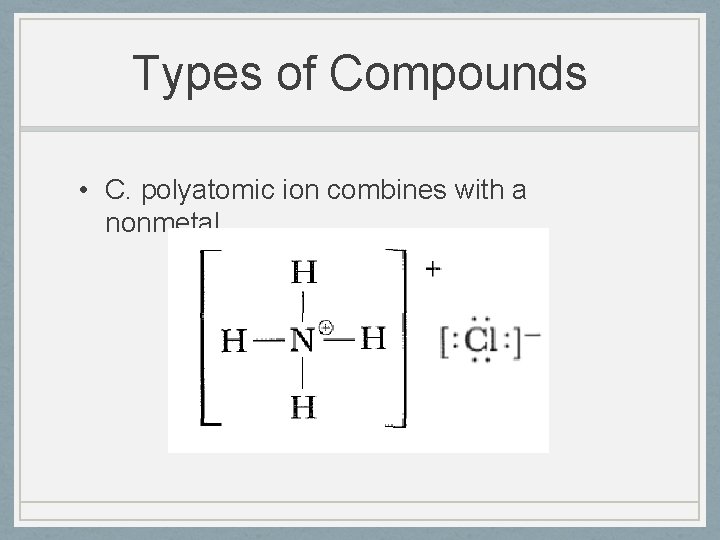
Types of Compounds • C. polyatomic ion combines with a nonmetal
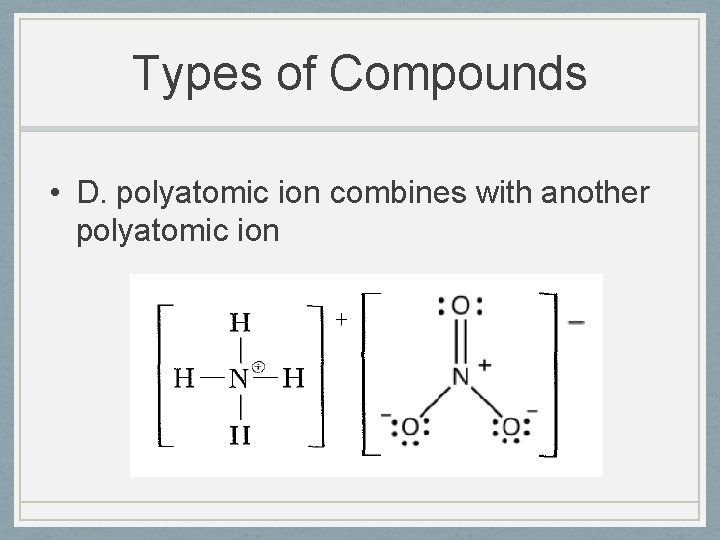
Types of Compounds • D. polyatomic ion combines with another polyatomic ion
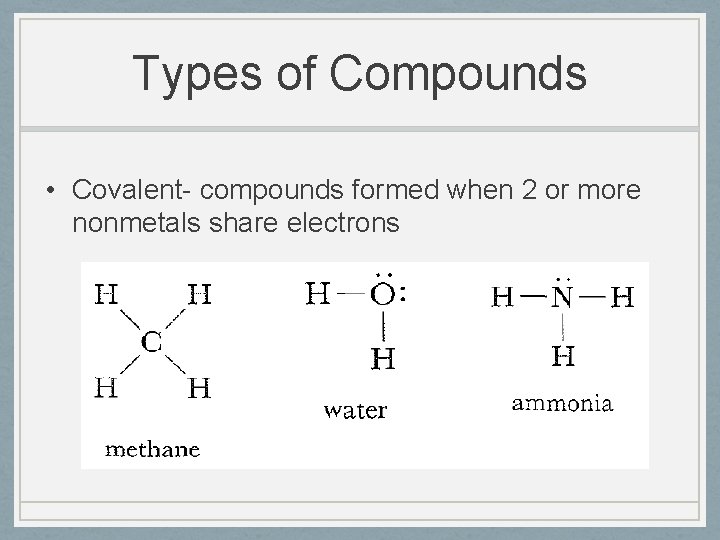
Types of Compounds • Covalent- compounds formed when 2 or more nonmetals share electrons
Ionic vs. Covalent

Metals • Pure substances found to the left of the staircase • Atoms of a metal do not bond with other metals • Metals share a sea of mobile valence electrons • Allows metals to conduct electric current (wave energy)

Ionic vs. Covalent • Ionic • Electrons transferred • Covalent • Electrons shared

Ionic vs. Covalent • Ionic: physical properties • High melting point/boiling point due to strong bonds • Electrolyte (conducts electric current in solution) • Hard

Ionic vs. Covalent • Covalent: physical properties • Low melting point/boiling point due to weak intermolecular forces • Nonelectrolyte (does not conduct electric current in solution) • Soft

Ionic vs. Covalent • The greater the electronegativity difference two elements the greater the percent ionic character • The closer the electronegativity difference is to zero, the greater the percent covalent character

Ionic vs. Covalent • We can classify a compound as ionic or covalent in one of two ways: • 1) Look at the elements in the compound • Metal and a nonmetal = ionic compound • Two nonmetals = covalent compound

Ionic vs. Covalent • 2) do the math: • The greater the electronegativity difference between the elements, the greater the ionic character • The closer the electronegativity difference between the elements, the greater the covalent character

Naming and Writing Chemical Formulas • Each element within a compound has its own charge • These charges are called oxidation numbers

Naming and Writing Chemical Formulas • Rules for assigning oxidation states (numbers) • 1) Uncombined elements (elements not bonded to any other type of element) have an oxidation number of zero • This includes any formula that has only one chemical symbol in it (single elements & diatomic elements)

Naming and Writing Chemical Formulas • 2) in compounds (they are neutral) the sum of the charges must add up to zero so the ions within a compound have oxidation numbers equal to their oxidation # found on the periodic table/individual charges • Usually the + element is written first and the - last

Naming and Writing Chemical Formulas • Group 1 Metals always have an oxidation number of +1 when in a compound (bonded to another species) • Group 2 metals always have a +2 oxidation number when located within a compound • Na. Cl Ca. Cl 2

Naming and Writing Chemical Formulas • 4) Fluorine is always a -1 in compounds. The other halogens are also -1 as long as they are the most electronegative element in the compound • Ex. Cu. F Ni. F 3 Cr. Cl 2 Fe. Br 3 • 5) Hydrogen is a +1 in compounds unless it is combined with a metal (and is at the end of the formula) then it is is -1 • Ex. HCl H 2 S Li. H Ca. H 2

Naming and Writing Chemical Formulas • 6) Oxygen is usually -2 in compounds • Ex. Fe 2 O 3 CO 2 H 2 O • When combined with fluorine (F), which is more electronegative, oxygen is +2 • Ex. OF 2 • When in a peroxide oxygen is -1. a peroxide is a compound that has a formula of X 2 O 2 • Ex. H 2 O 2 Na 2 O 2 Li 2 O 2

Naming and Writing Chemical Formulas • 7) the sum of the oxidation numbers in a polyatomic ions must equal the charge in the Ion (table E) • Compounds containing polyatomic ions have both ionic and covalent bonds

IUPAC Naming Rules • IUPAC – universal way of naming compounds • Compounds have a common name and a chemical name • There is a systematic method for naming ionic compounds

IUPAC Naming Rules for Ionic Compounds • Formula chemical name • 1. Name the positive element first (the metal) • 2. Name the negative element second (nonmetal) • Replace the ending of the negative element with “ide” • Ex. fluorine- fluoride • oxygen- oxide

IUPAC Naming Rules for Ionic Compounds • 3. Polyatomic ions are named as they are seen on Table E (periodic table) • Ex. Na. OH sodium hydroxide • KNO 3 Potassium Nitrate

IUPAC Naming Rules for Ionic Compounds • 4. Transition Metals usually have more than one oxidation number • Roman numerals are used to indicate oxidation number within a compound • The roman numeral appears in parenthesis after the element symbol • Ex. Cobalt Chloride • Co. Cl 2 • cobalt (II) chloride Co. Cl 3 cobalt (III) chloride

IUPAC Naming Rules for Covalent Compounds • Covalent nonmetal + nonmetal • Binary compound= 2 elements bonded together • Ternary compound= 3 elements bonded together • Similar to naming ionic compounds • Use prefixes to show many of each element you have in the covalent compound
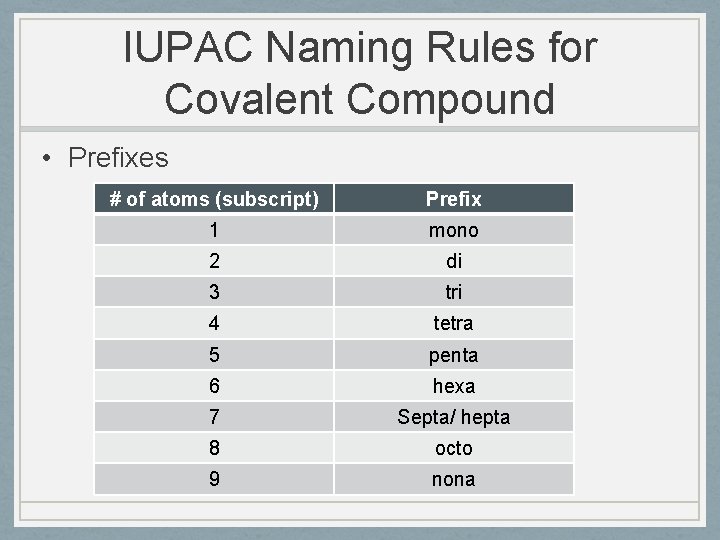
IUPAC Naming Rules for Covalent Compound • Prefixes # of atoms (subscript) Prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 Septa/ hepta 8 octo 9 nona

IUPAC Naming Rules for Covalent Compounds • One exception: drop the Mono prefix if there is only atom of the first element in the compound name • Final O’s or A’s of prefix are dropped when an element begins with a vowel • ex. Carbon Monoxide

IUPAC Naming Rules for Covalent Compounds • Writing the chemical formula • 1. least electronegative element is written first • 2. most electronegative is written last • Subscripts tell you the prefix of each element in the formula • Ex. CO 2 = carbon dioxide
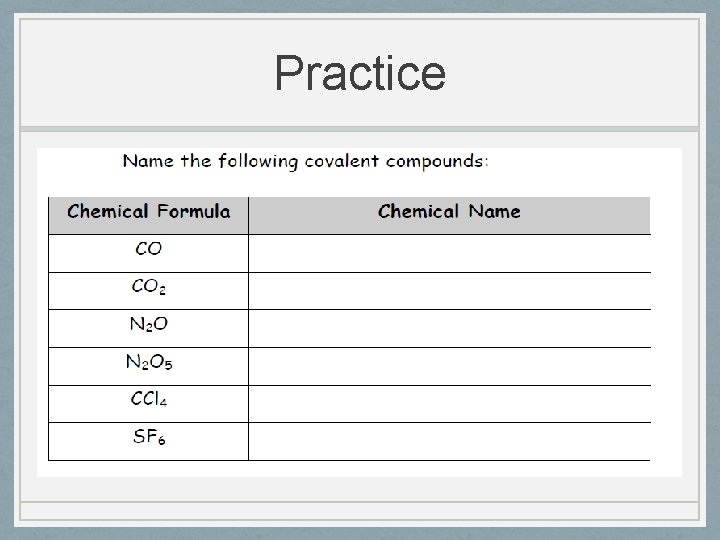
Practice
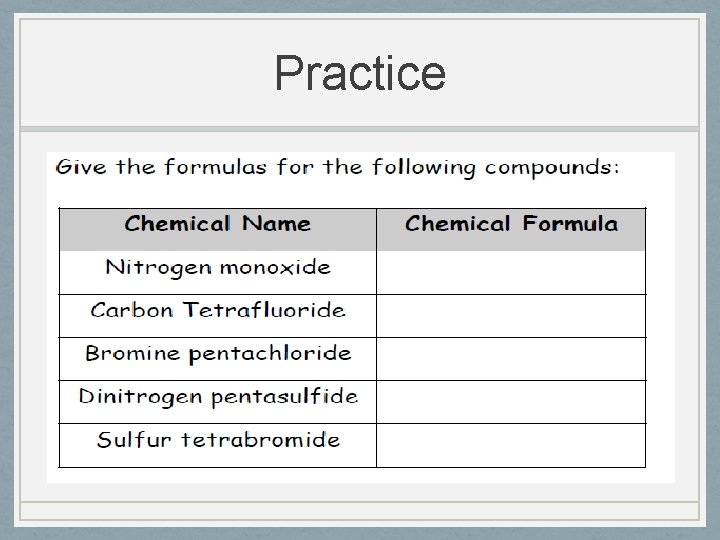
Practice

Lewis Dot Diagrams for Ionic Compounds • Within ionic compounds there is a transfer of electrons from metal to nonmetal • 1. Draw all elements and their individual Lewis Dot Diagrams • 2. Draw an arrow indicating the transfer of electrons from metal to nonmetal • 3. Redraw the bonded compound with dots and new charges

Lewis Dot Diagrams for Ionic Compounds • When you draw electrons from the metal to the nonmetal your goal is to match all unpaired electrons • There should be none left when diagram is done

Practice

Lewis Dot Diagrams for Covalent Compounds • 1. Write the element symbols and draw their valence electrons • If there are more than two atoms, find the element with the most unpaired valence electrons • This is the central atom • Ex. H 2 O

Lewis Dot Diagrams for Covalent Compounds • 2. Draw dash/lines (between the elements) connecting all lone electrons • Goal is to pair up ALL unpaired electrons
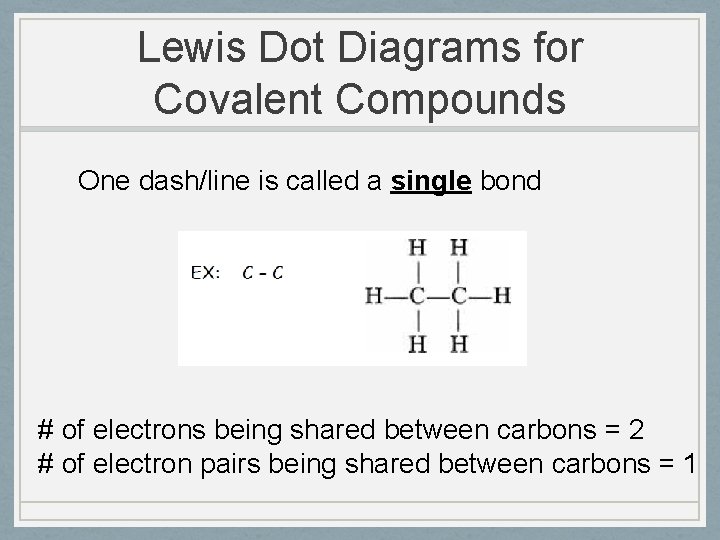
Lewis Dot Diagrams for Covalent Compounds One dash/line is called a single bond # of electrons being shared between carbons = 2 # of electron pairs being shared between carbons = 1
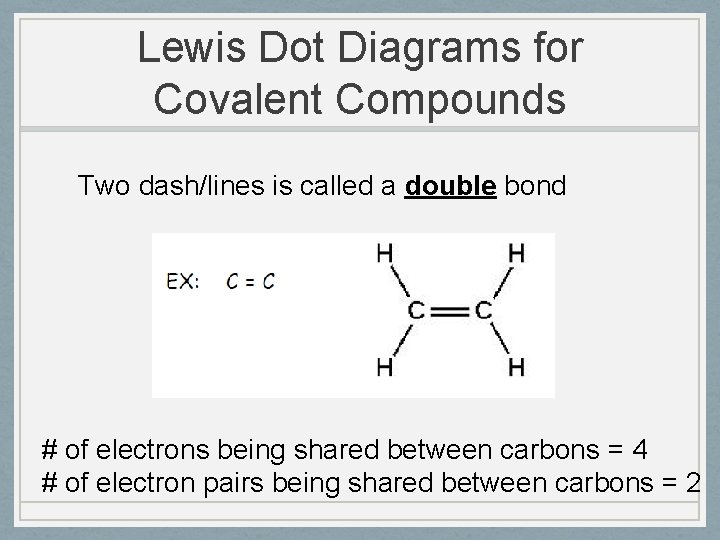
Lewis Dot Diagrams for Covalent Compounds Two dash/lines is called a double bond # of electrons being shared between carbons = 4 # of electron pairs being shared between carbons = 2
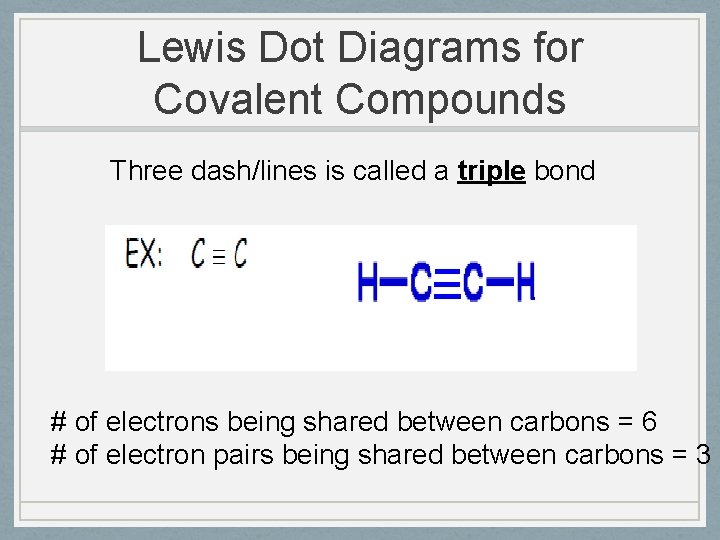
Lewis Dot Diagrams for Covalent Compounds Three dash/lines is called a triple bond # of electrons being shared between carbons = 6 # of electron pairs being shared between carbons = 3

Practice

Practice

Polar vs. Nonpolar • Molecule= covalently bonded substance; always 2 or more nonmetals bonded together • Molecular compound = covalent compound

Polar vs. Nonpolar • 1. Polar Molecule • Asymmetrical molecules • Unequal sharing of electrons • Doesn’t pass mirror test • Cant be folded to reflect itself • 2 atoms different elements/electronegativities • More than 2 atoms unbonded e- or lone pairs around the central atom
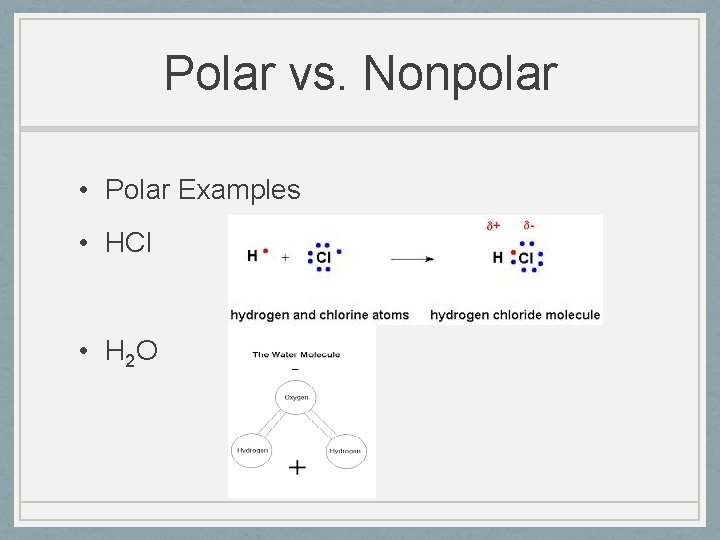
Polar vs. Nonpolar • Polar Examples • HCl • H 2 O

Polar vs. Nonpolar • 2. Nonpolar • Symmetrical • Equal sharing of electrons or • Does pass the mirror test • Can be folded to reflect itself • 2 atoms same element/electronegativities • More than 2 atoms no unbonded e- or lone pairs around the central atom
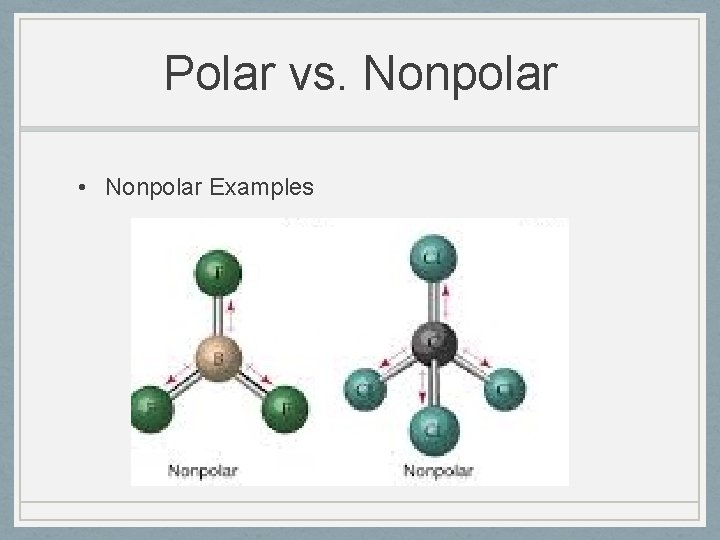
Polar vs. Nonpolar • Nonpolar Examples
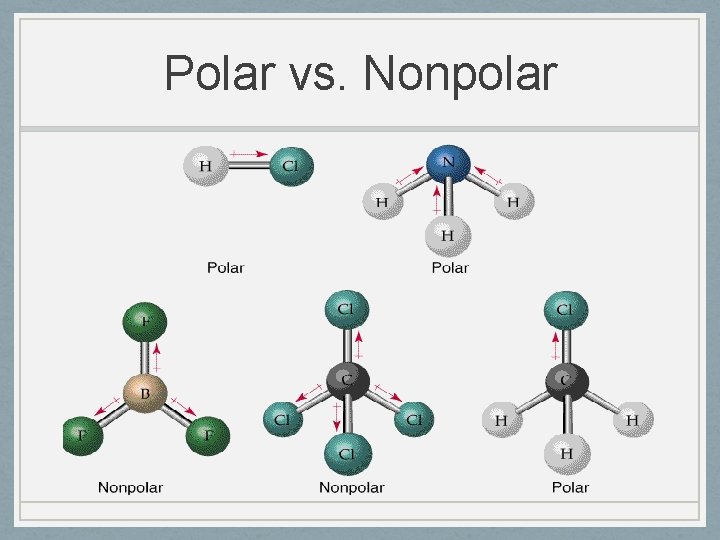
Polar vs. Nonpolar

Polar vs. Nonpolar • Can be polar bonds between elements in nonpolar molecules

Polar Covalent Bonds • Polar covalent bonds are a particular type of covalent bond. In a polar covalent bond, the electrons shared by the atoms spend a greater amount of time, on the average, closer to one element • Ex. H 2 O • Spend more time with oxygen

Nonpolar Covalent Bonds • Nonpolar covalent bonds are a type of bond that occurs when two atoms share a pair of electrons with each other. • These shared electrons glue two or more atoms together to form a molecule.

Intermolecular Forces • IMF • Only in covalent, never ionic compounds • Weak forces that act between molecules that hold molecules to each other • Only exists in gaseous and liquid state • Called weak forces because they are much weaker that chemical bonds

Intermolecular Forces • Remember: IMFs occur between molecules, whereas bonding occurs within molecules
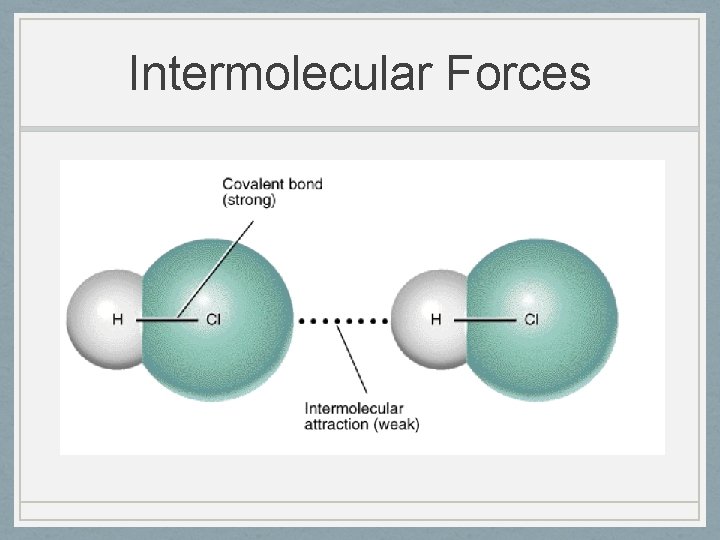
Intermolecular Forces

Intermolecular Forces • IMFs are not bonds

Intermolecular Forces Type of IMF London Dispersion Forces • • Description/Example Weakest of all IMFs Only important for nonpolar molecules More electrons= greater LDF Electron-electron repulsion creates brief dipoles in atoms/molecules

Intermolecular Forces Type of IMF Description/Example Dipole • Molecules such as HCl have both + (dipole-dipole) and – ends/poles • Two poles= dipole • Result from an unequal/asymmetrical sharing of electrons • Dipole-Dipole= 2 molecules with permanent dipoles are attracted to each other temporarily • Dipole moment= measure of the strength of the dipole within a molecule (polarity)

Intermolecular Forces • * the greater the electronegativity difference between atoms, the greater the polarity/dipole moment • *the higher the dipole moment, the stronger the intermolecular forces • *the higher the IMFs, the higher the melting/boiling points

Intermolecular Forces Type of IMF Hydrogen Bonds • • Description/Example Specific type of dipole interaction In a polar bond, hydrogen is basically reduced to a bare proton with almost no atomic radius Strongest of all IMFs Only occur in molecules containing hydrogen and fluorine, oxygen or nitrogen
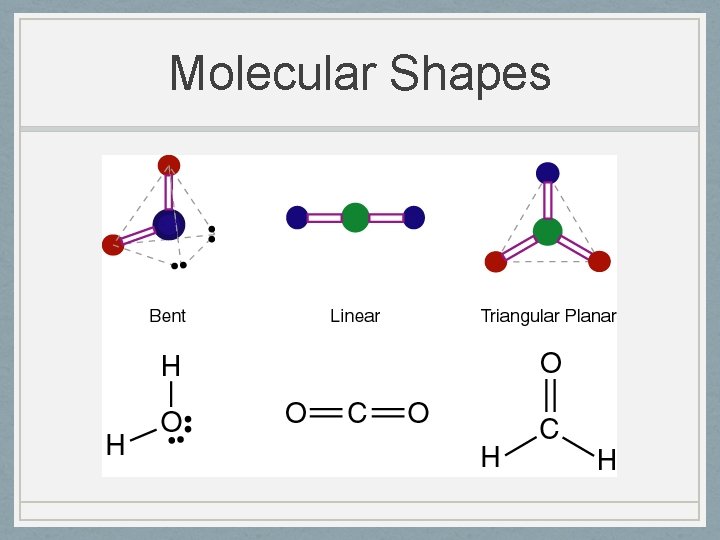
Molecular Shapes
- Slides: 66