BONDING Molecules and Covalent Bonding Part 2 Shapes

BONDING Molecules and Covalent Bonding Part 2 Shapes of Molecules and VSEPR Bond Strengths

Lewis structures are an excellent tool to help you determine the number of atoms, valence electron numbers and the placement and bonding of atoms or ions in a molecule. They give you no information about the shape of the molecule. One way of accomplishing this task is through VSEPR theory. VSEPR stands for: Valence Shell Electron Pair Repulsion The basic principle is: That electrons pairs surrounding a central atom in a molecule repel each other and that they will arrange themselves in such a way as to minimize these repulsions. Furthermore: lone pairs (LP) repel more than bonding pairs (BP) such that for repulsions: LP-LP > LP-BP > BP-BP

Steps for predicting molecular shape using VSEPR: 1. Draw a Lewis structure 2. Determine the VSEPR notation for that molecule 3. Find shape in chart (you will need to memorize this for the AP exam)
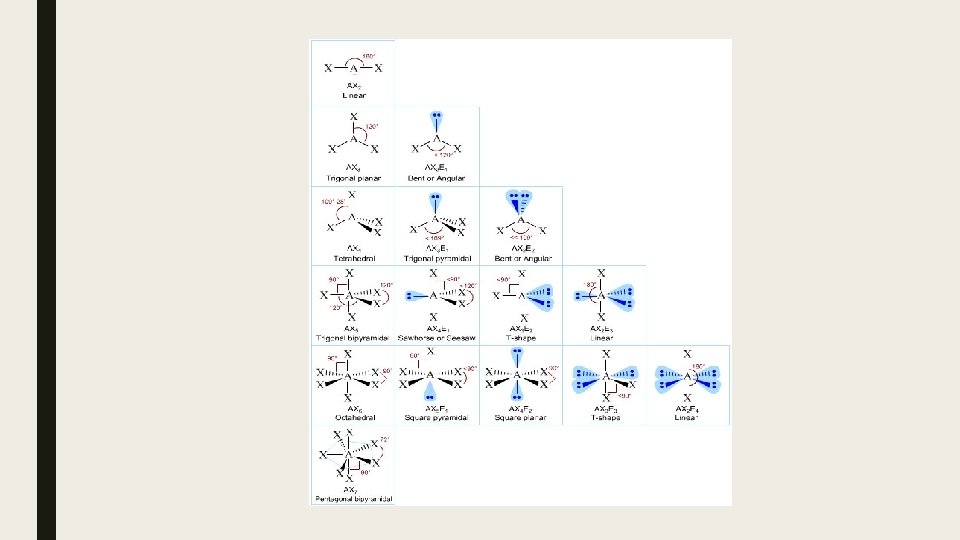

Examples: angles. a) PF 5 Predict and draw the shape of each of the following. Indicate the bond

b) Br. Cl 4 -

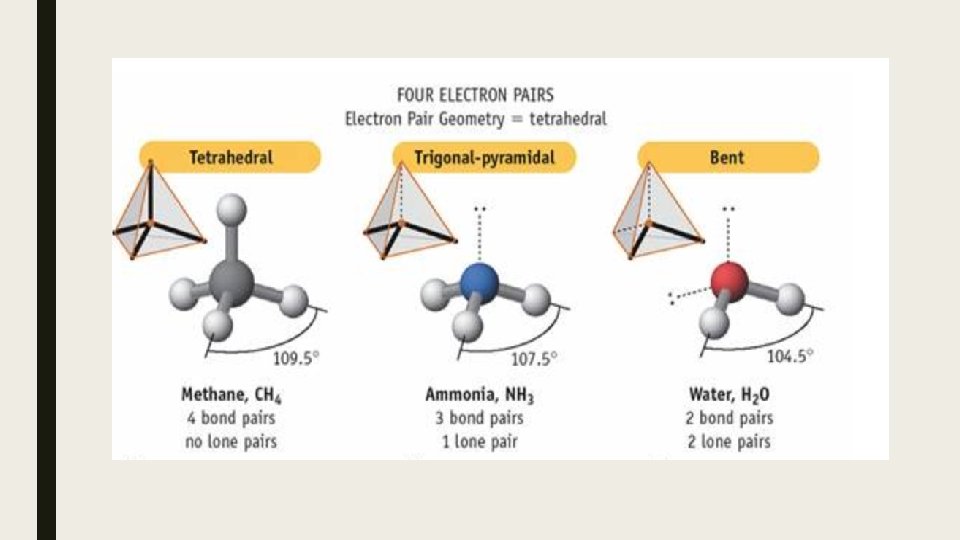
Bond Angles Effect of LP There are differences in angles are due to repulsions between lone pairs being greater than repulsions between bonding pairs – LP-LP repulsion > LP-BP repulsion > BP-BP repulsions – This is because the lone pair is on average closer to the nucleus than a bonding pair and therefore repels other electrons more strongly. – In these 3 molecules, all are sp 3 hybridized and their structure is based on a tetrahedron. However, since NH 3 has one BP and H 2 O has 2 BP, the angles are not all 109. 5 o.

For example: For the molecule SO 2 which of the following best describes the O-S-O angle: a) 120 o b) <120 o c) >120 o
AP practice questions

The Relationship between Molecular Shape and Molecular Polarity A molecule’s shape and polarity are directly related. ∆EN is the principle factor that determines the type of chemical bonding between two atoms. When a bond between two atoms is polar covalent, the electrons are shared unequally. Because one atom is more electronegative than the second atom in the bond, it has a greater attraction for the electrons shared in the bond. It develops a partial negative charge ( d- ) and the atom that is less electronegative develops a partial positive charge ( d+ ). This polarity in a bond is called a dipole, and ∆EN is often referred to as the dipole moment. The greater the ∆EN, the greater the dipole moment.
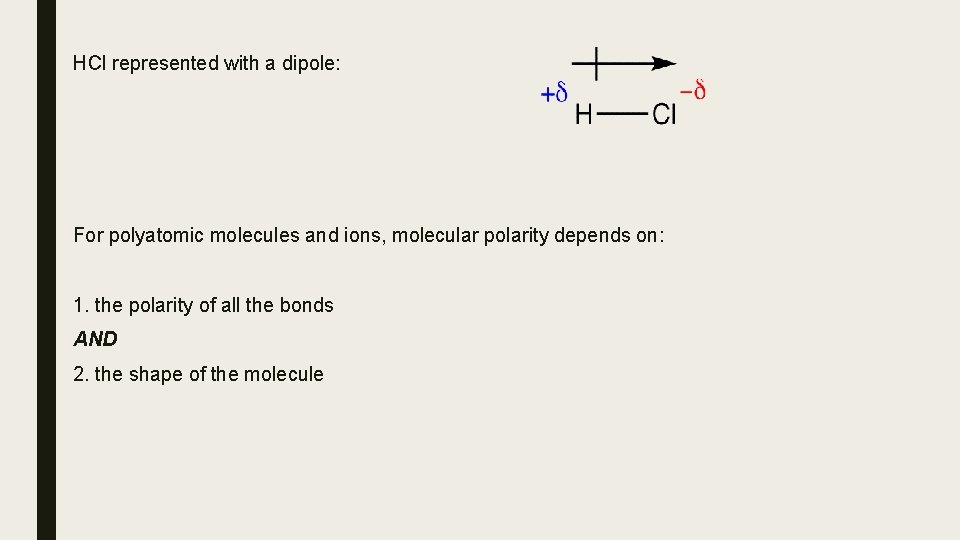
HCl represented with a dipole: For polyatomic molecules and ions, molecular polarity depends on: 1. the polarity of all the bonds AND 2. the shape of the molecule

Molecules must have polar bonds AND two distinct “sides” for them to be polar. Examples: H 2 S, CCl 4 and CHF 3

AP practice question

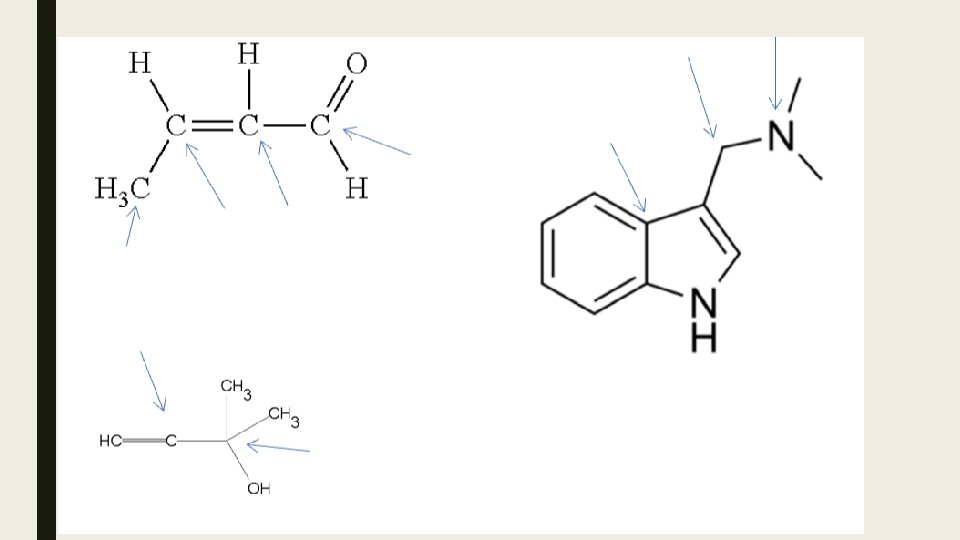
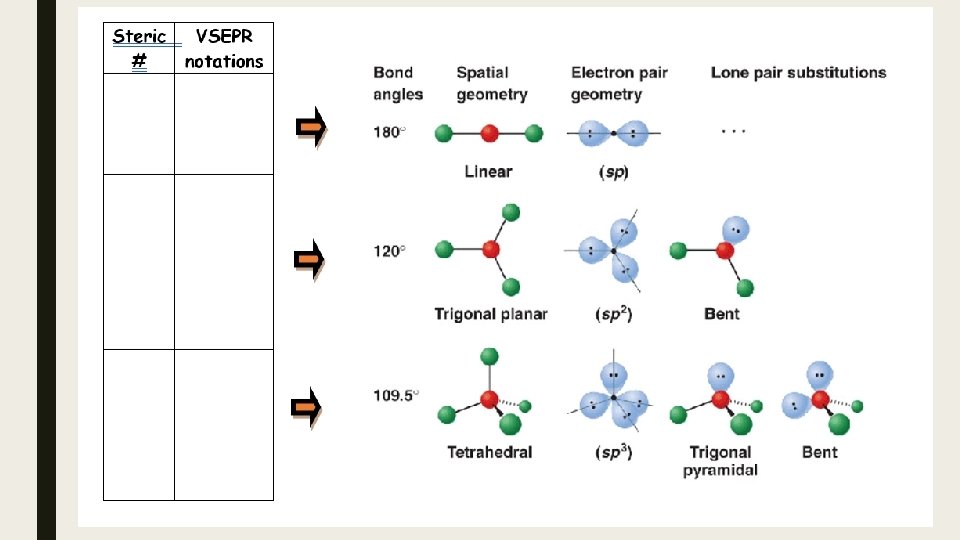
Hybrid Orbitals and Molecular Shape Hybrid orbitals are used to describe orbitals during bonding. Each of these types of hybrid orbitals is associated with specific VSEPR notations (and from there specific molecular shapes). Steric number is the number of atoms bonded to a central atom of a molecule plus the number of lone pairs attached to the central atom. The steric number of a molecule is used in VSEPR (valence shell electron pair repulsion) theory to determine the molecular geometry of a molecule. VSEPR notations AX 2 Steric Basic shape number made by BP and LP 2 linear Hybrid orbital at A sp AX 3, AX 2 E 3 trigonal planar sp 2 AX 4, AX 3 E, AX 2 E 2 4 tetrahedral sp 3
AP practice questions
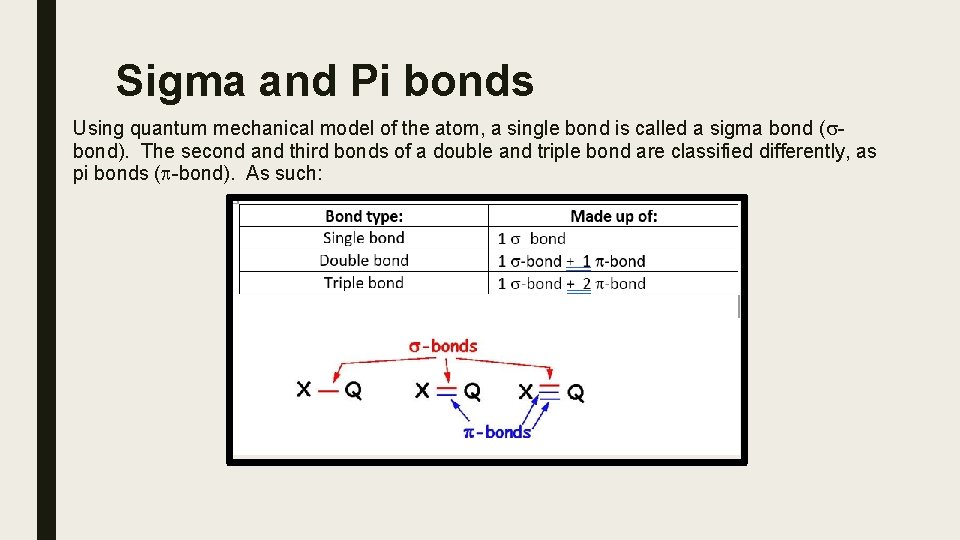
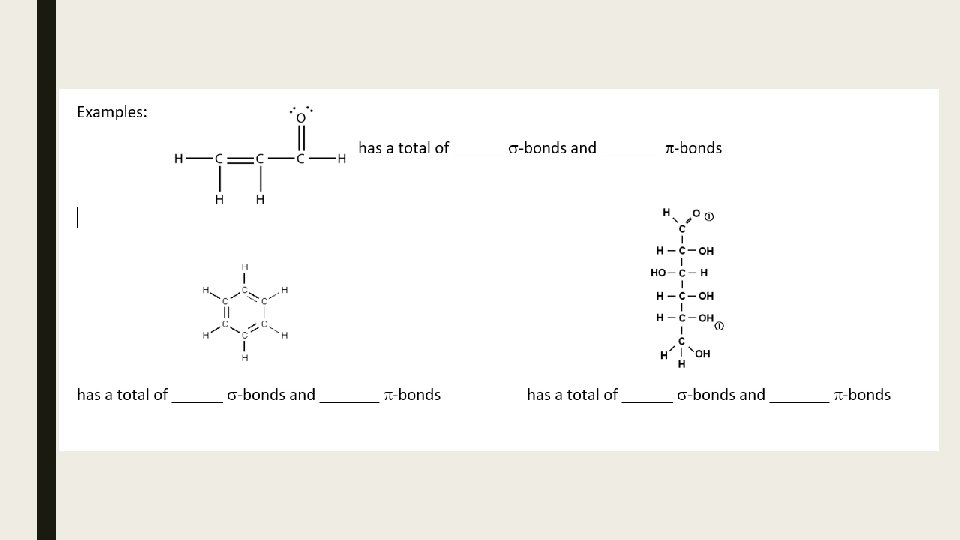
Sigma and Pi bonds Using quantum mechanical model of the atom, a single bond is called a sigma bond (sbond). The second and third bonds of a double and triple bond are classified differently, as pi bonds (p-bond). As such:
AP practice questions

Hybridization and Bonding in Molecules
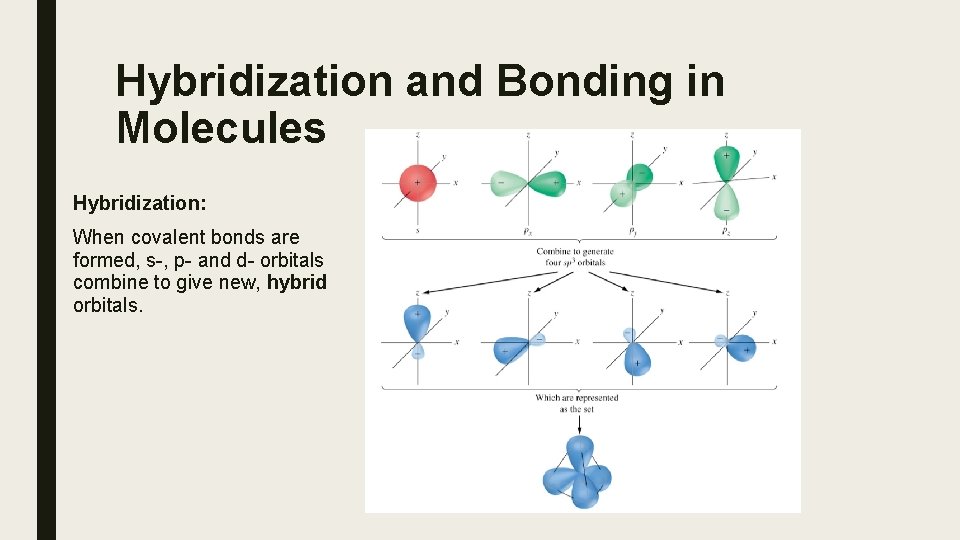
Hybridization and Bonding in Molecules Hybridization: When covalent bonds are formed, s-, p- and d- orbitals combine to give new, hybrid orbitals.
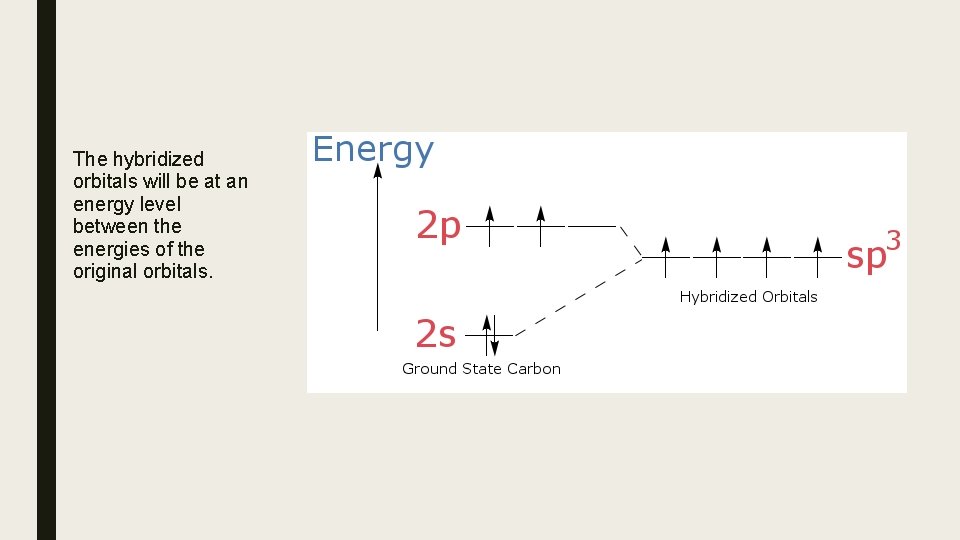
The hybridized orbitals will be at an energy level between the energies of the original orbitals.
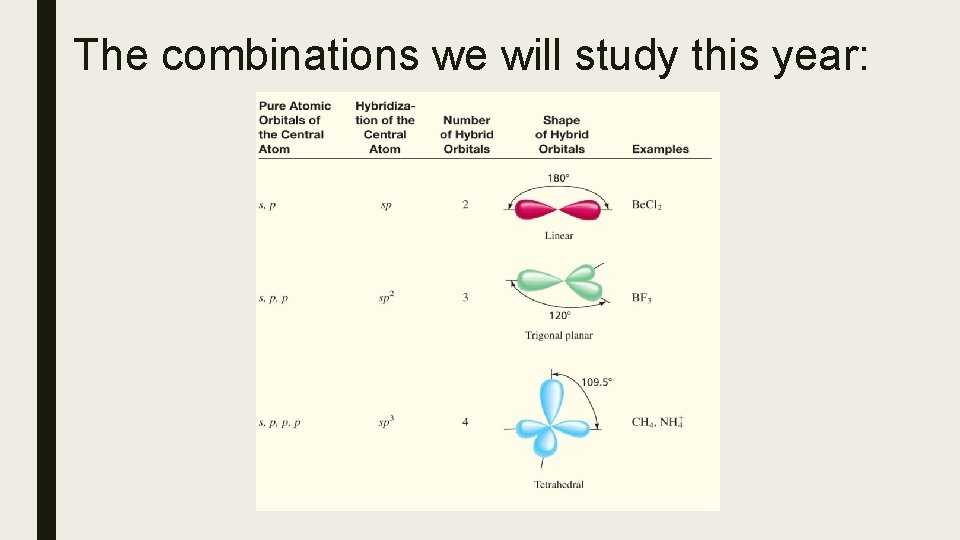
The combinations we will study this year:

To determine the hybrid orbitals formed when bonding occurs: 1. Draw the Lewis Structure. 2. Determine the shape using the VSEPR model. 3. Specify the hybrid orbitals needed to make that shape based on the steric number.

Bonding s-bonds involve the direct overlap of orbitals p-bonds involve the indirect (parallel) overlap of unhybridized p-orbitals.
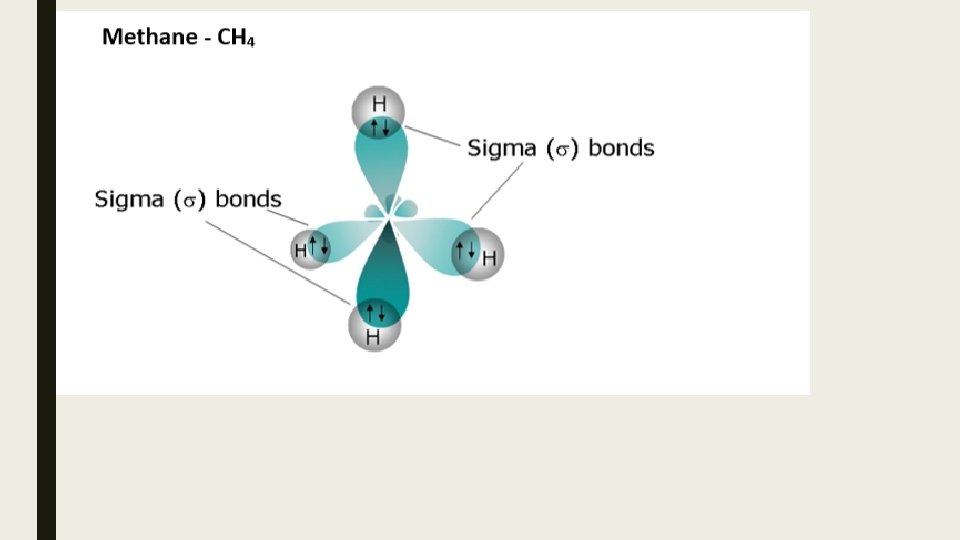
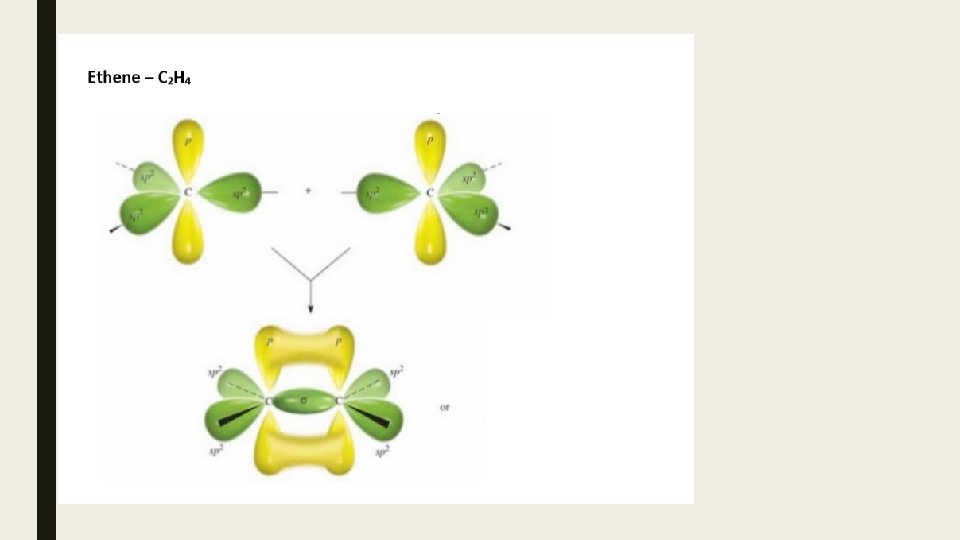
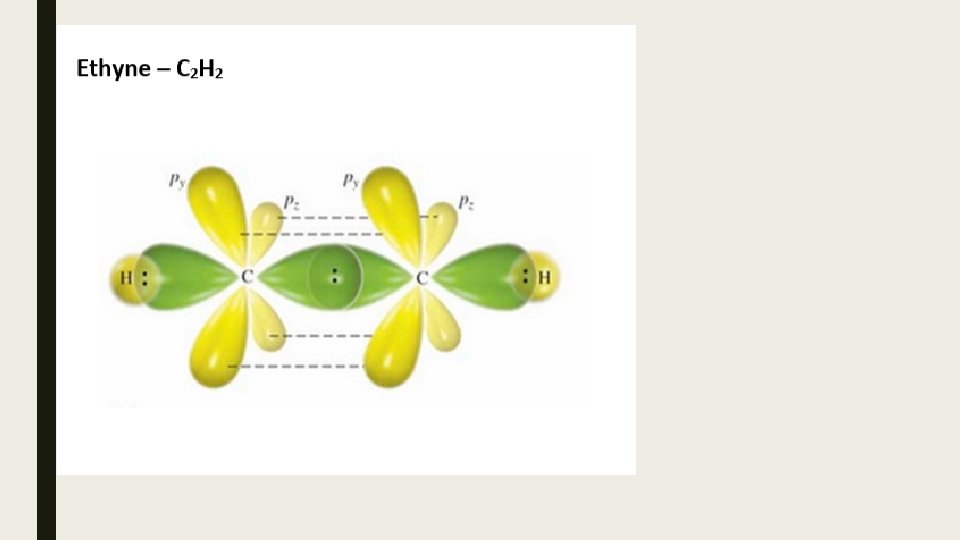

Bond Strengths Since s- bonds involve the direct overlap of orbitals, they are the strongest. p- bonds are not as strong due to the indirect overlap of unhybridized p-orbitals.

AP practice question
- Slides: 32