Bonding Lewis Dot Structures Ionic Covalent Coordinate Covalent

Bonding Lewis Dot Structures Ionic Covalent Coordinate Covalent Nonpolar Covalent Polar Covalent Kenneth E. Schnobrich

Ionic Bonding • • • Generally takes place when you are combining a METAL and a NONMETAL Metals - tend to lose electrons and form (+) ions to achieve an inert gas configuration Nonmetals - tend to gain electrons and form (-) ions to achieve an inert gas configuration Metals are enclosed in [ ] and the positive charge equals the number of electrons lost Nonmetals are enclosed in [ ] and the electrons gained are shown along with the negative charge (# electrons gained) [Na]+1[ Cl ]-1 x Na 2 -8 -1 Cl 2 -8 -7

Ionic Bonding Examples • • Na 2 S Al. Cl 3 KF Ca. I 2 K 3 N Li 2 O Na. H

Covalent Bonding • • • Covalent bonding generally takes place between NONMETAL atoms. Covalent implies that 1, 2, or 3 electron pairs are being shared between two nonmetal atoms. NONMETAL atoms share electrons so they can each complete an inert gas configuration. NONPOLAR COVALENT bonding implies that the electrons are being shared equally between the two nonmetal nuclei. The electronegativity difference is <0. 5 between the two atoms. POLAR COVALENT bonding implies that the electrons are not being shared equally between the two nonmetal nuclei. The electronegativity difference can be 0. 5 but generally <1. 7. **Electronegativity is a measure of the attractive force that an atom has for electrons during bonding

Covalent Examples • • • • H 2 Br 2 N 2 O 2 HCl H 2 O H 3 O+1 HCN CO 2 CH 2 O OF 2 NH 3 NH 4+1 SO 2 SO 3 - Remember each atom wants to achieve an inert gas configuration It is possible to share 1, 2, or 3 pairs of electrons between two nonmetal atoms Look up electronegativities to determine if the bond is nonpolar or polar <0. 5 nonpolar 0. 5 up to <1. 7 generally polar Coordinate covalent bonds - when one atom contributes both electrons of the shared pair Identify lone-pairs and bonded-pairs of electrons Lone-pairs are not bonded to another atom Bonded-pairs are bonded to another atom

Lewis Dot Structures • • • Write the symbol of the element (represents the kernel of the atom) Place the valence electrons around the symbol Placement of the valence electrons must help each of the two bonding atoms complete an inert gas configuration (either 2 electrons or 8 electrons) In ionic bonding the metal will appear to have no valence electrons and the nonmetal will have gained enough to complete the octet of electrons (or 2 electrons in the case of H-1). In covalent bonding (polar, nonpolar, coordinate) the electron pairs must be placed so that each atom either has 2 electrons (like H 2) or 8 electrons (an octet). There cannot be 3 electron bonds nor can there be “dangling” single electrons.

What Molecules Look Like Are They Polar or Nonpolar • When deciding what a molecule will look like you must ask yourself several questions: – (1) Are there any lone-pairs of electrons on the central atom? – (2) Does the structure look symmetrical or asymmetrical – (3) Symmetrical = nonpolar molecule // Asymmetrical = polar molecule • Remember that: – (1) Lone-pairs tend to push other lone-pairs and bondedpairs away from them strongly – (2) Bonded-pairs do not push other bonded-pairs or lonepairs away as strongly as lone-pairs
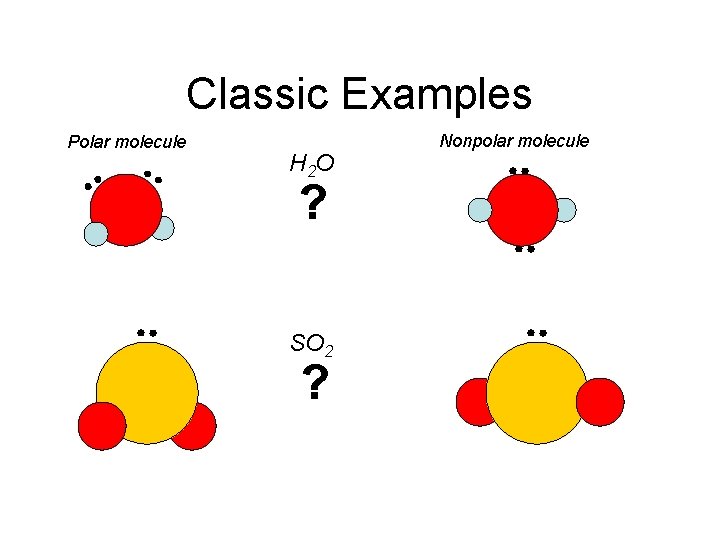
Classic Examples Polar molecule H 2 O ? SO 2 ? Nonpolar molecule
- Slides: 8