Bonding ionic covalent and metallic The names BondIonic

Bonding ionic, covalent, and metallic

The name’s Bond…Ionic Bond • Ionic bonds are formed between ions of opposite charges • Formed due to the TRANSFER of electrons • Formed between a metal cation and a nonmetal anion • Substances held together by ionic bonds are called ionic compounds
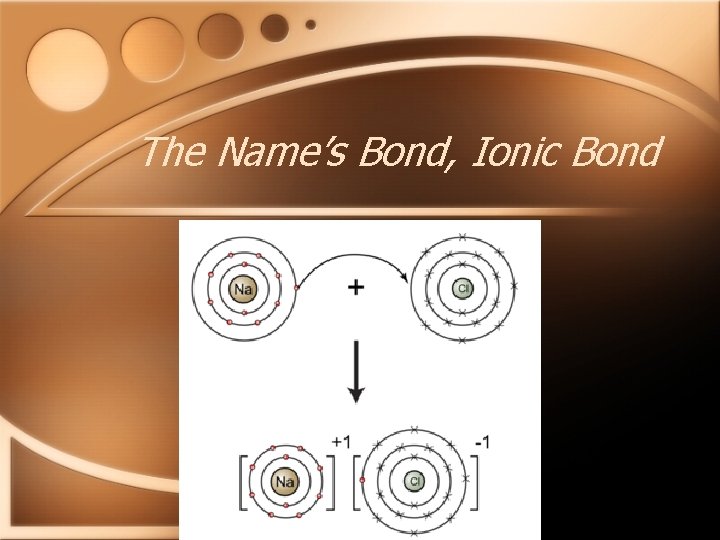
The Name’s Bond, Ionic Bond


The Name’s Bond, Ionic Bond • Properties of Ionic Compounds • • Form crystals Hard and brittle VERY high melting points Good conductors when dissolved in water

The name’s Bond, Metallic Bond • Formed between atoms of metallic elements • Positively charged nuclei held together by a “sea of electrons”

The name’s Bond, Metallic Bond • Properties of Metallic bonds • Very good conductors • Lustrous • Very high melting points

The name’s Bond, Metallic Bond • Since electrons are free to move around in metallic bonds, metals don’t bond with other metals • Instead they form mixtures called Alloys • Alloy examples: steel, brass, bronze, and pewter

The name’s Bond, Covalent Bond • Formed between nonmetallic atoms • Formed due to SHARING of electrons • Substances held together by covalent bonds are called molecules
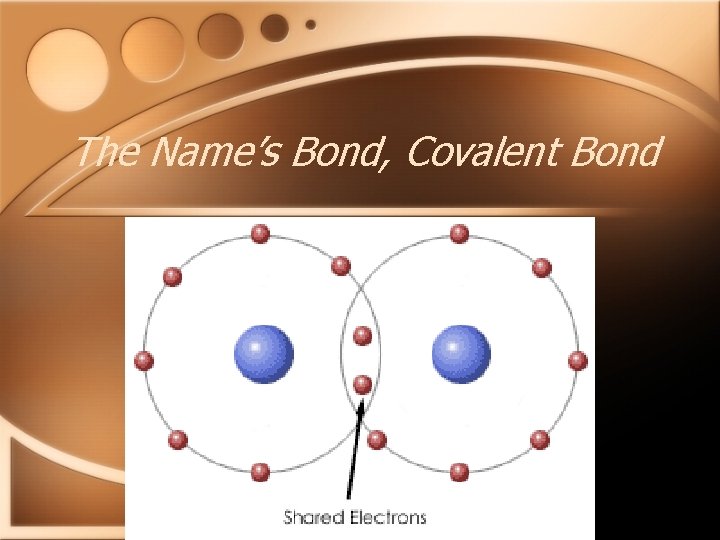
The Name’s Bond, Covalent Bond

The name’s Bond, Covalent Bond • Properties of covalent molecules • Low melting points • Softer and ‘squishier’ then ionic compounds • Do not conduct electricity as solids or while dissolved in water.

The name’s Bond, Covalent Bond • How evenly the electrons are shared, depends on the electronegativity of the two elements involved • Electronegativity: the ability of an atom to attract electrons to itself when bonded to another atom
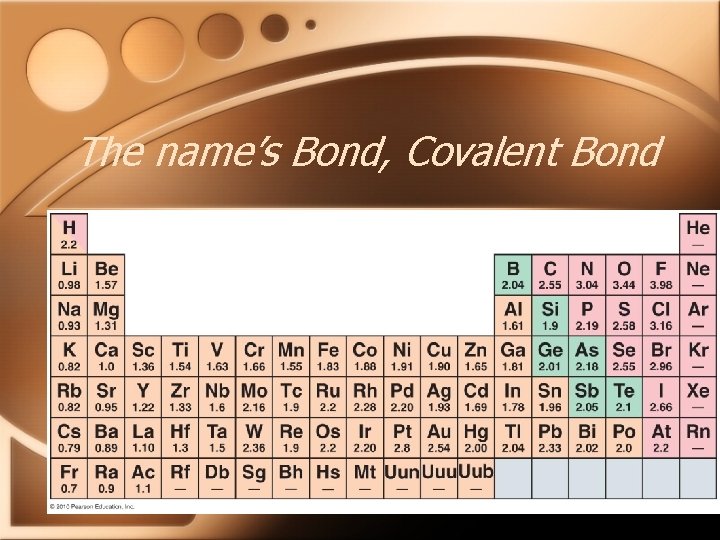
The name’s Bond, Covalent Bond

The name’s Bond, Covalent Bond • When the electronegativity difference between the bonded atoms is small, the electrons are shared evenly the bond is said to be non-polar

The name’s Bond, Covalent Bond • When the electronegativity difference between the bonded atoms is great, the electrons are unevenly shared and the bond is said to be polar.

The name’s Bond, Covalent Bond • In polar covalent bonds, one atom becomes partially positively charged and one becomes partially negatively charged • This is shown using + and -
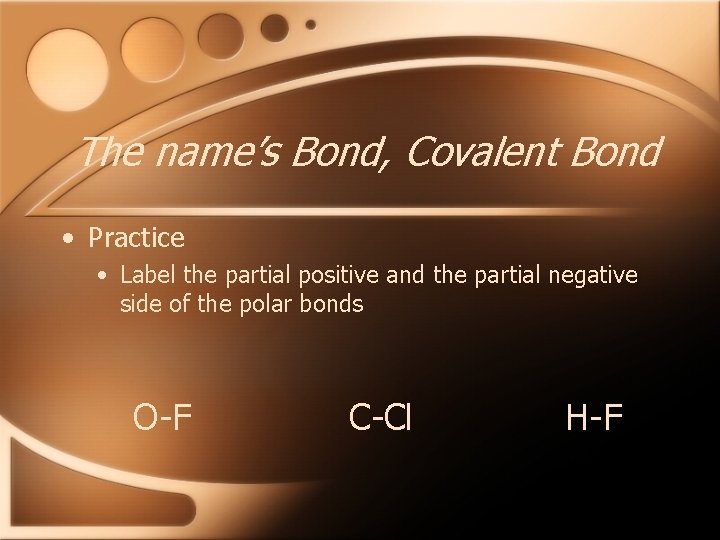
The name’s Bond, Covalent Bond • Practice • Label the partial positive and the partial negative side of the polar bonds O-F C-Cl H-F
- Slides: 17