Bonding Chemical Bond Attractive force between atoms or

Bonding • Chemical Bond – Attractive force between atoms or ions that binds them together as a unit – Bonds form in order to… • • decrease potential energy (PE) increase stability bond forming is always exothermic bonds can be metallic, ionic, or covalent

Bonding (valence electrons) • The electrons responsible for the chemical properties and bonding of atoms are those in the outer energy level. • Valence electrons - The s and p electrons in the outer energy level. • Core electrons -those in the energy levels below.
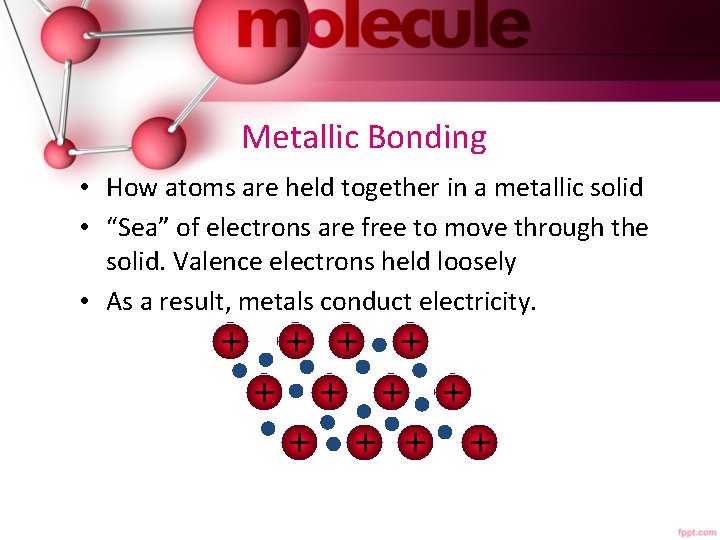
Metallic Bonding • How atoms are held together in a metallic solid • “Sea” of electrons are free to move through the solid. Valence electrons held loosely • As a result, metals conduct electricity. + + +
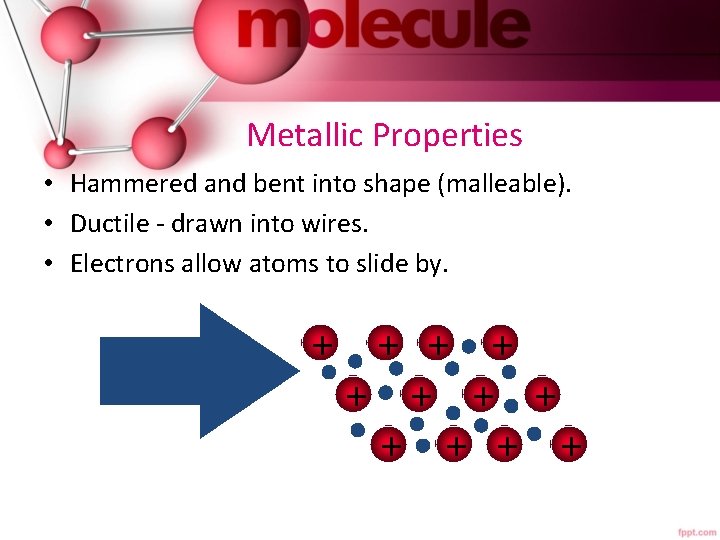
Metallic Properties • Hammered and bent into shape (malleable). • Ductile - drawn into wires. • Electrons allow atoms to slide by. + + +

Ionic Bonding - Crystal Lattice Table salt
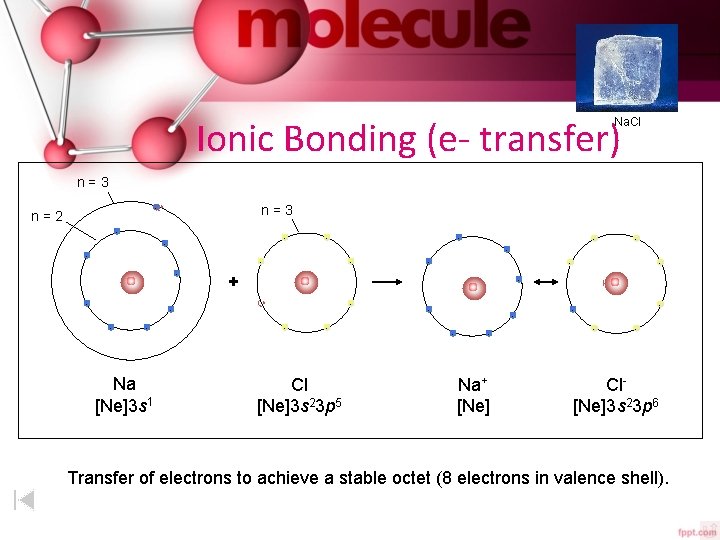
Ionic Bonding (e- transfer) Na. Cl n=3 - n=2 - - - - Na [Ne]3 s 1 - - - + - - - - - Cl [Ne]3 s 23 p 5 - - - Na+ [Ne] - - - Cl[Ne]3 s 23 p 6 Transfer of electrons to achieve a stable octet (8 electrons in valence shell).
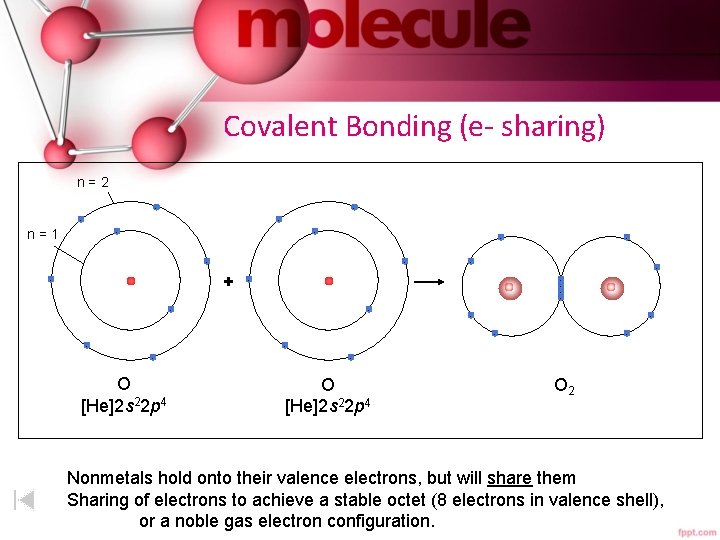
Covalent Bonding (e- sharing) n=2 - - n=1 - - - + - - - - O [He]2 s 22 p 4 O 2 Nonmetals hold onto their valence electrons, but will share them Sharing of electrons to achieve a stable octet (8 electrons in valence shell), or a noble gas electron configuration.
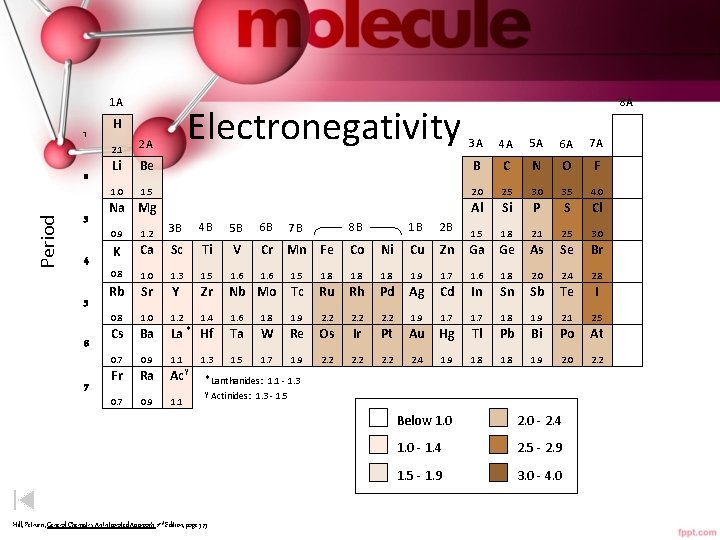
1 A 1 Period 2 3 4 5 6 7 Electronegativity H 8 A 3 A 4 A 5 A 6 A 7 A Be B C N O F 1. 5 2. 0 2. 5 3. 0 3. 5 4. 0 Al Si P S Cl 1. 5 1. 8 2. 1 2. 5 3. 0 2. 1 2 A Li 1. 0 Na Mg 1 B 2 B Ni Cu Zn Ga Ge As Se Br 1. 8 1. 9 1. 7 1. 6 1. 8 2. 0 2. 4 2. 8 Ru Rh Pd Ag Cd In Sn Sb Te I 1. 9 2. 2 1. 9 1. 7 1. 8 1. 9 2. 1 2. 5 W Re Os Ir Pt Au Hg Tl Pb Bi Po At 1. 7 1. 9 2. 2 2. 4 1. 9 1. 8 1. 9 2. 0 2. 2 0. 9 1. 2 3 B 4 B 5 B 6 B 7 B 8 B K Ca Sc Ti V Cr Mn Fe Co 0. 8 1. 0 1. 3 1. 5 1. 6 1. 5 1. 8 Rb Sr Y Zr Nb Mo Tc 0. 8 1. 0 1. 2 1. 4 1. 6 1. 8 Cs Ba La * Hf Ta 0. 7 0. 9 1. 1 1. 3 1. 5 Fr Ra Ac 0. 7 0. 9 1. 1 y * Lanthanides: 1. 1 - 1. 3 y Actinides: Hill, Petrucci, General Chemistry An Integrated Approach 2 nd Edition, page 373 1. 3 - 1. 5 Below 1. 0 2. 0 - 2. 4 1. 0 - 1. 4 2. 5 - 2. 9 1. 5 - 1. 9 3. 0 - 4. 0

Electronegativity • A measure of how strongly the atoms attract electrons in a bond. • The larger the electronegativity difference the more polar the covalent bond. 0. 0 0. 1 - 2. 0 > 2. 0 Covalent nonpolar Covalent polar Ionic
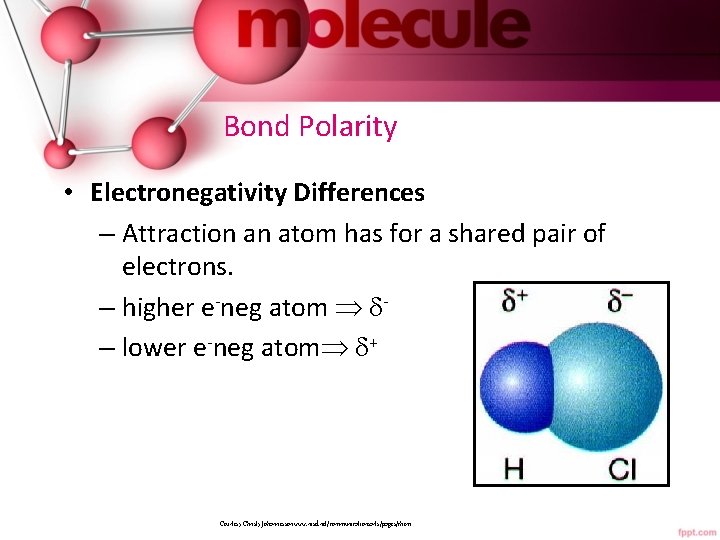
Bond Polarity • Electronegativity Differences – Attraction an atom has for a shared pair of electrons. – higher e-neg atom – lower e-neg atom + Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
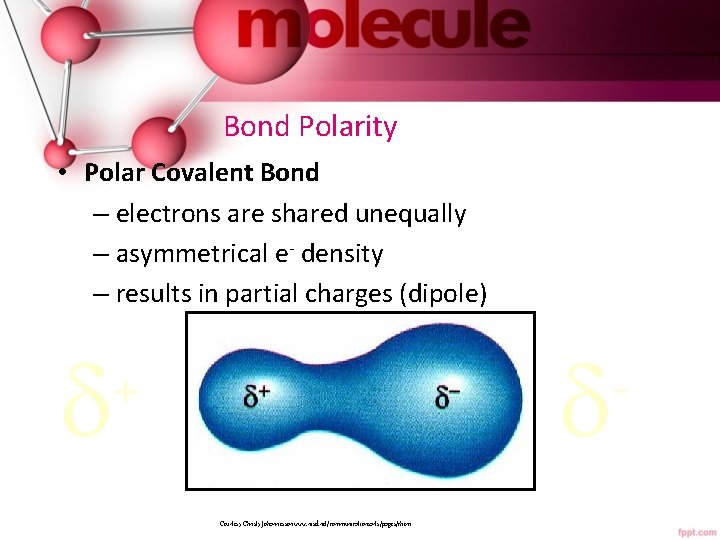
Bond Polarity • Polar Covalent Bond – electrons are shared unequally – asymmetrical e- density – results in partial charges (dipole) + Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
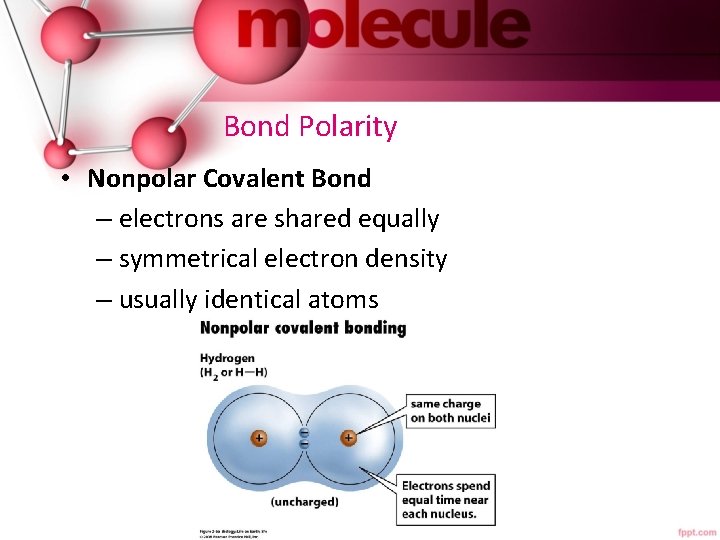
Bond Polarity • Nonpolar Covalent Bond – electrons are shared equally – symmetrical electron density – usually identical atoms
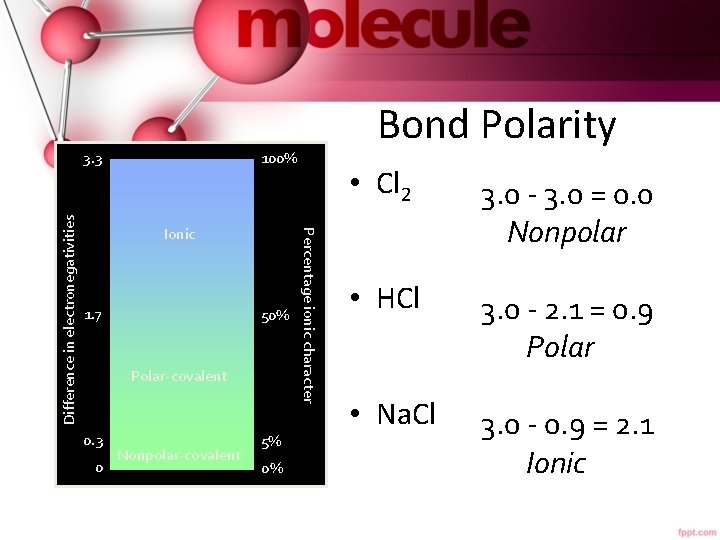
100% Ionic 1. 7 50% Polar-covalent 0. 3 0 Nonpolar-covalent 5% 0% Percentage ionic character Difference in electronegativities 3. 3 Bond Polarity • Cl 2 3. 0 - 3. 0 = 0. 0 Nonpolar • HCl 3. 0 - 2. 1 = 0. 9 Polar • Na. Cl 3. 0 - 0. 9 = 2. 1 Ionic
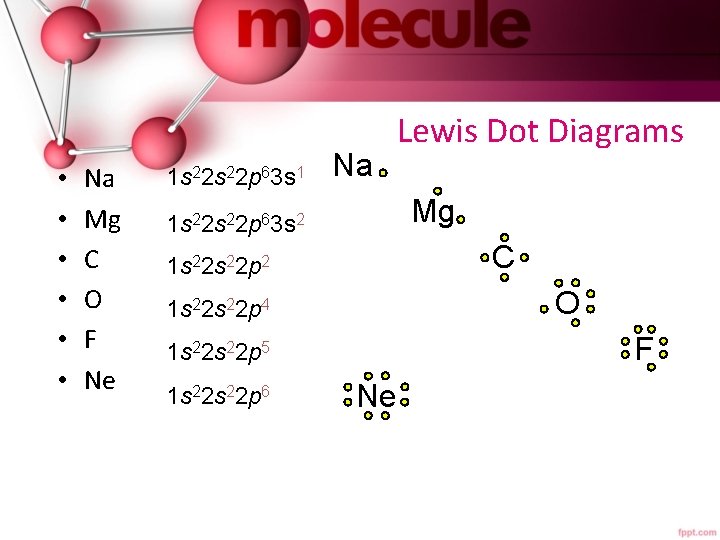
• • • Na Mg C O F Ne 1 s 22 p 63 s 1 Na Lewis Dot Diagrams Mg 1 s 22 p 63 s 2 C 1 s 22 p 2 O 1 s 22 p 4 F 1 s 22 p 5 1 s 22 p 6 Ne

Covalent Bonds • Single bond - two atoms share one pair (2) of valence electrons. • Different from an ionic bond because they actually form molecules, not crystal lattices. • Double bond - two atoms share two pair (4) of valence electrons. • Triple bond - two atoms share three pair (6) of valence electrons.

Covalent Bonding “Rules” • Atoms of four elements ALWAYS have full octet (8) valence e(Carbon, Nitrogen, Oxygen, and Fluorine). • Atoms of four elements will have fewer than full octet (8) valence e- (Hydrogen, Boron, Beryllium, and Aluminum). • Atoms of other elements will have AT LEAST an octet (8) valence e-. NOTE: Helium generally does not bond, but could only hold two valence e-. • Central atom is nearly always the one with lowest electronegativity. Hydrogen is never a central atom. • If more than one possible stable structure exists, multiple bonds will appear with larger EN atoms. • Molecules strive for simplicity (i. e. single and C-H bonds).

Drawing Lewis Structures 1. Determine central atom (least electronegative) and draw all remaining atoms around it. Hydrogen is always on far outside. 2. Determine the total number of valence electrons to be accounted for (represented by dots). 3. Connect all atoms with two dots (one bond). 4. Surround all outer atoms with electrons (to achieve stability). Unbonded electrons must be in pairs. 5. If electron are left over, place on central atom (reside in d orbitals). 6. If there is shortage of electrons to achieve stability, move unbonded electrons to form double or triple bond(s).
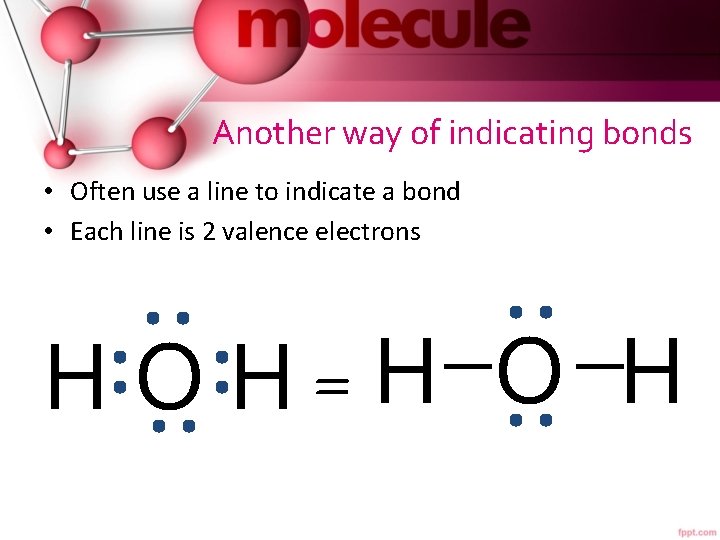
Another way of indicating bonds • Often use a line to indicate a bond • Each line is 2 valence electrons H O H=H O H
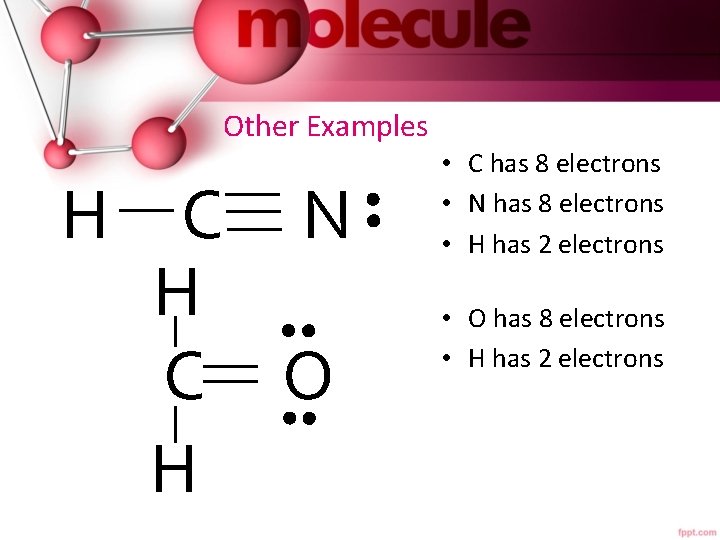
Other Examples H C N H C O H • C has 8 electrons • N has 8 electrons • H has 2 electrons • O has 8 electrons • H has 2 electrons

Polyatomic Ions • To find total number of valence electrons: – Add 1 electron for each negative charge. – Subtract 1 electron for each positive charge. • Place brackets around the ion and label the charge.
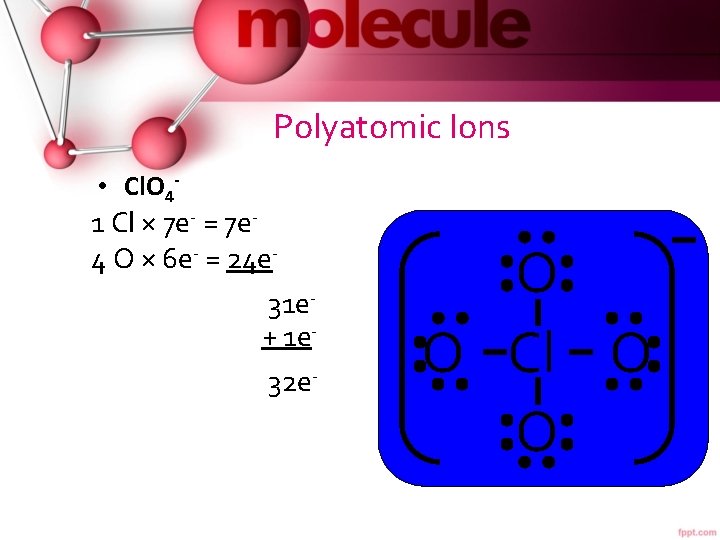
Polyatomic Ions • Cl. O 4 - 1 Cl × 7 e- = 7 e 4 O × 6 e- = 24 e 31 e- + 1 e 32 e- 8 e 24 e- O O Cl O O
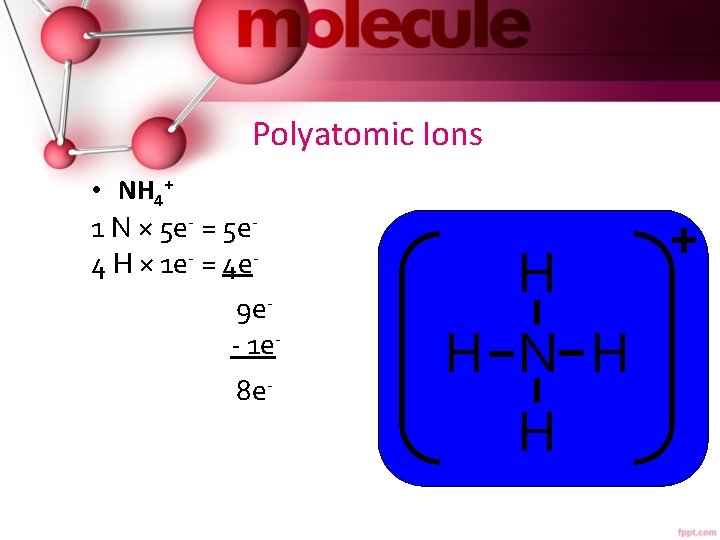
Polyatomic Ions • NH 4+ 1 N × 5 e- = 5 e 4 H × 1 e- = 4 e 9 e- 1 e 8 e- 8 e 0 e- H H N H H
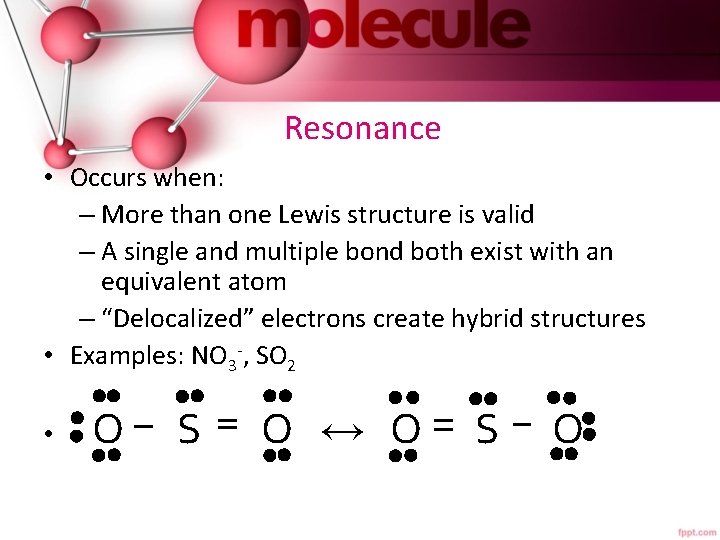
Resonance • Occurs when: – More than one Lewis structure is valid – A single and multiple bond both exist with an equivalent atom – “Delocalized” electrons create hybrid structures • Examples: NO 3 -, SO 2 • O S O ↔ O S O

VSEPR • Valence Shell Electron Pair Repulsion. Predicts three dimensional geometry of molecules. Name tells you theory. Valence shell - outside electrons. Electron Pair repulsion - electron pairs try to get as far away as possible. • Can determine the angles of bonds. • •
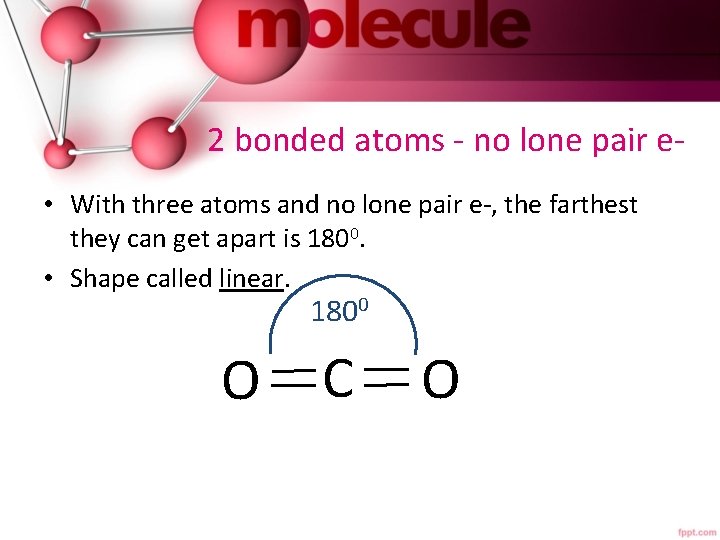
2 bonded atoms - no lone pair e • With three atoms and no lone pair e-, the farthest they can get apart is 1800. • Shape called linear. 1800 O C O
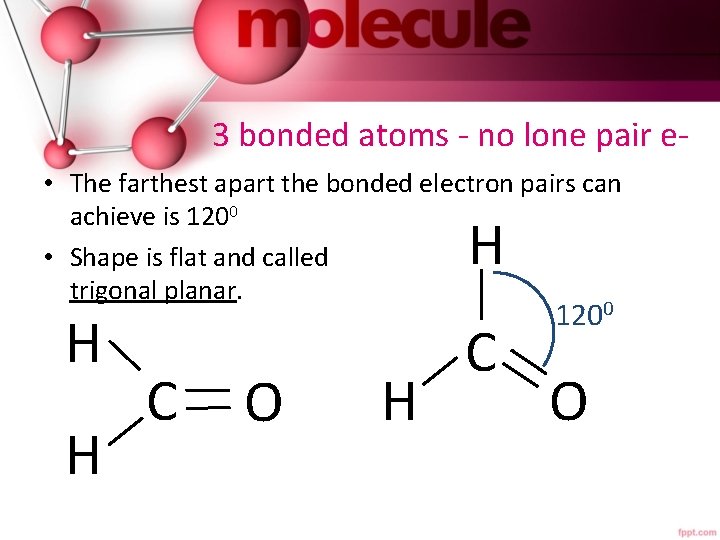
3 bonded atoms - no lone pair e • The farthest apart the bonded electron pairs can achieve is 1200 • Shape is flat and called trigonal planar. H H H C O H C 1200 O

H 4 atoms bonded H C H H • Single bonds fill atoms. • There are 4 pairs of electrons pushing away. • The furthest they can get away is 109. 50.
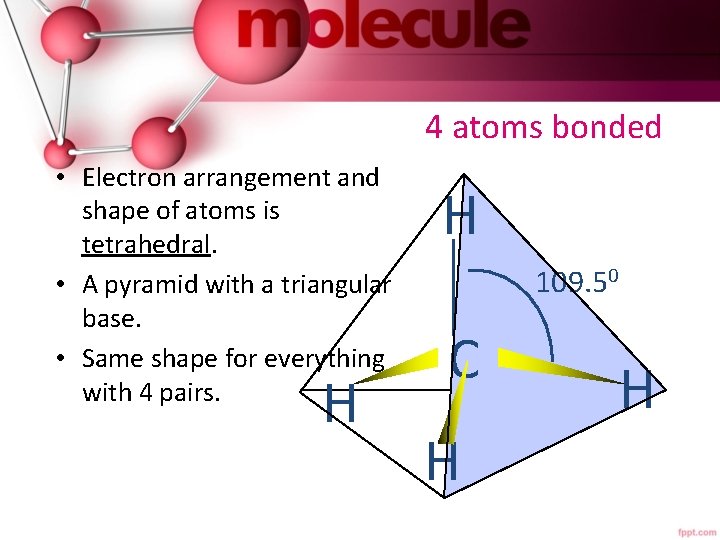
4 atoms bonded • Electron arrangement and shape of atoms is tetrahedral. • A pyramid with a triangular base. • Same shape for everything with 4 pairs. H H 109. 50 C H H
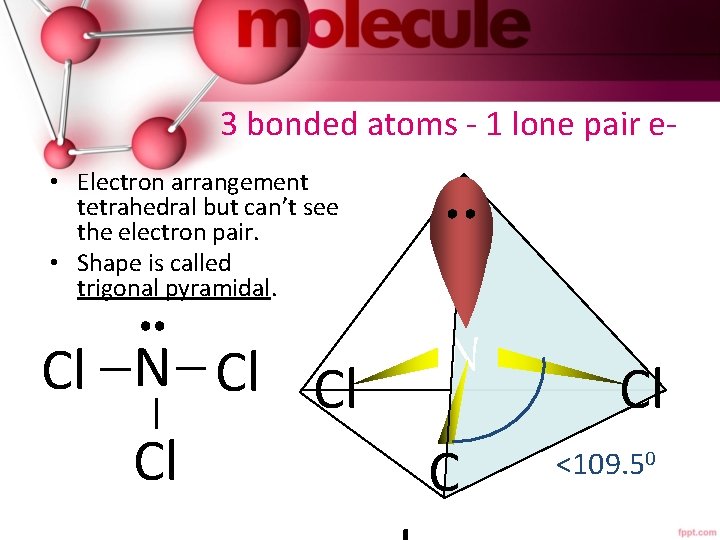
3 bonded atoms - 1 lone pair e • Electron arrangement tetrahedral but can’t see the electron pair. • Shape is called trigonal pyramidal. Cl N Cl Cl Cl CC N Cl <109. 50
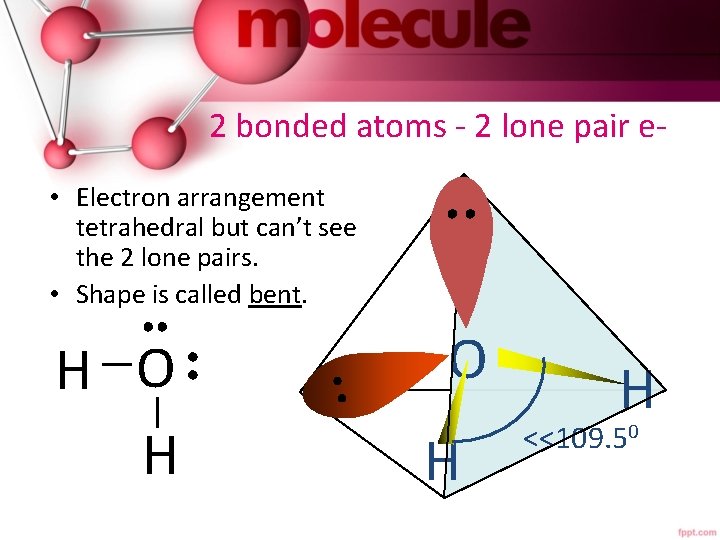
2 bonded atoms - 2 lone pair e • Electron arrangement tetrahedral but can’t see the 2 lone pairs. • Shape is called bent. H O H H <<109. 50
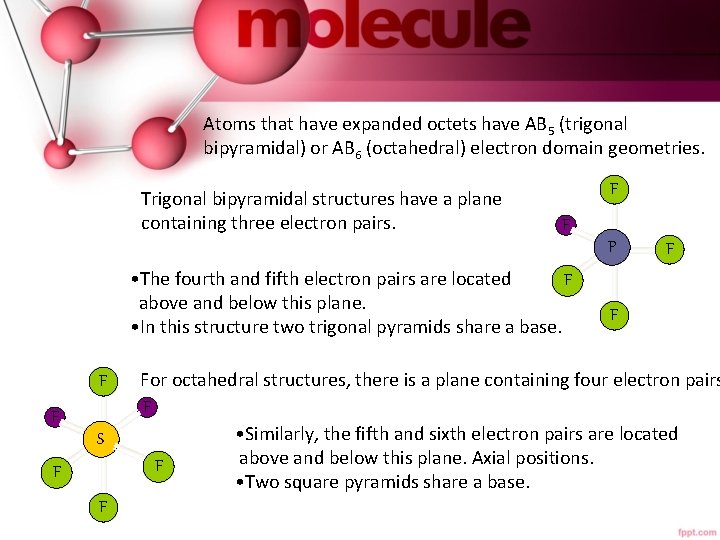
Atoms that have expanded octets have AB 5 (trigonal bipyramidal) or AB 6 (octahedral) electron domain geometries. Trigonal bipyramidal structures have a plane containing three electron pairs. F F P • The fourth and fifth electron pairs are located F above and below this plane. • In this structure two trigonal pyramids share a base. F F S F F For octahedral structures, there is a plane containing four electron pairs F • Similarly, the fifth and sixth electron pairs are located above and below this plane. Axial positions. F • Two square pyramids share a base.
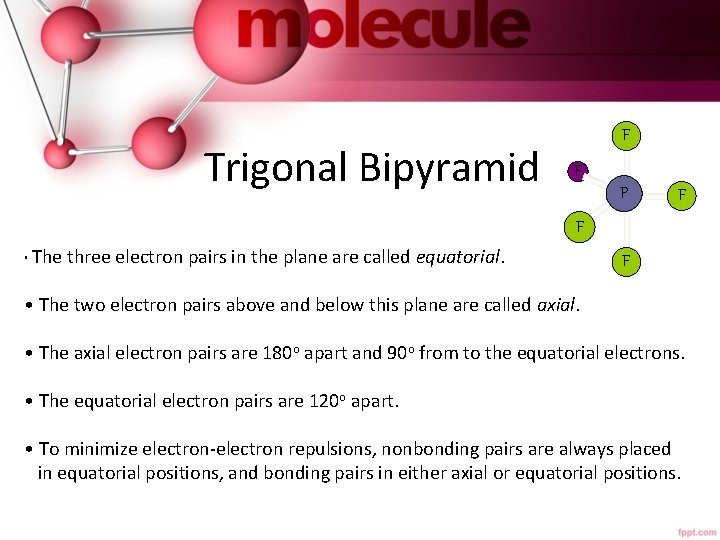
Trigonal Bipyramid F F P F F • The three electron pairs in the plane are called equatorial. F • The two electron pairs above and below this plane are called axial. • The axial electron pairs are 180 o apart and 90 o from to the equatorial electrons. • The equatorial electron pairs are 120 o apart. • To minimize electron-electron repulsions, nonbonding pairs are always placed in equatorial positions, and bonding pairs in either axial or equatorial positions.
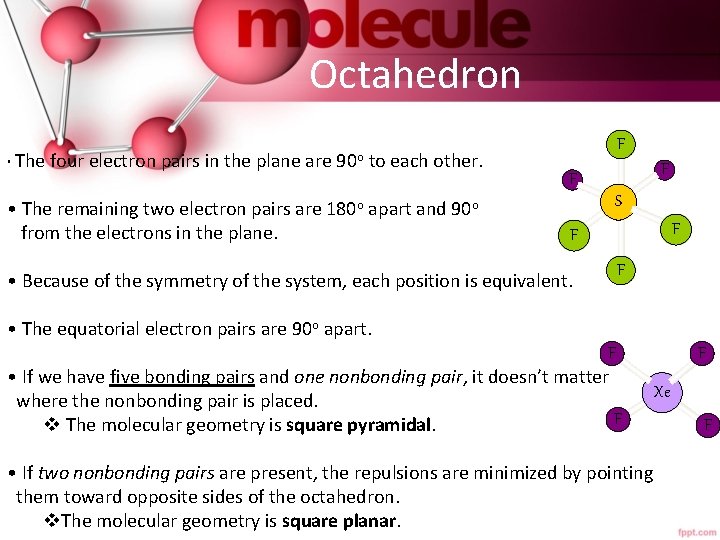
Octahedron • The four electron pairs in the plane are 90 o to each other. • The remaining two electron pairs are 180 o apart and 90 o from the electrons in the plane. F F F S F F • Because of the symmetry of the system, each position is equivalent. F • The equatorial electron pairs are 90 o apart. F • If we have five bonding pairs and one nonbonding pair, it doesn’t matter where the nonbonding pair is placed. F v The molecular geometry is square pyramidal. • If two nonbonding pairs are present, the repulsions are minimized by pointing them toward opposite sides of the octahedron. v. The molecular geometry is square planar. F Xe F
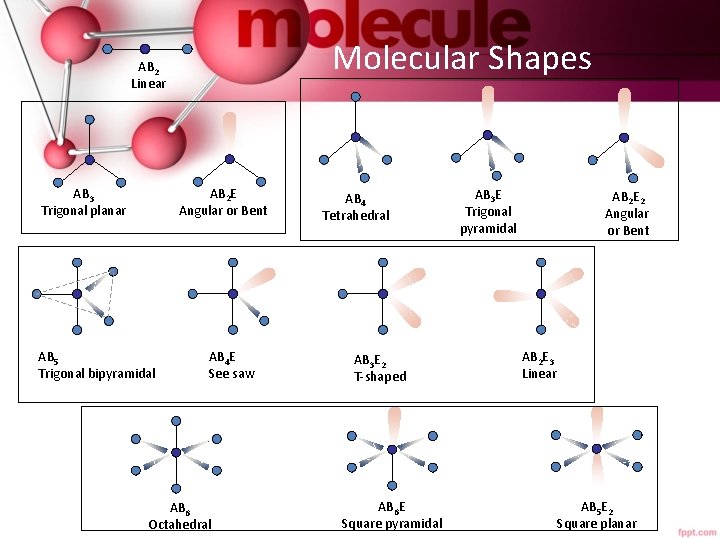
Molecular Shapes AB 2 Linear AB 2 E Angular or Bent AB 3 Trigonal planar AB 5 Trigonal bipyramidal AB 4 E See saw AB 6 Octahedral AB 4 Tetrahedral AB 3 E 2 T-shaped AB 6 E Square pyramidal AB 3 E Trigonal pyramidal AB 2 E 2 Angular or Bent AB 2 E 3 Linear AB 5 E 2 Square planar

Polarity of Molecules • In order for a molecule to be polar it must: 1. Have polar bonds. • Electronegativity difference—theory. • Bond dipole moments—measured. 2. Have an unsymmetrical shape. • Vector addition. • Polarity effects the intermolecular forces of attraction. Tro's Introductory Chemistry, Chapter 10 35
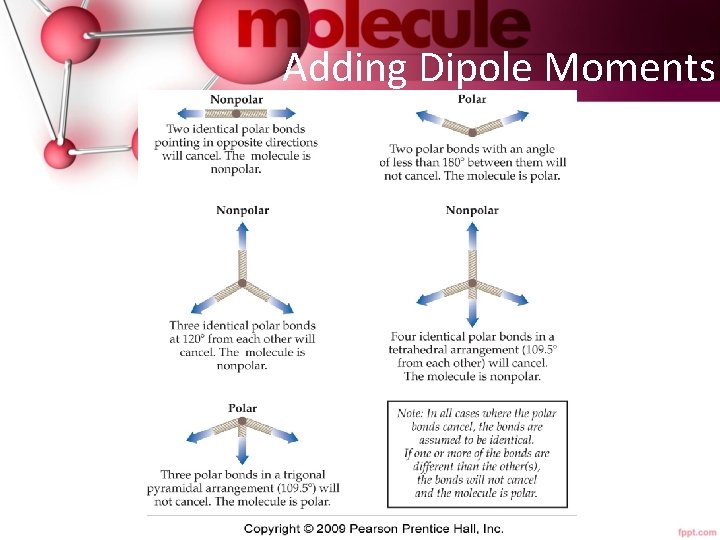
Adding Dipole Moments 36
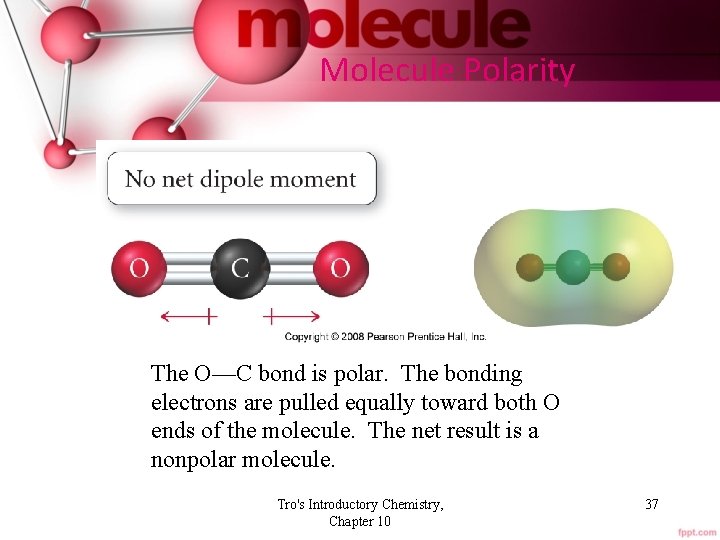
Molecule Polarity The O—C bond is polar. The bonding electrons are pulled equally toward both O ends of the molecule. The net result is a nonpolar molecule. Tro's Introductory Chemistry, Chapter 10 37

Molecule Polarity The H—O bond is polar. Both sets of bonding electrons are pulled toward the O end of the molecule. The net result is a polar molecule. Tro's Introductory Chemistry, Chapter 10 38
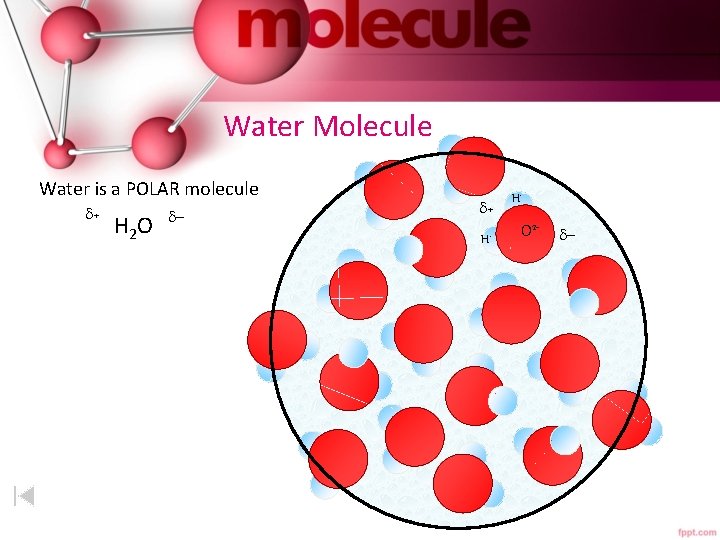
Water Molecule Water is a POLAR molecule + H 2 O - + H+ H+ O 2 - -
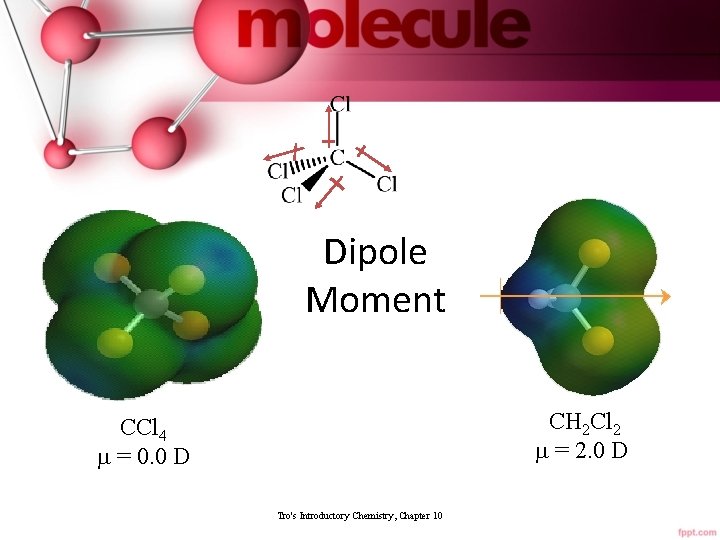
Dipole Moment CH 2 Cl 2 m = 2. 0 D CCl 4 m = 0. 0 D Tro's Introductory Chemistry, Chapter 10 40
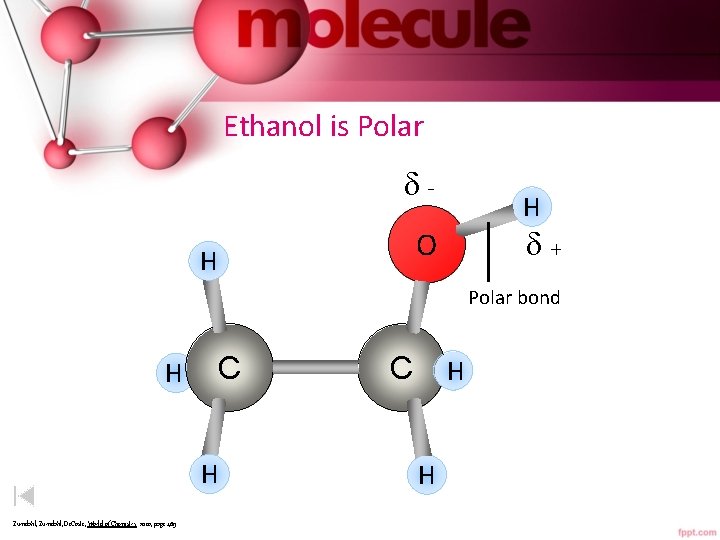
Ethanol is Polar - H + O H Polar bond H C H Zumdahl, De. Coste, World of Chemistry 2002, page 469 C H H
- Slides: 41