Bonding Between Atoms Why do Atoms Form Bonds

Bonding Between Atoms

Why do Atoms Form Bonds? l To get a stable octet of valence electrons. l Called a “noble gas configuration”

Changes in Energy l Energy is released when bonds form. (exothermic) l Results in lower energy, more stability l Know example…

Bonding Type #1 Ionic Bonding

Atoms Become Ions l Lose electrons, become a (+) ion (cation) l Ex: Mg is 2 -8 -2 l Loses 2 electrons to become Mg+2 2 -8

l Gain electrons, become a (–) ion (anion) l Ex: Cl is 2 -8 -7 l Gains 1 electron to become Cl-1 2 -8 -8
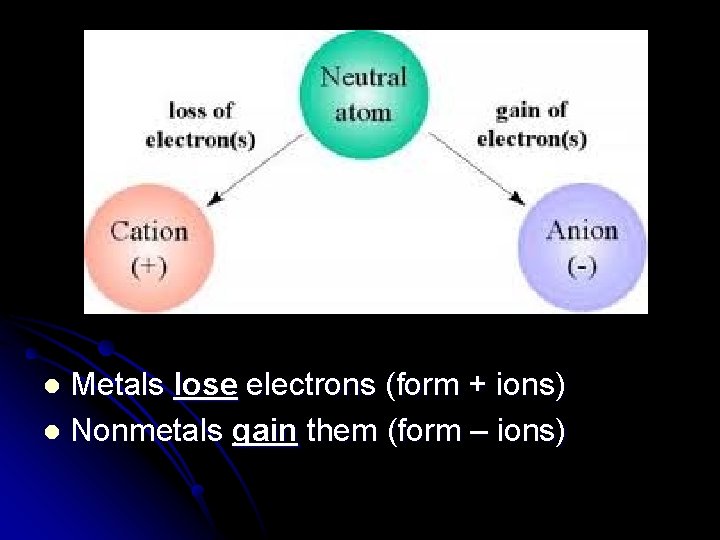
Metals lose electrons (form + ions) l Nonmetals gain them (form – ions) l
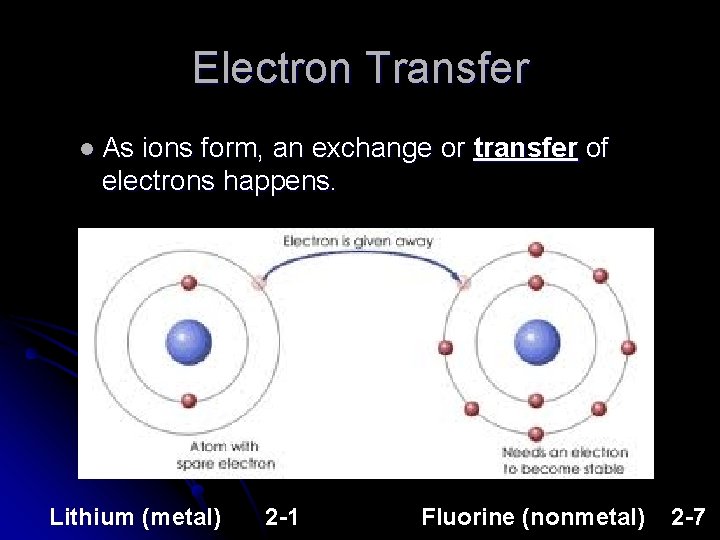
Electron Transfer l As ions form, an exchange or transfer of electrons happens. Lithium (metal) 2 -1 Fluorine (nonmetal) 2 -7

Electronegativity l Atoms in ionic compounds have a large difference in their EN values > 1. 7

l Ex: EN Values l Metal l Nonmetal l Na = 0. 9 Cl = 3. 2 Nonmetal with higher EN “takes” electron(s) from the metal

l A positively charged ion and a negatively charged ion attract each other. - + ion l This attraction forms an IONIC BOND

l Ionic compounds have a uniform crystalline lattice structure.

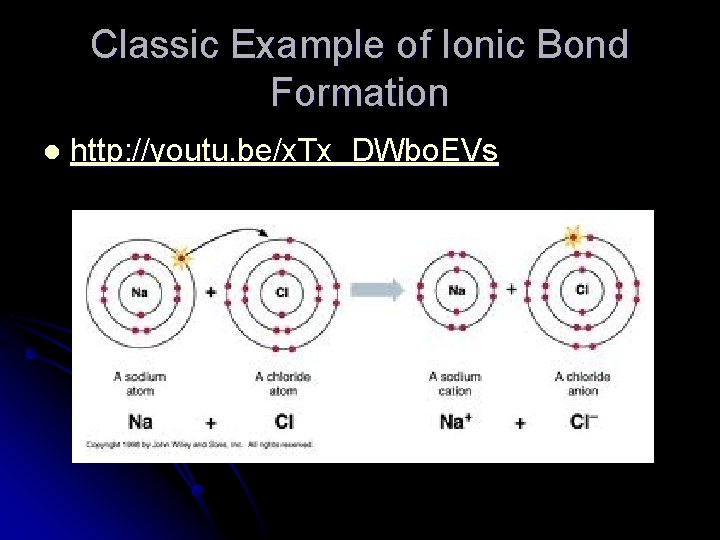
Classic Example of Ionic Bond Formation l http: //youtu. be/x. Tx_DWbo. EVs

Properties of Ionic Compounds
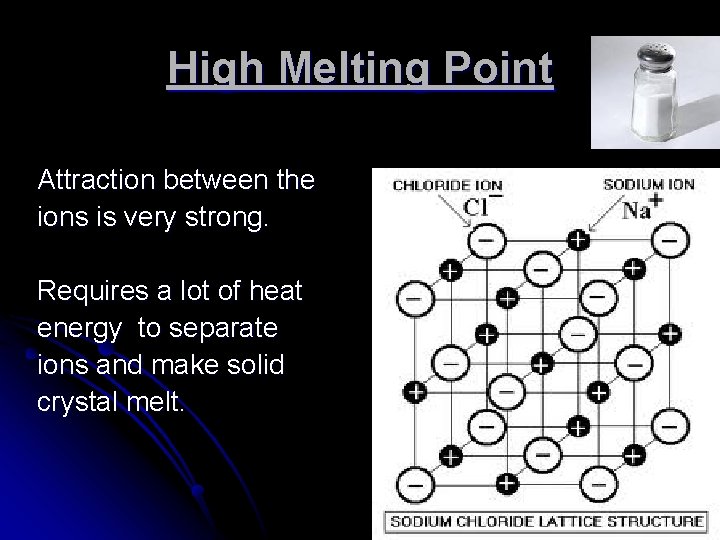
High Melting Point Attraction between the ions is very strong. Requires a lot of heat energy to separate ions and make solid crystal melt.

Solubility in Water l Most ionics will dissolve in water, or be “soluble”. l When dissolved in water they are “aqueous” Ex: Na. Cl (aq)
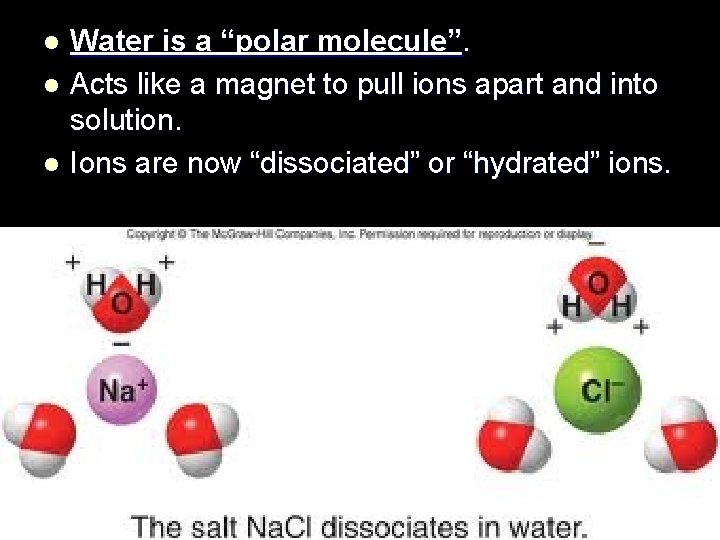
Water is a “polar molecule”. l Acts like a magnet to pull ions apart and into solution. l Ions are now “dissociated” or “hydrated” ions. l


http: //youtu. be/g. N 9 euz 9 jzwc l http: //youtu. be/EBf. Gc. TAJF 4 o l

Conductivity l Ionic compounds conduct when ions are “mobile” or free to move about.

Conductivity l Ionic compounds conduct when: l Molten (melted/liquid) (l) l Aqueous (aq) Note: They DO NOT conduct when solid as the ions are locked in place.

Types of Ionic Compounds l Binary: Contain 2 elements Ex: Mg. Cl 2, Al 2 O 3, Na. Cl

l Ternary: Contain 3 elements l Polyatomic ion present. l Contain both ionic and covalent bonds!! Ex: Na. NO 3, Ca 3(PO 4)2, NH 4 Cl Elements inside the polyatomic ion are covalently bonded (all nonmetals).

Ionic Compounds are Neutral l Criss-Cross charges if necessary to balance the formula

Roman Numeral (Stock System) l Roman numeral is used in the name of ionic compounds in which the metal can have more than one possible charge. Ex: Ni. Br 2 Ni. Br 3 Nickel II Bromide Nickel III Bromide

Bonding Type #2
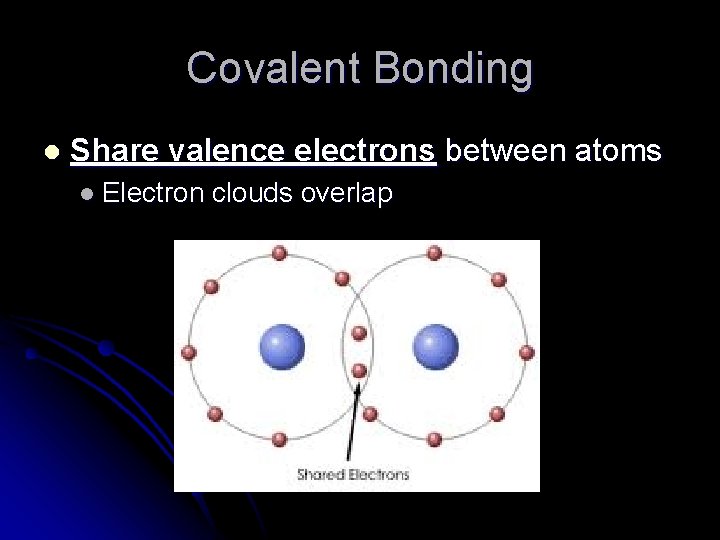
Covalent Bonding l Share valence electrons between atoms l Electron clouds overlap

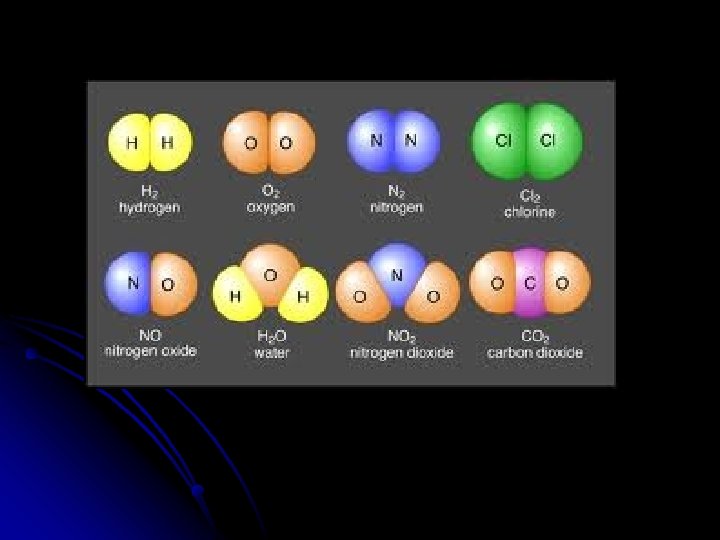
l Happens between nonmetals Ex: H 2 O CH 4 C 6 H 12 O 6 NH 3 CO 2

Electronegativity l Difference in EN is smaller than in ionics and is usually < 1. 7 l Ex: HCl H = 2. 1 Cl = 3. 2 Difference = 1. 1

Molecular Formulas l All covalent compounds are called molecules. l Molecular formulas: show actual number of atoms of each element present in compound Ex: H 2 O 2 hydrogen atoms and 1 oxygen

Empirical Formulas l Empirical formulas: l Show simplest whole number ratio of atoms or ions in the compound. l All l Ex: ionic compounds have empirical formulas Mg. Cl 2 1 : 2 ion ratio

You can simplify some molecular formulas to make them empirical ratios Ex: C 6 H 12 O 6 Simplest ratio of atoms CH 2 O

Structural Formulas Show the atoms are bonded together in a molecule. l Use “lines” to show covalent bonds l
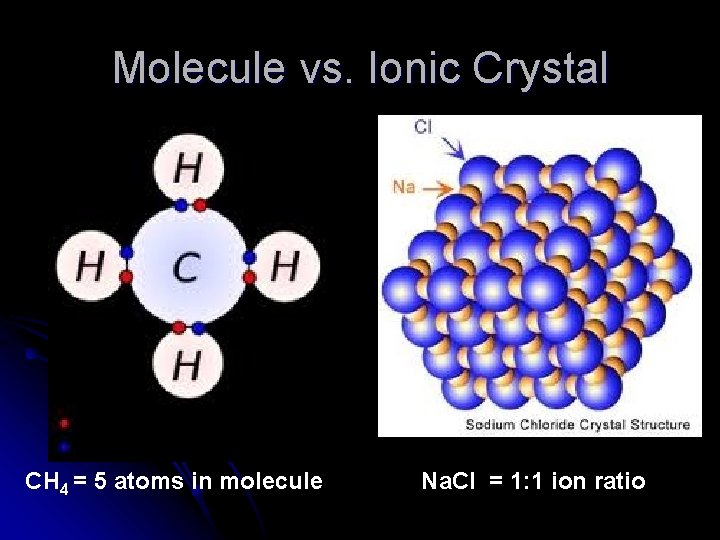
Molecule vs. Ionic Crystal CH 4 = 5 atoms in molecule Na. Cl = 1: 1 ion ratio

Ionic Character l Note: The greater the EN difference is between atoms the more “Ionic Character” the bond has.

Single, Double, Triple Bonds l Atoms can share single double or triple bonds between them. l Each bond represents a shared pair of electrons. l http: //youtu. be/1 wp. Dic. W_MQQ
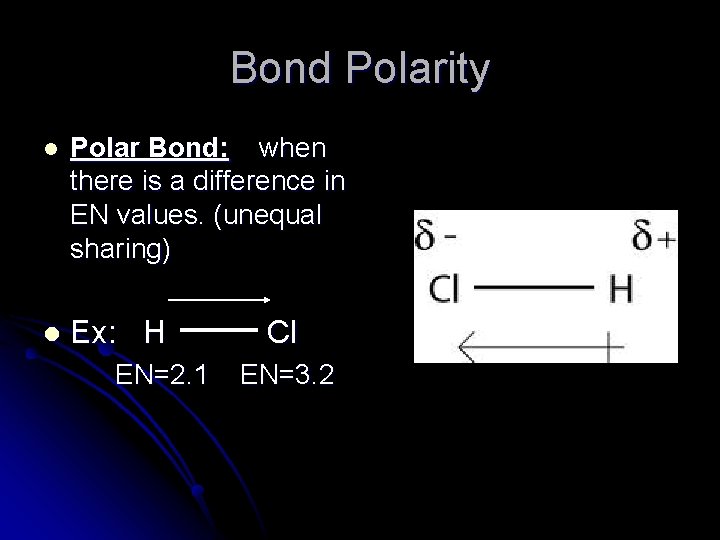
Bond Polarity l Polar Bond: when there is a difference in EN values. (unequal sharing) l Ex: H EN=2. 1 Cl EN=3. 2

l Non. Polar Bond: no difference in EN values. (equal sharing) Ex: O 2, N 2, Cl 2, H 2 (all the diatomics!)


Shapes of Molecules

VSPER l Valence Shell Electron Pair Repulsion l Valence electrons will orient themselves around the “central” atom to be as far apart from each other as possible. l This influences the “shape” of the molecule.

Polarity of a Molecule l Polar Molecules: l Have l polar bonds and are not symmetrical Nonpolar Molecules l Have nonpolar bonds OR l Have polar bonds and are symmetrical
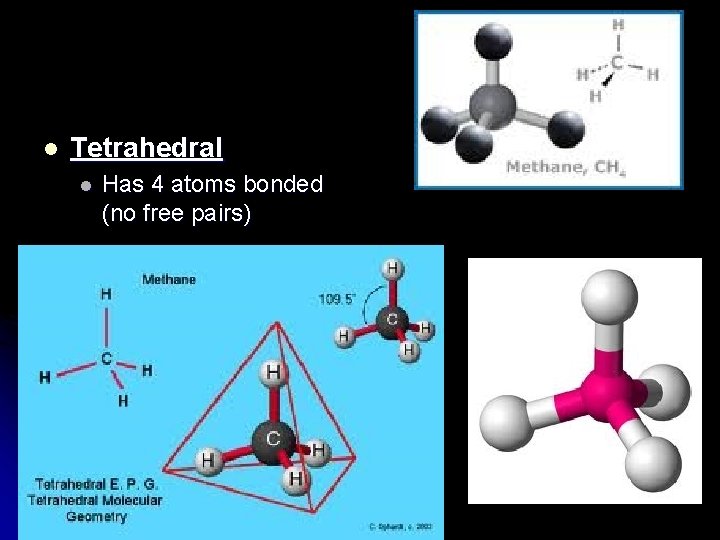
l Tetrahedral l Has 4 atoms bonded (no free pairs)
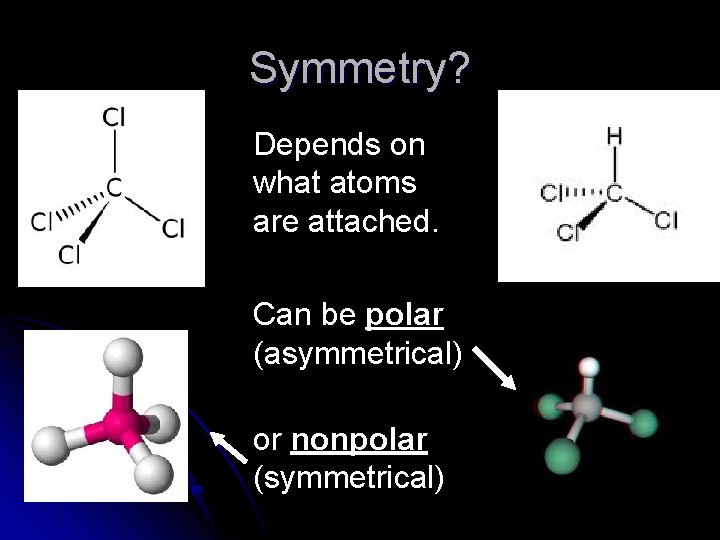
Symmetry? Depends on what atoms are attached. Can be polar (asymmetrical) or nonpolar (symmetrical)
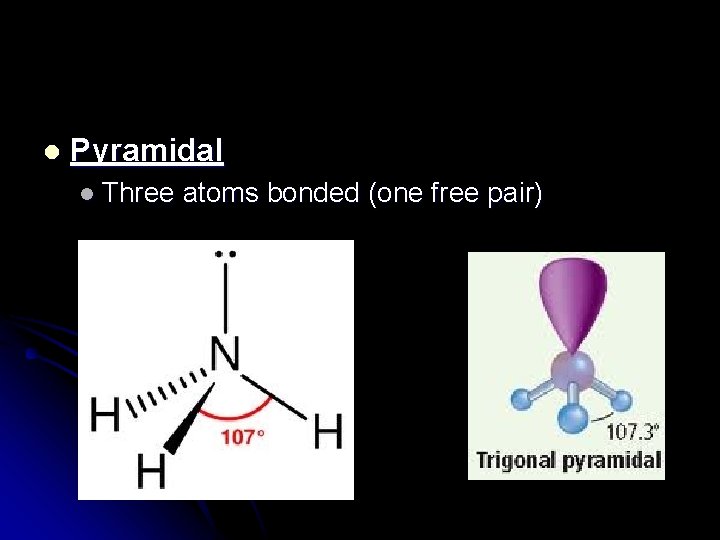
l Pyramidal l Three atoms bonded (one free pair)
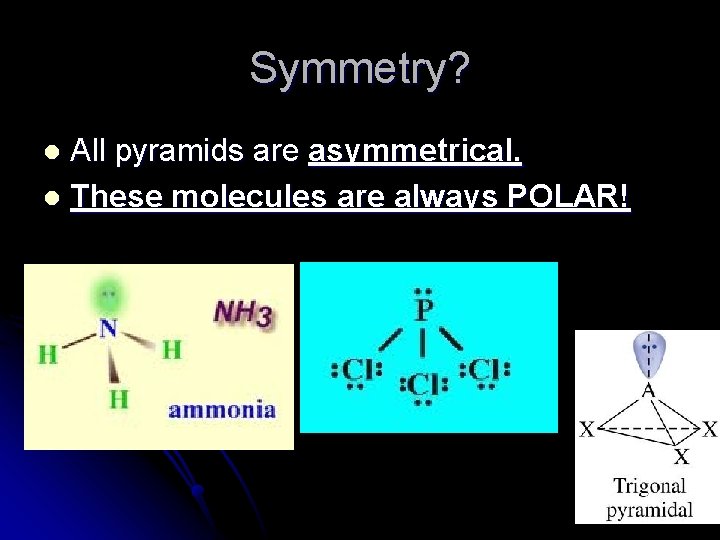
Symmetry? All pyramids are asymmetrical. l These molecules are always POLAR! l
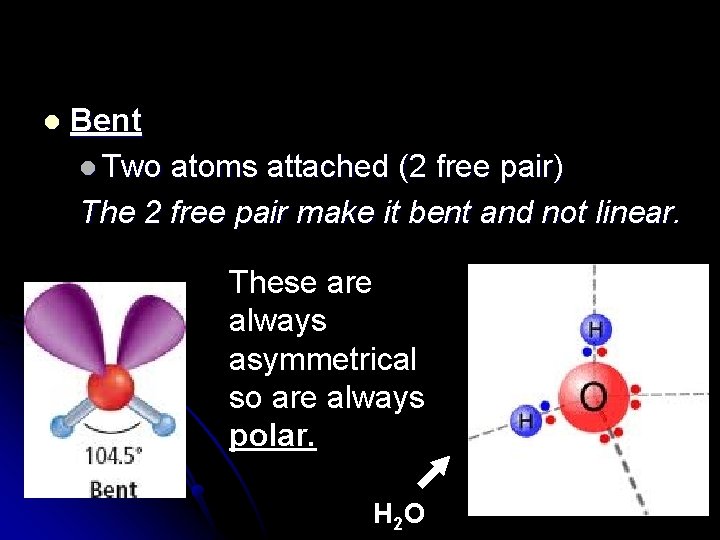
l Bent l Two atoms attached (2 free pair) The 2 free pair make it bent and not linear. These are always asymmetrical so are always polar. H 2 O

Hey, Water is Polar!!!!! Never forget this!!!
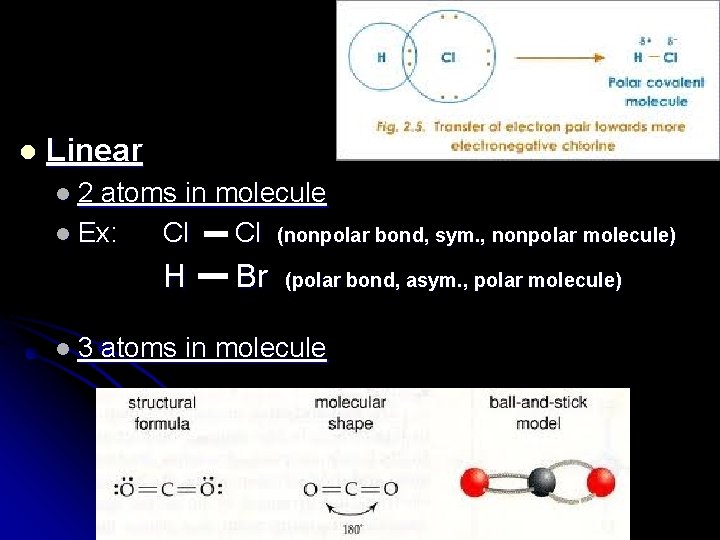
l Linear l 2 atoms in molecule l Ex: Cl Cl (nonpolar bond, sym. , nonpolar molecule) H l 3 Br (polar bond, asym. , polar molecule) atoms in molecule

Properties of Covalent Compounds

Melting Point Lower than Ionics l To melt, you are only separating the weak bonds between molecules (not within). l

Melting Point l Polar Molecules (dipoles/”mini-magnets”): l Have higher melting points than non-polars because they are harder to separate.

Solubility l Polar Molecules dissolve in Polar Solvents (like H 2 O, CHCl 3, NH 3 etc. ) l Non-polar Molecules dissolve in Non-Polar Solvents (like hexane, CCl 4) “Like Dissolves Like”

Conductivity l Covalent Molecules do not conduct well as they do not form ions. l Except Acids!!!! l Covalently bonded acids in water (aqueous) will ionize and conduct current.

Decompose l If the heat gets high enough covalent compounds will break down and decompose. l (Remember the lab, sugar melted first, then it burned and turned into black carbon)

Molecular to Empirical Formula l Molecular formulas tell the exact number of atoms in a covalent molecule. l They can be “reduced” or made into “empirical ratios”. l Ex: C 6 H 12 O 6 becomes CH 2 O

Drawing Polyatomic Ions Follow same steps for drawing covalents. l Add or subtract electrons from total valence depending on charge. l Draw brackets around ion and indicate charge. l l Ex: (SO 4) -2 6 + 4(6) + 2 = 32 electrons

Coordinate Covalent Bonding Covalent bond in which one of the bonding atoms donates both of the electrons to the bond. l The other atom donates nothing. l
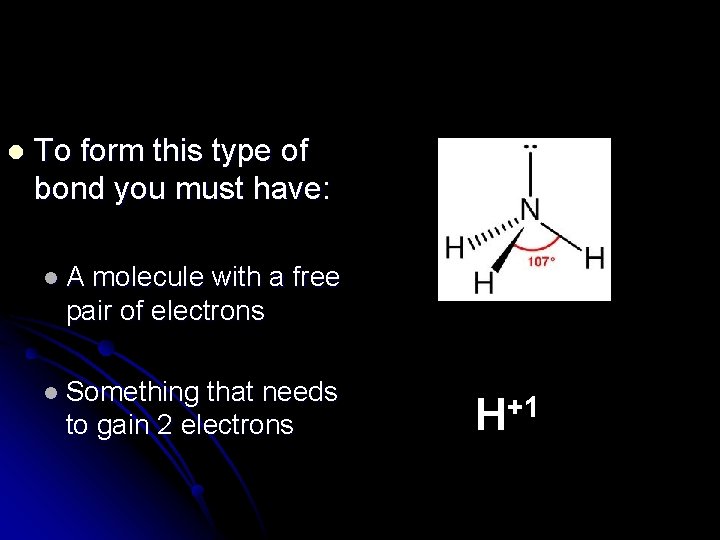
l To form this type of bond you must have: l. A molecule with a free pair of electrons l Something that needs to gain 2 electrons H+1


Exceptions to the Octet Rule (Honors) l Less than Octet l Ex: l Odd Number of Valence Electrons l Ex: l Be. H 2 NO More than Octet l Ex: SF 6
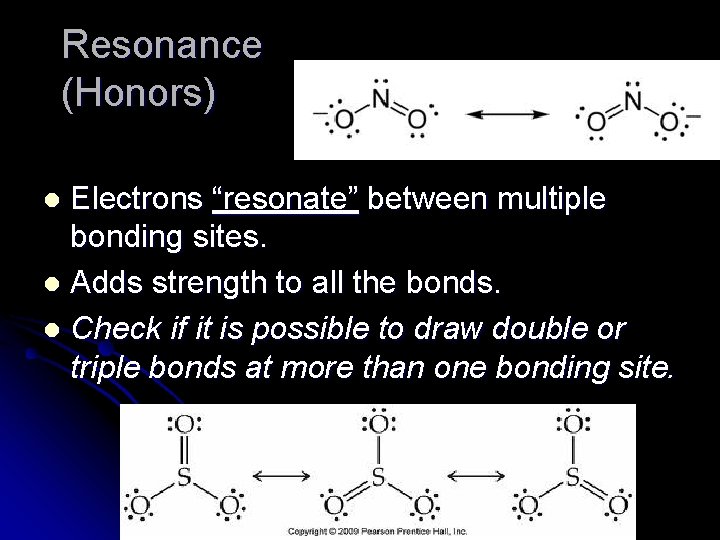
Resonance (Honors) Electrons “resonate” between multiple bonding sites. l Adds strength to all the bonds. l Check if it is possible to draw double or triple bonds at more than one bonding site. l

Sigma and Pi Bonds (Honors) l Sigma Bond: l l End to end overlap of orbitals All single bonds are sigma One of the bonds in a multiple bond is a sigma Pi Bond: l l Side to side overlap of orbitals. Found in double or triple bonds.

Network Solids Giant network of covalently bonded atoms. l Large macromolecules l Extremely strong structures l l Unusually high M. P. l Do not dissolve Diamonds are a giant network of carbon atoms.
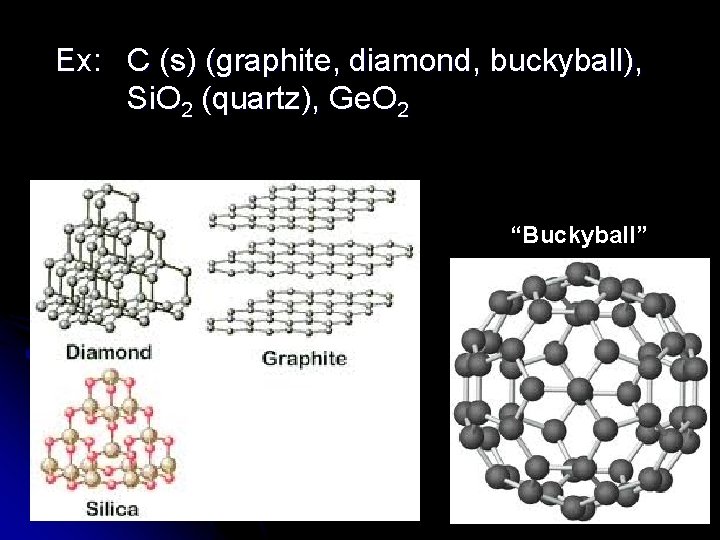
Ex: C (s) (graphite, diamond, buckyball), Si. O 2 (quartz), Ge. O 2 “Buckyball”

Metallic Bonding l Pure metals or alloys. l Ex: Mg, Fe, Brass, Au, Ni, Cu l “Delocalized” valence electrons move about between all the metal atoms.


Metallic Bonding Properties l Conducts heat and electricity very well Conducts as a solid too! l Does not dissolve in solvents l Malleable and Ductile l Relatively high melting point. l Higher MP than covalents. l Similar MP to most ionics
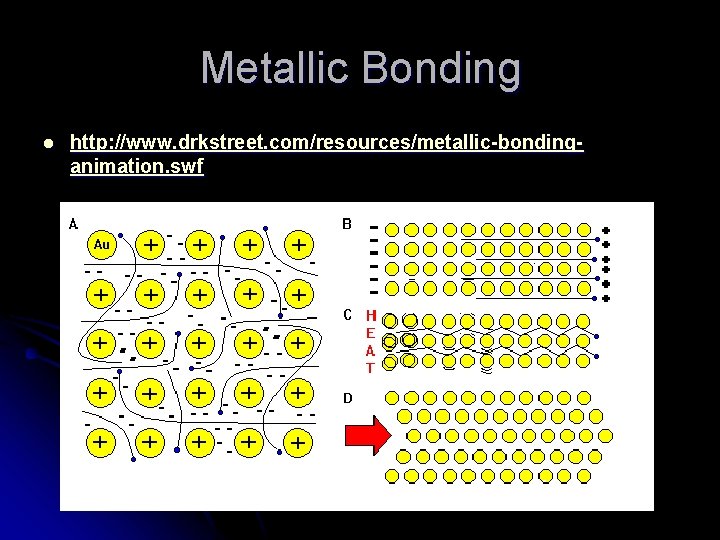
Metallic Bonding l http: //www. drkstreet. com/resources/metallic-bondinganimation. swf

VSEPR SHAPES FOR HONORS l http: //youtu. be/Fjjh. UI 4 w. FTE l Example of Shapes (honors) l http: //youtu. be/i 3 FCHVl. SZc 4 l

l l l l l Ionic vs. Covalent Bonding (short video) http: //youtu. be/Qqjc. Cvz. Wwww Songs It’s a chemical bond baby” http: //youtu. be/w. WUYHHo-z. B 0 Dancin Queen (Ionic/Covalent Bonds) http: //youtu. be/BCYr. NU-7 Sf. A Isn’t it Ionic http: //youtu. be/rw. Rtfrg. JL 5 E

Types of Bonding l http: //youtu. be/yjge 1 Wd. CFPs l http: //youtu. be/Qqjc. Cvz. Wwww l Water l http: //youtu. be/o. Swl. Z 4 Sio 9 Q l Electronegativity l http: //youtu. be/Kj 3 o 0 Xvh. Vq. Q l
- Slides: 74