Bonding and Nomenclature Ionic Bonds How Bonds Form

Bonding and Nomenclature

Ionic Bonds
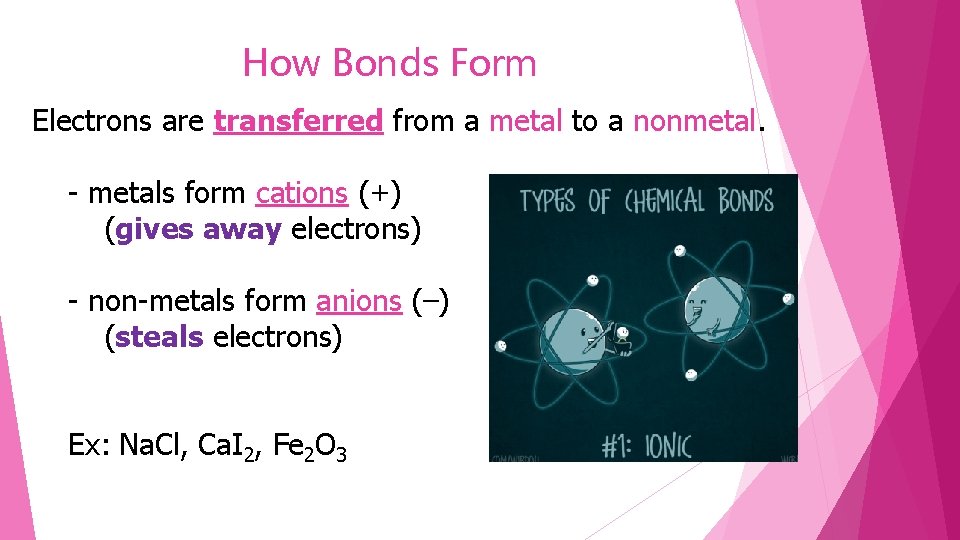
How Bonds Form Electrons are transferred from a metal to a nonmetal. - metals form cations (+) (gives away electrons) - non-metals form anions (–) (steals electrons) Ex: Na. Cl, Ca. I 2, Fe 2 O 3

Properties – Think SALT!! • ALWAYS crystalline solid at room temperature • VERY HIGH melting points and boiling points • Hard and brittle • Conducts electricity when melted or dissolved in water (ions – aqueous solution) • Usually very soluble in water (dissolves)
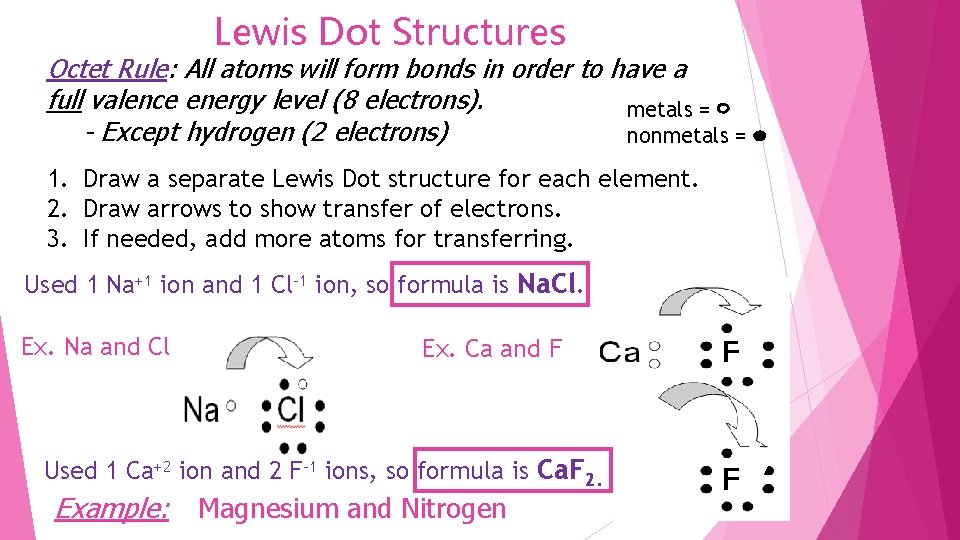
Lewis Dot Structures Octet Rule: All atoms will form bonds in order to have a full valence energy level (8 electrons). metals = - Except hydrogen (2 electrons) nonmetals = 1. Draw a separate Lewis Dot structure for each element. 2. Draw arrows to show transfer of electrons. 3. If needed, add more atoms for transferring. Used 1 Na+1 ion and 1 Cl-1 ion, so formula is Na. Cl. Ex. Na and Cl Ex. Ca and F Used 1 Ca+2 ion and 2 F-1 ions, so formula is Ca. F 2. Example: Magnesium and Nitrogen

Naming 1. Write the name of the metal. (LEAVE SPACE HERE!) 2. Change the ending of the nonmetal to –ide. Ex 1. Na. Cl = sodium chlorine = sodium chloride Ex 2. Li 2 O = lithium oxygen = lithium oxide Ex 3. Mg 3 N 2 = magnesium nitrogen = magnesium nitride

Writing Chemical Formulas 1. Write the oxidation # (charge) of each ion above symbol. 2. If the charges cancel out, only 1 of each ion is needed. 3. If the charges for each ion do NOT cancel out, use the criss cross method to determine subscripts. 4. Write the formula using ONLY symbols and subscripts. Ex. potassium phosphide +1 K P -3 K 3 P

Writing Chemical Formulas (examples) Ex 1. Calcium sulfide +2 Ca S -2 Ca. S Ex 2. Iron IV nitride +4 Fe N -3 Fe 3 N 4

Naming 1. Write the name of the metal. • Use Roman numerals for transition metals to identify the charge. • Check the charge of the nonmetal to help determine the transition metal’s charge. 2. Change the ending of the nonmetal to –ide. Ex 4. Cr 2 S 3 = chromium sulfur = chromium III sulfide Ex 5. Cu. O = copper oxygen = copper II oxide Ex 6. Cu 2 O = Copper oxygen = copper I oxide

Naming (with polyatomic ions) 1. Write the name of the metal. • Use Roman numerals for transition metals to identify the charge. • Check the charge of the nonmetal to help determine the transition metals charge. • OR ammonium (NH 4) 2. Change the ending of the nonmetal to –ide. • OR name the polyatomic ion (Do NOT change the ending!)

Naming (examples) Ex 7. NH 4 Br = ammonium bromide Ex 8. K 2 CO 3 = potassium carbonate Ex 9. Ca(NO 3)2 = Ex 10. Fe 3(PO 4)2 = calcium nitrate iron II phosphate

Writing Chemical Formulas (examples) Ex 3. sodium phosphate +1 Na (PO 4) -3 Na 3 PO 4 or Na 3(PO 4) Ex 4. beryllium nitrite +2 Be (NO 2) -1 Be(NO 2)2 Ex 5. nickel II sulfate +2 Ni (SO 4) -2 Ni. SO 4 or Ni(SO 4)

Covalent Bonds

How Bonds Form Electrons are shared between 2 or more nonmetal atoms. Can also be between a nonmetal and a metalloid. - EN value difference less than 1. 5 Ex: H 2 O, CO 2, C 6 H 12 O 6 All nonmetal atoms!

Properties – think sugar! • • • Can be solid, liquid, or gas at room temp. Low melting and boiling points Cannot conduct electricity Insoluble in water Polar Covalent bonds – electrons shared unequally (EN difference 0. 5 – 1. 5) Creates partial + and – ends • Nonpolar Covalent bonds – electrons shared equally (EN difference 0 – 0. 5) • Compounds are called “molecules”
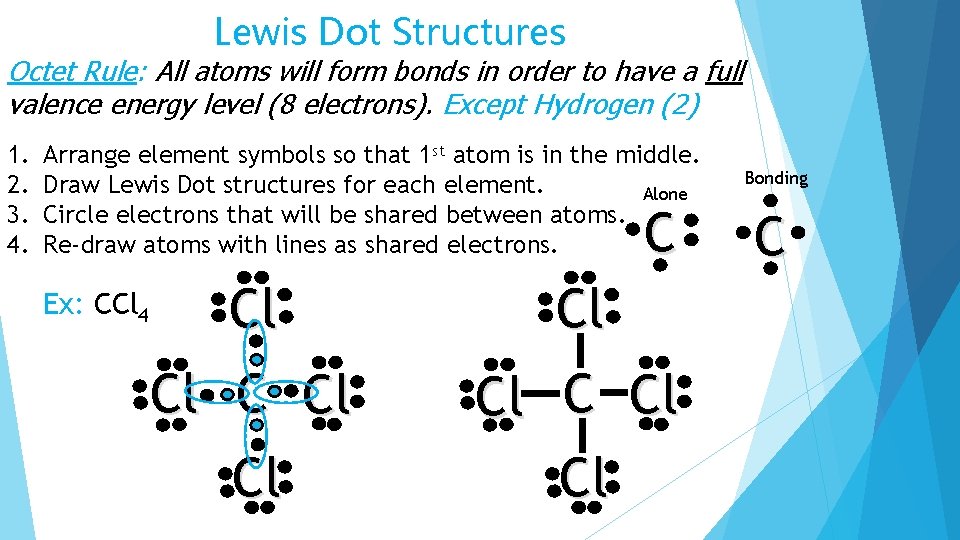
Lewis Dot Structures Octet Rule: All atoms will form bonds in order to have a full valence energy level (8 electrons). Except Hydrogen (2) 1. 2. 3. 4. Arrange element symbols so that 1 st atom is in the middle. Draw Lewis Dot structures for each element. Alone Circle electrons that will be shared between atoms. Re-draw atoms with lines as shared electrons. C Ex: CCl 4 Cl Cl Cl Cl Bonding C

Covalent Bonds between 2 elements can vary! • 1 pair of electrons shared = SINGLE bond • 2 pairs of electrons shared = DOUBLE bond • 3 pairs of electrons shared = TRIPLE bond

Naming 1. List the prefix and name of 1 st nonmetal. Don’t use “mono” on 1 st element! 2. Prefix and change the ending of the 2 nd nonmetal to -ide. Prefixes Examples 1 = mono 6 = hexa CO 2 Carbon dioxide 7 = hepta 2 = di S 2 O 3 Disulfur trioxide 8 = octa 3 = tri N 4 F Tetranitrogen monofluoride 4 = tetra 9 = nona CO Carbon monoxide 5 = penta 10 = deca

Writing Chemical Formulas 1. Write the symbol of the first nonmetal and subscript to match the prefix. 2. Repeat for 2 nd nonmetal. DO NOT SIMPLIFY SUBSCRIPTS! Diatomic elements: H 2 N 2 O 2 F 2 Cl 2 Br 2 I 2 Ex: Triphosphorus pentachloride P 3 Cl 5 Ex: Tricarbon hexasulfide C 3 S 6 Ex: Antimony tetrabromide Sb. Br 4
- Slides: 19