Bonding and Chemical Reactions Compounds and Molecules Chemical

Bonding and Chemical Reactions

Compounds and Molecules • Chemical Bond: forces that hold atoms together in a compound. • Chemical Structure: the arrangement of atoms in a substance

Chemical Structure • compounds with networks are strong solids • ex: diamond, quartz, many rocks • networked compounds have boiling points • ex: salts, rocks, diamonds melting and

Chemical Structure • Some compounds are made of molecules • ex: H , O , N , CO are all gases with weak 2 2 attraction • hydrogen bonds are very strong
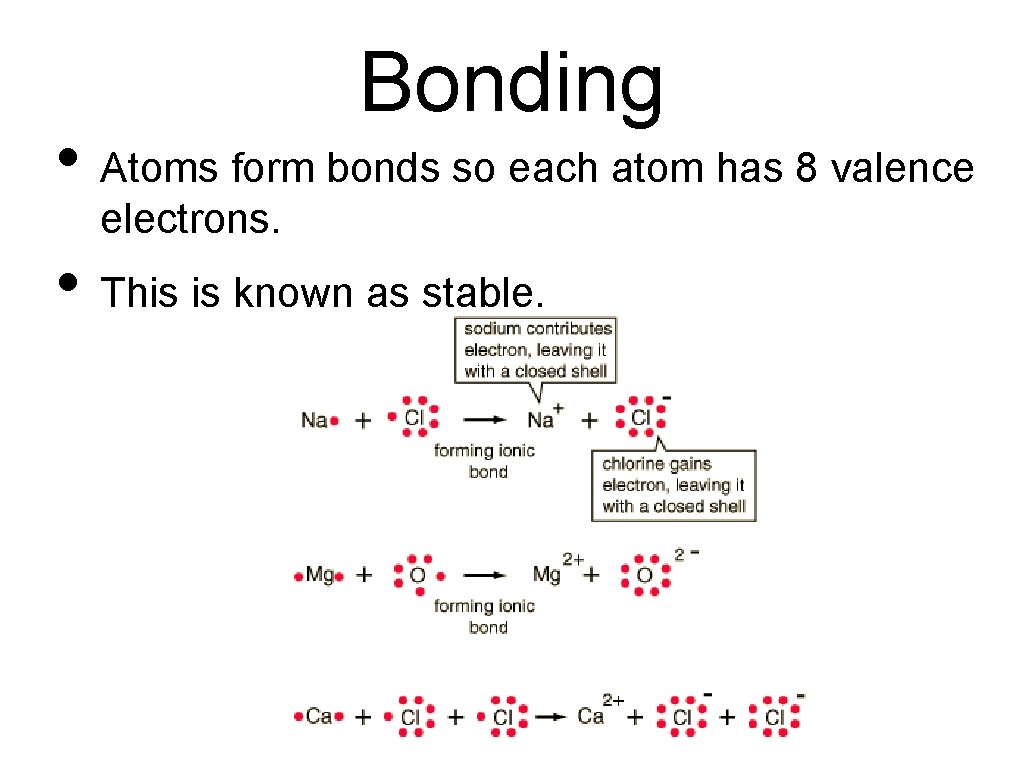
Bonding • Atoms form bonds so each atom has 8 valence electrons. • This is known as stable.
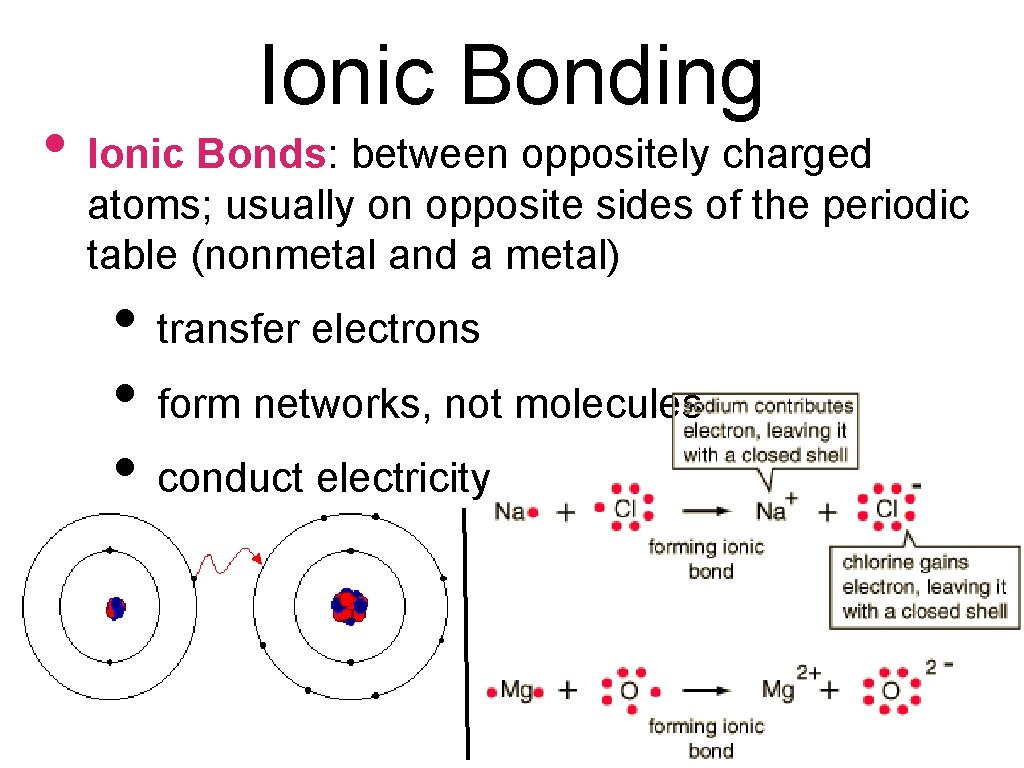
Ionic Bonding • Ionic Bonds: between oppositely charged atoms; usually on opposite sides of the periodic table (nonmetal and a metal) • transfer electrons • form networks, not molecules • conduct electricity
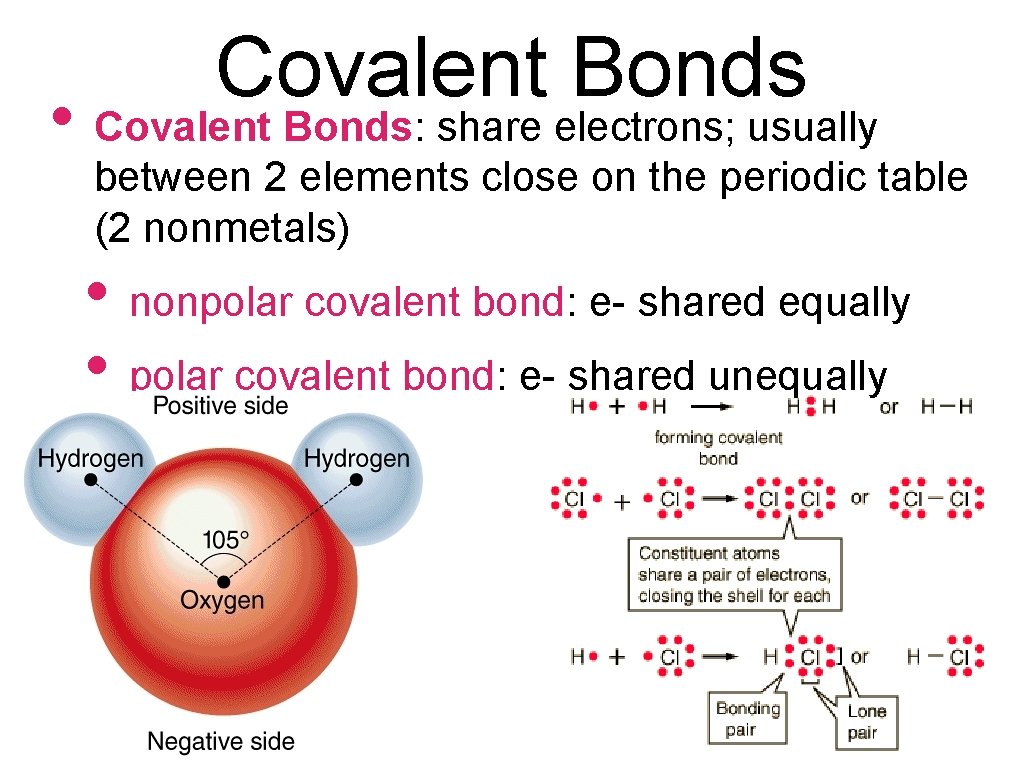
Covalent Bonds • Covalent Bonds: share electrons; usually between 2 elements close on the periodic table (2 nonmetals) • nonpolar covalent bond: e- shared equally • polar covalent bond: e- shared unequally

Compound Names and Formulas • Compounds have specific names so you can tell how many and what atoms are in the compound/molecule.

Ionic Naming • Cations are just the name of the element • Anions have -ide at the end of the element’s name Cation is ALWAYS FIRST Sodium Chloride Anion is ALWAYS LAST Ionic compounds MUST have a total charge of zero!! Na+1 + Cl-1 = Na. Cl = 0 charge!

Transition Metal Naming • Transition metals show their charges as Roman Numerals because they can change charge!! Fe. O = Fe+2 + O-2 Iron (II) Oxide Fe 2 O 3 = Fe+3 + O-2 Iron (III) Oxide

What is the formula for Lithium Oxide?

What is the name for Cu. Cl 2?

Covalent Naming • To name covalent compounds, you must put a prefix in front of the element’s name. 1. mono 2. di 3. tri 4. tetra 5. penta- 6. hexa 7. hepta 8. octa 9. nona 10. deca- If you only have 1 cation, you don’t need a prefix!

What is the name of H 2 O?

Formulas • Molecular Formula: how many atoms are in a molecule of a compound GLUCOSE Molecular Formula C 6 H 12 O 6

Chemical Reactions 1. Rearrange atoms 2. Involve changes in energy • • Exothermic: release energy (heat, fire, fizz, etc) Endothermic: absorb energy (drop in temp. )
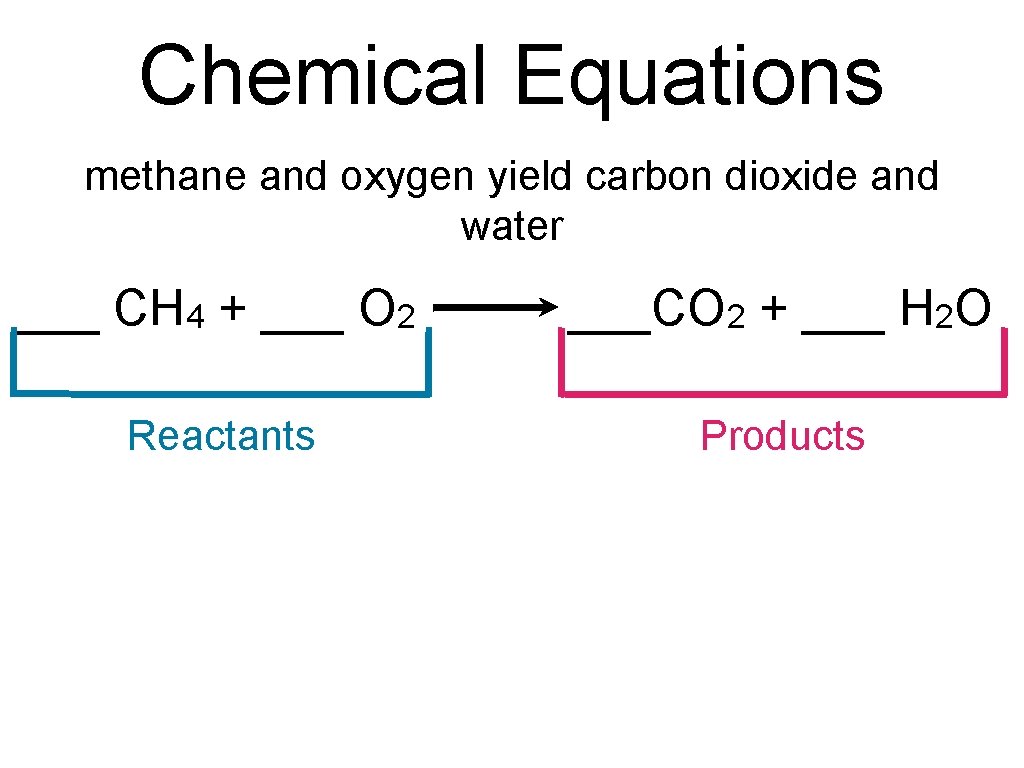
Chemical Equations methane and oxygen yield carbon dioxide and water ___ CH 4 + ___ O 2 ___CO 2 + ___ H 2 O Reactants Products
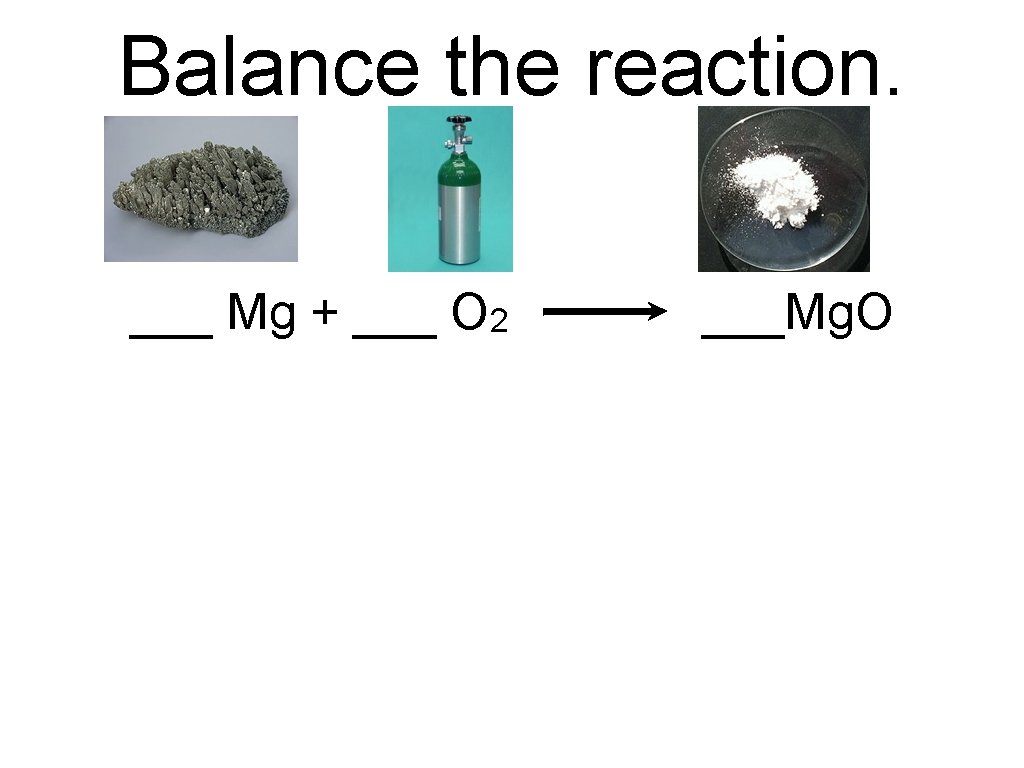
Balance the reaction. ___ Mg + ___ O 2 ___Mg. O
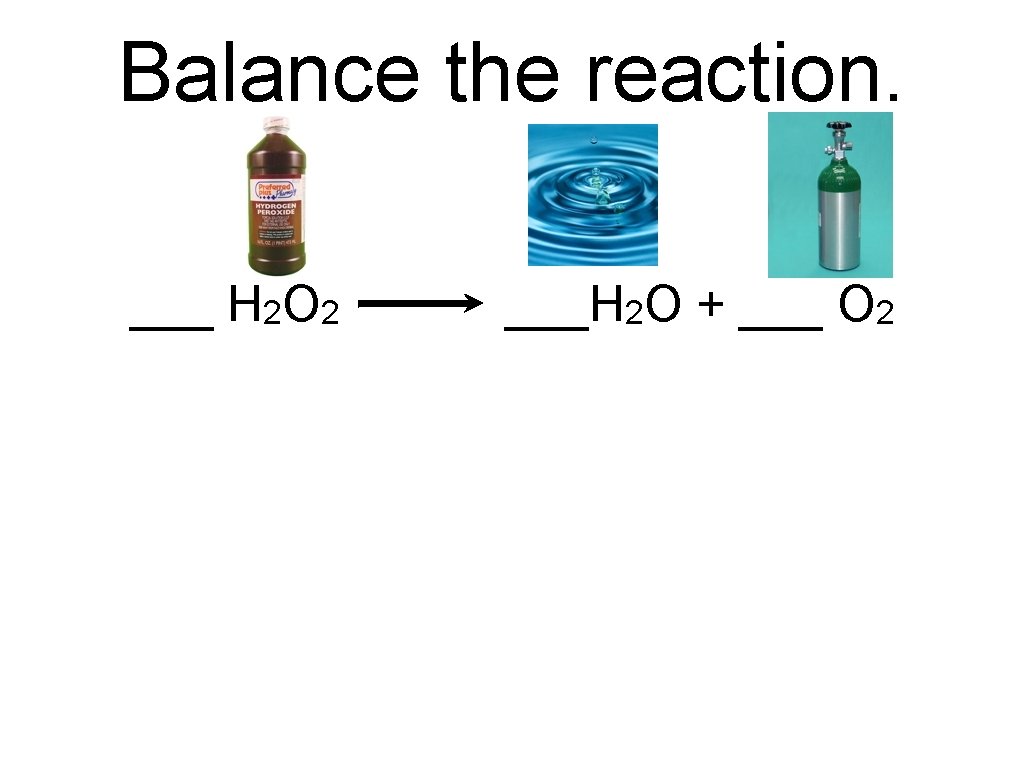
Balance the reaction. ___ H 2 O 2 ___H 2 O + ___ O 2

Reaction Types • Synthesis: • A+B AB • Decomposition: • AB A+B • Combustion: ALWAYS HAS O 2 AS A REACTANT! • AB + O 2 AO + BO

Reaction Types • Single-Replacement (single-displacement): • AX + B A + BX • Double-Replacement (double-displacement): • AX + BY AY + BX
- Slides: 21