BONDING A Review BONDING REVIEW 1 Be able

BONDING A Review

BONDING REVIEW 1. Be able to determine the valence electrons of representative elements using the periodic table. (Do not worry about transition or rare earth elements. ) Group 1 Group 2 Group 13 Group 14 1 valence 2 valence 3 valence 4 valence You get the idea…. .
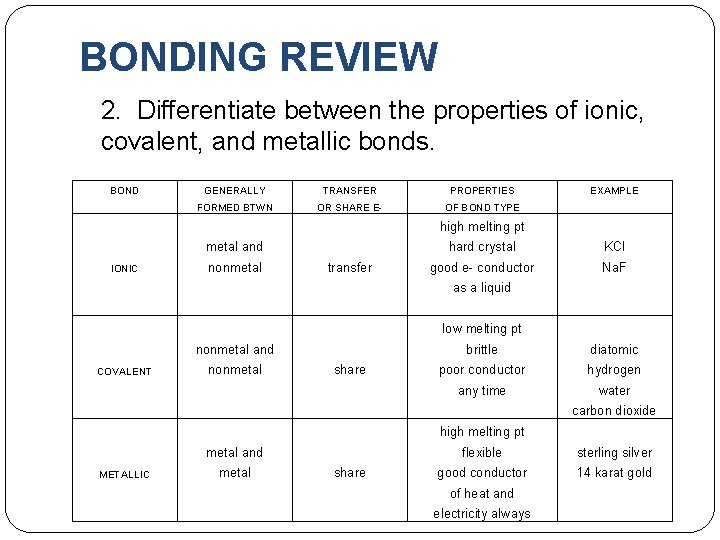
BONDING REVIEW 2. Differentiate between the properties of ionic, covalent, and metallic bonds. BOND GENERALLY TRANSFER PROPERTIES EXAMPLE FORMED BTWN OR SHARE E- OF BOND TYPE high melting pt metal and hard crystal KCl good e- conductor Na. F as a liquid IONIC nonmetal low melting pt nonmetal and brittle diatomic COVALENT nonmetal poor conductor hydrogen any time water carbon dioxide high melting pt metal and flexible sterling silver good conductor 14 karat gold of heat and electricity always METALLIC metal transfer share
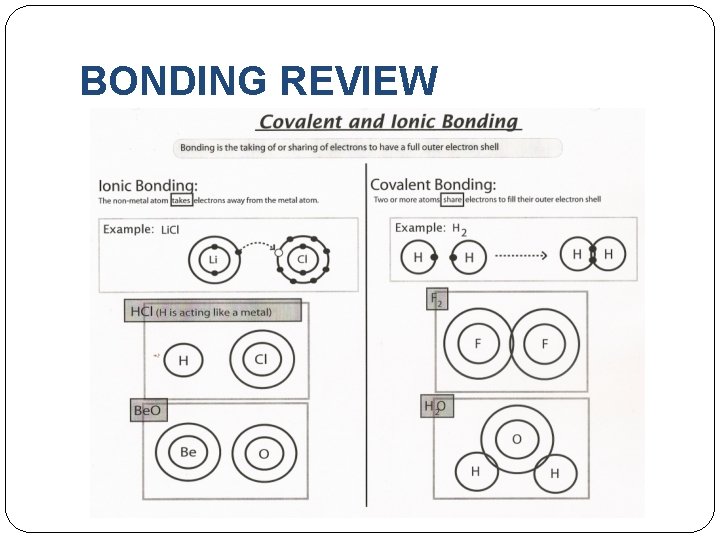
BONDING REVIEW

BONDING REVIEW 3. Given two atoms involved in a bond, determine if the bond is ionic or covalent using just the positions of the elements on the PERIODIC TABLE. Be able to do for any compound. metal + nonmetal = ionic nonmetal + nonmetal = covalent metal + metal = metallic

BONDING REVIEW 4. Predict the type of bond formed (ionic, polar covalent, nonpolar covalent) using ELECTRONEGATIVITY VALUES. Be able to do for any compound. <0. 5 covalent 0. 5 to 1. 67 covalent nonpolar

BONDING REVIEW 5. Use the VSEPR theory to explain the basic shapes of simple molecules. Valence Shell Electron Pair Repulsion theory is based on the number of regions of high electron density around a central atom. can be used to predict structures of molecules or ions by minimizing the electrostatic repulsion between the regions of high electron density. can also be used to predict structures of molecules or ions that contain multiple bonds or unpaired electrons.

BONDING REVIEW 6. Be able to identify the basic shapes of simple molecules from their chemical formula - linear, trigonal planar, tetrahedral, pyramidal, and bent. Be able to fill out the chart you have had for homework and for lab. You will see it on the test.

BONDING REVIEW 7. Be able to identify the polarity of simple molecules. (Not the polarity of the bond, but the polarity of the whole molecule. ) Terminal Atoms alike different �Linear nonpolar �Trigonal planar nonpolar �Tetrahedral nonpolar �Pyramidal polar �Bent polar Terminal Atoms polar polar

BONDING REVIEW 8. Describe hybrid orbitals and be able to determine the hybridization of the central atom involved in bonding. You have three choices: - linear 1 s and 1 p = sp usually 2 A group elements - trigonal planar 1 s and 2 p = usually 3 A group elements - tetrahedral 1 s and 3 p = usually 4 A group elements sp 2 sp 3
- Slides: 10