Bond Polarity and Electronegativity In a covalent bond

Bond Polarity and Electronegativity • In a covalent bond, electrons are shared. • Sharing of electrons to form a covalent bond does not imply equal sharing of those electrons. • There are some covalent bonds in which the electrons are located closer to one atom than the other. • Unequal sharing of electrons results in polar bonds. (

Electronegativity • Electronegativity: The ability of one atom in a molecule to attract electrons to itself. • Pauling set electronegativities on a scale from 0. 7 (Cs) to 4. 0 (F). • Electronegativity increases • across a period and • up a group.
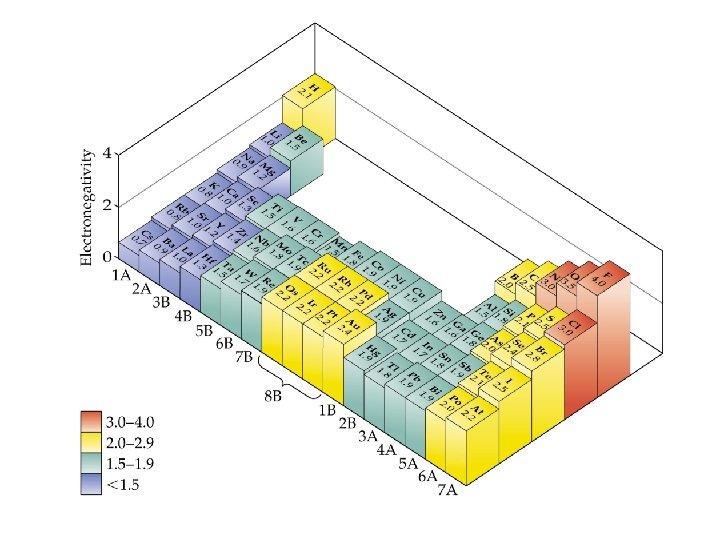

Electronegativity and Bond Polarity • Difference in electronegativity is a gauge of bond polarity (4. 2. 5): • electronegativity differences less than 0. 5 result in non-polar covalent bonds (equal or almost equal sharing of electrons); • electronegativity differences around from 0. 5 to 2 result in polar covalent bonds (unequal sharing of electrons); • electronegativity differences greater than 2 result in ionic bonds (transfer of electrons).
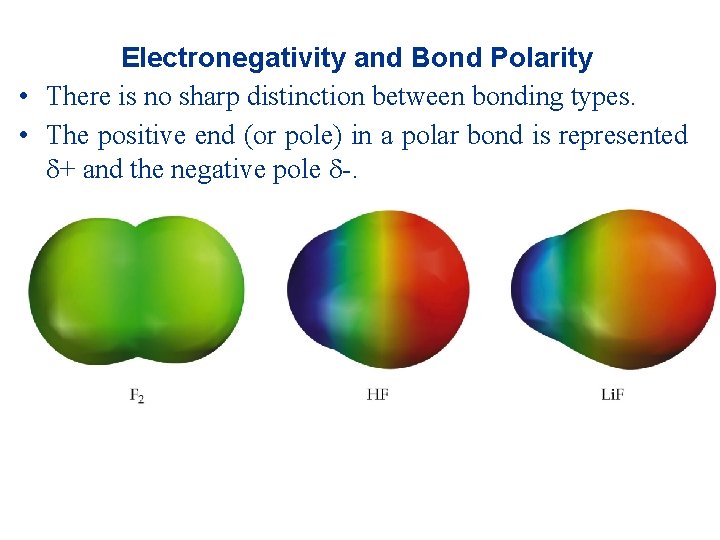
Electronegativity and Bond Polarity • There is no sharp distinction between bonding types. • The positive end (or pole) in a polar bond is represented + and the negative pole -.

Dipole Moments • Consider HF: • The difference in electronegativity leads to a polar bond. • There is more electron density on F than on H. • Since there are two different “ends” of the molecule, we call HF a dipole. • Dipole moment, m, is the magnitude of the dipole: where Q is the magnitude of the charges. • Dipole moments are measured in debyes, D.

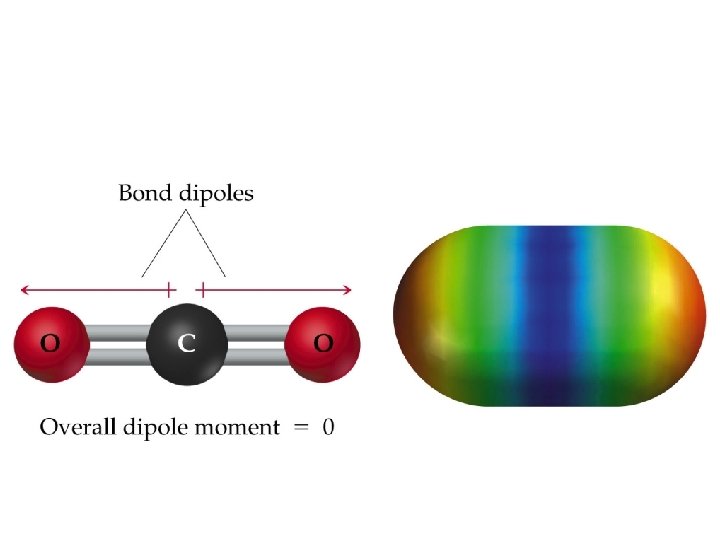
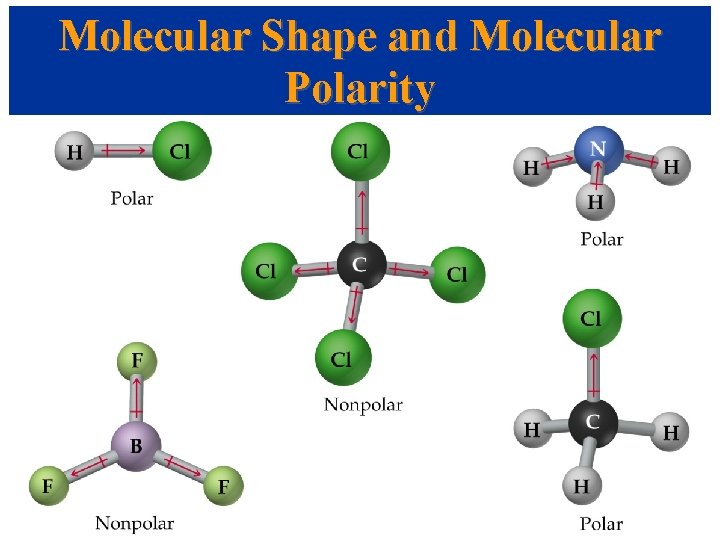
Molecular Shape and Molecular Polarity (4. 2. 6) • When there is a difference in electronegativity between two atoms, then the bond between them is polar. • It is possible for a molecule to contain polar bonds, but not be polar. • For example, the bond dipoles in CO 2 cancel each other because CO 2 is linear.

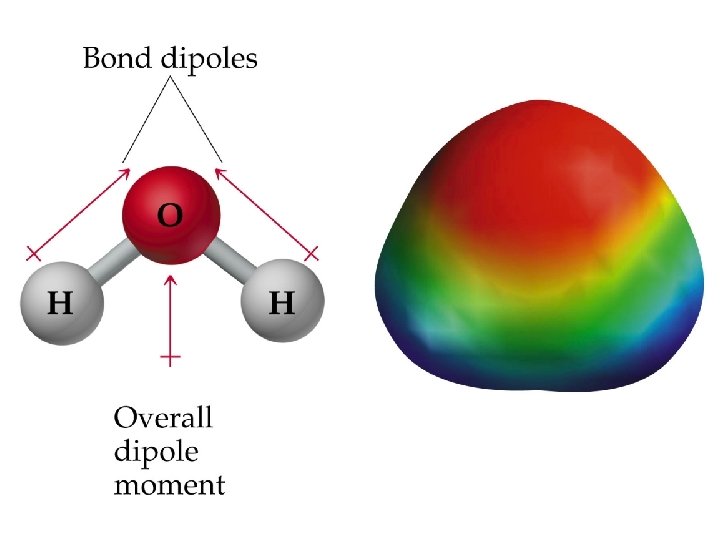
• In water, the molecule is not linear and the bond dipoles do not cancel each other. • Therefore, water is a polar molecule. • The overall polarity of a molecule depends on its molecular geometry (symmetry).
Molecular Shape and Molecular Polarity

Examples – Determine whether each is polar or nonpolar: 1. CCl 4 2. PCl 3 3. BF 3 4. I 35. SO 3

Examples – Determine whether each is polar or nonpolar: 1. CCl 4 nonpolar 2. PCl 3 polar 3. BF 3 nonpolar 4. I 3 nonpolar 5. SO 3 nonpolar

A Molecular Comparison of Liquids and Solids • Physical properties of substances understood in terms of kinetic molecular theory: – Gases are highly compressible, assume shape and volume of container: • Gas molecules are far apart and do not interact much with each other. – Liquids are almost incompressible, assume the shape but not the volume of container: • Liquids molecules are held closer together than gas molecules, but not so rigidly that the molecules cannot slide past each other.

– Solids are incompressible and have a definite shape and volume: • Solid molecules are packed closely together. The molecules are so rigidly packed that they cannot easily slide past each other.
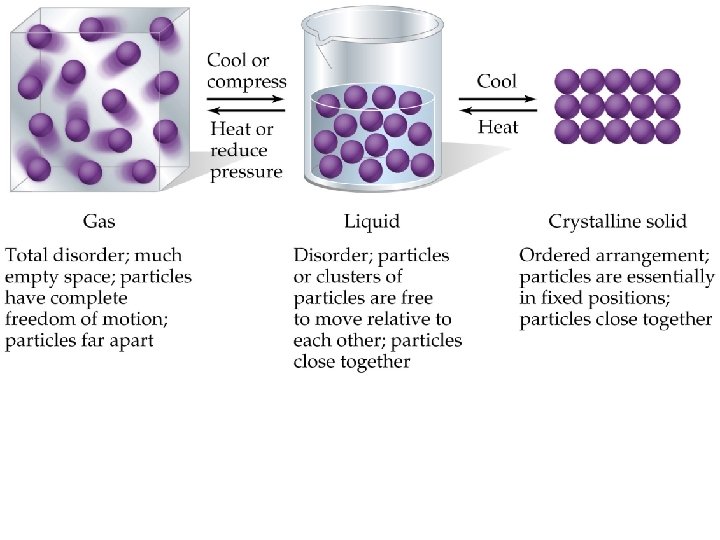

• Converting a gas into a liquid or solid requires the molecules to get closer to each other: – cool or compress. • Converting a solid into a liquid or gas requires the molecules to move further apart: – heat or reduce pressure. • The forces holding solids and liquids together are called intermolecular forces.

• The covalent bond holding a molecule together is an intramolecular force. • The attraction between molecules is an intermolecular force. • Intermolecular forces are much weaker than intramolecular forces (e. g. 16 k. J/mol vs. 431 k. J/mol for HCl). • When a substance melts or boils the intermolecular forces are broken (not the covalent bonds).
- Slides: 18