Bond Ionic Bond To get started We should

Bond. Ionic Bond.

To get started… We should refresh our memories about the structure of atoms: _____ Protons and _____ neutrons are found inside the nucleus, _____ are found in electrons energy levels outside the nucleus. _______ The electrons in the energy level farthest from the nucleus are called ______ valence electrons.

Valence matters Valence electrons are the only particles that are involved in _________. chemical reactions Every atoms wants to have a ______ full valence shell – it’s more ____ stable than an unfilledone. _____ This gives them the same electron noble gas configuration as a ______!

Octet Rule This filling of your valence shell is known as the _______Octet _____. rule This means that atoms will _____ combine in such a way that they have ___8 electrons in their valence shells – just like a Noble gas _______.

Win, Lose or Draw There are ___ 3 options for filling your valence shell: 1. You can _______ lose electrons. 2. You can _______ gain electrons. 3. You can _______ share electrons. The option you choose all depends on how many _____ electrons you _______ valence with… start

Alkali Metals Consider ____ Li for a moment… Lithium has ___ 1 valence electron. 3 P 4 N But its valence shell can hold ___. 8 So Li has to ____ gain 7 electrons to fill its valence shell…?

What if…? What if Li gave up its one valence electron? 3 P 4 N

What if…? What if Li gave up its one valence electron? 3 P 4 N

What if…? What if Li gave up its one valence electron? 3 P 4 N

What if…? What if Li gave up its one valence electron? We are left with _____ one energy level, which now becomes the _____ shell! valence (And would you look at that – it’s FULL already _______!!!!) 3 P 4 N

The Path of Least Resistance Atoms will take the _____ shortest path to a full valence shell. **Keep in mind that this may be backward _____!*
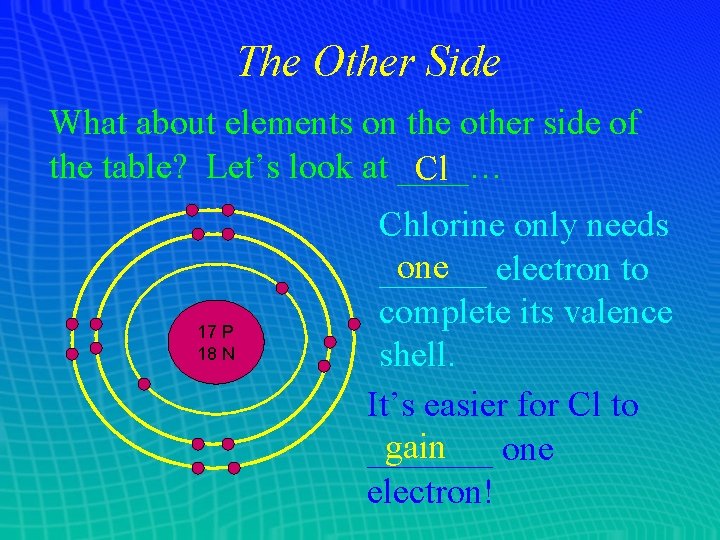
The Other Side What about elements on the other side of the table? Let’s look at ____… Cl 17 P 18 N Chlorine only needs one electron to ______ complete its valence shell. It’s easier for Cl to gain _______ one electron!

Which way? Here’s the general rule: If you have ______ less than 4 valence electrons, you will tend to ______ lose them. If you have ______ more than 4 valence gain them. electrons, you will tend to _____ metals tend to ______ lose Therefore, _____ nonmetals tend to electrons, and ______ gain them!

Charges, charges… Remember what you become when you lose or gain electrons? (An _____!) ion **An ion is a ____ charged particle – with an ____ unequal number of _____ protons and electrons _____. And remember that electrons have a _____ negative charge, so if you _______ lose electrons, you become a _____ positive ion!
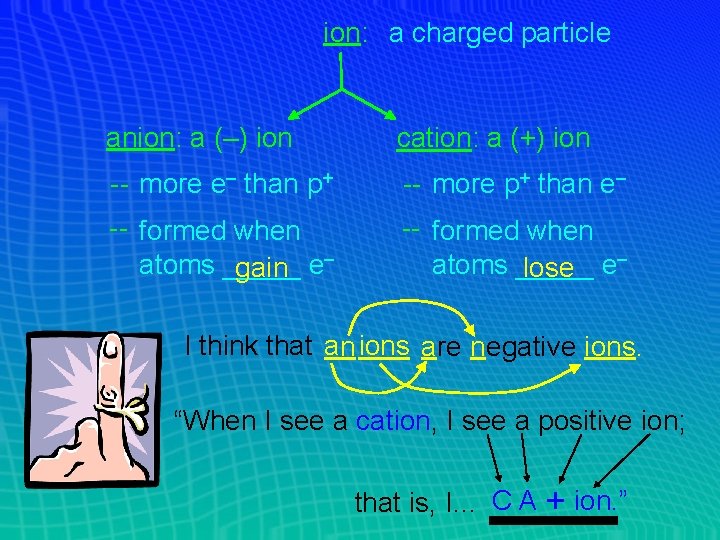
ion: a charged particle anion: a (–) ion cation: a (+) ion -- more e– than p+ -- more p+ than e– -- formed when atoms _____ gain e– -- formed when atoms _____ lose e– I think that anions are negative ions. “When I see a cation, I see a positive ion; that is, I… C A + ion. ”

What now…? What you end up with is an atom that will _____lose electrons and become a _____ ion… positive Right next to an atom that will ______ gain electrons and become a ____ negative ion… Hmmm… What do we know about opposites and attraction? ?
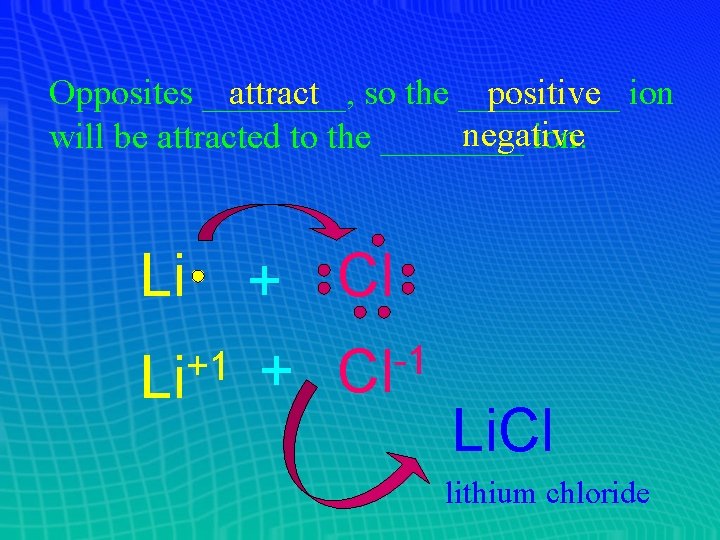
Opposites ____, attract so the _____ positive ion negative will be attracted to the ____ ion. Li + Cl -1 +1 Li + Cl Li. Cl lithium chloride

Match made in Heaven! This ____ transfer of electrons is known as an Ionic Bond _______! Ionic bonds are always formed between a _______ metal and a _____! nonmetal transfer of electrons creates The _____ ions attractive forces ______, and the ______ between the ions are what hold the compound ______ together.

Those ‘Dot’ things again? !? To show ionic bonds, we use the Electron Dot diagrams – and draw _____ to show the _____ of electrons arrows transfer between atoms. Make sure you show the ____ ion that results from that transfer of electrons!
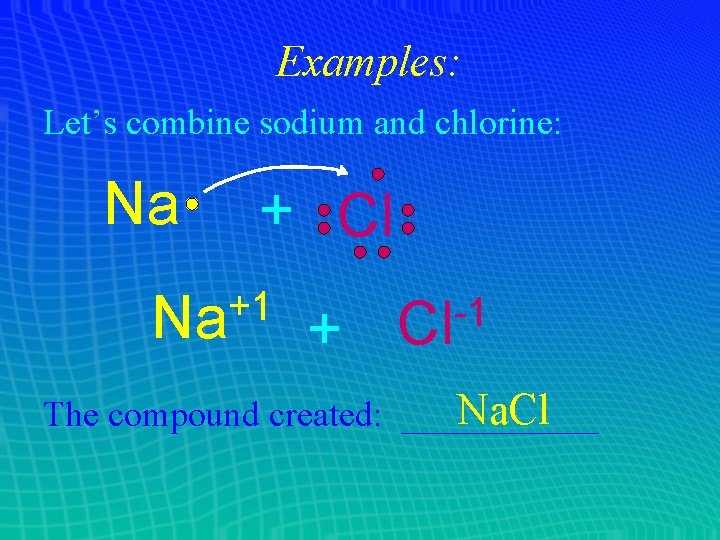
Examples: Let’s combine sodium and chlorine: Na + Cl +1 Na + -1 Cl Na. Cl The compound created: ______

Naming Ionic Compounds Ionic compounds are always formed metal and a _____. nonmetal between a _______ To name them: 1. ____ Metal goes first. 2. ______ Nonmetal goes second – but you change the end of the nonmetal’s name to “-ide” _______. Example: Na. Cl = sodium chloride

More than one? ? ? What happens if one atom needs to lose 2 electrons, but the other only gets __? 1 ___ Mg Cl Now Cl’s valence shell is _____, complete but Mg still has one electron to unload! Each Cl atom can only ____ accept 1 electron – but who says there’s only one Cl hanging around?
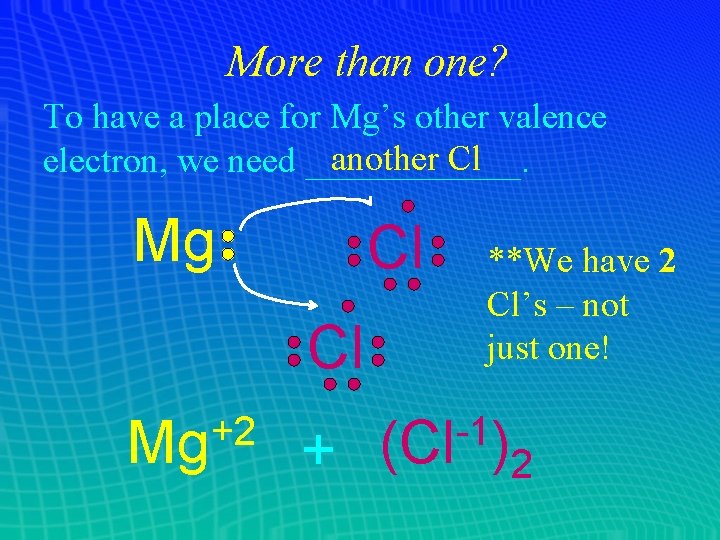
More than one? To have a place for Mg’s other valence another Cl electron, we need ______. Mg Cl Cl +2 Mg + **We have 2 Cl’s – not just one! -1 (Cl ) 2

More than one? So our formula is really: +2 Mg + -1 Cl -- OR -Mg. Cl 2 Name: _________ magnesium chloride

Things to remember… • Ionic bonds are formed between a metal and a _____. nonmetal ____ • Metals ______ lose electrons and become ___ + nonmetals ______gain ions; them and become ____ ions. arrows to show the ____ transfer of • Draw ____ ionswith their electrons, then draw the _________. charges • When naming, it’s _______, metal then _____ nonmetal -- ending in _______. “-ide”
- Slides: 25