Bond insertion in metalcarbon dioxide anionic clusters studied

Bond insertion in metal-carbon dioxide anionic clusters studied by infrared photodissociation spectroscopy Leah G. Dodson, Michael C. Thompson, J. Mathias Weber JILA, NIST, and Department of Chemistry and Biochemistry University of Colorado, Boulder, Colorado RA 03 73 rd International Symposium on Molecular Spectroscopy June 21, 2018
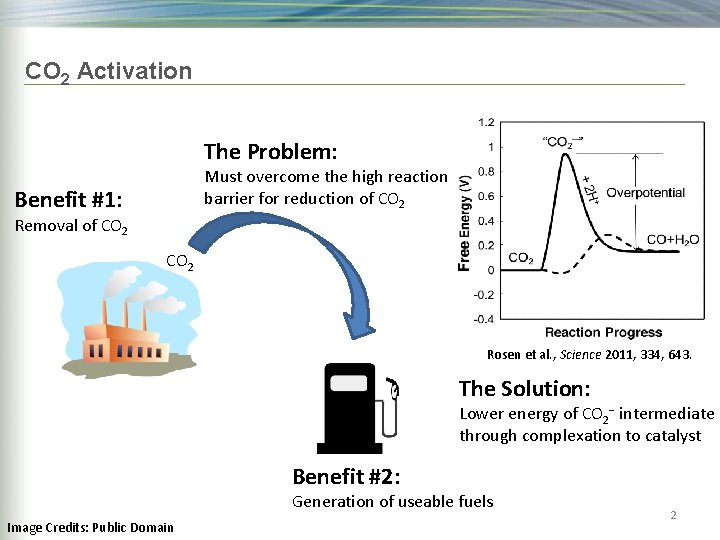
CO 2 Activation The Problem: Must overcome the high reaction barrier for reduction of CO 2 Benefit #1: Removal of CO 2 Rosen et al. , Science 2011, 334, 643. The Solution: Lower energy of CO 2− intermediate through complexation to catalyst Benefit #2: Generation of useable fuels Image Credits: Public Domain 2
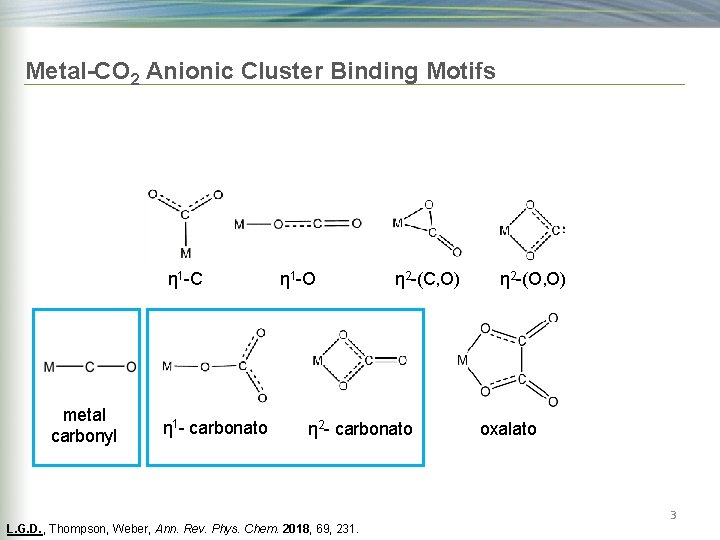
Metal-CO 2 Anionic Cluster Binding Motifs η 1 -C metal carbonyl η 1 - carbonato η 1 -O η 2 -(C, O) η 2 - carbonato η 2 -(O, O) oxalato 3 L. G. D. , Thompson, Weber, Ann. Rev. Phys. Chem. 2018, 69, 231.
![Infrared Photodissociation Spectroscopy [MOx(CO 2)y]− - Laser ablation - CO 2 expansion M CO Infrared Photodissociation Spectroscopy [MOx(CO 2)y]− - Laser ablation - CO 2 expansion M CO](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-4.jpg)
Infrared Photodissociation Spectroscopy [MOx(CO 2)y]− - Laser ablation - CO 2 expansion M CO 2 4
![[MOx(CO 2)y]− (x = 0– 3, y ≥ 0) Formation [M(CO 2)y]−, Supersonic Expansion [MOx(CO 2)y]− (x = 0– 3, y ≥ 0) Formation [M(CO 2)y]−, Supersonic Expansion](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-5.jpg)
[MOx(CO 2)y]− (x = 0– 3, y ≥ 0) Formation [M(CO 2)y]−, Supersonic Expansion CO 2 [MO(CO 2)y]−, [MO 2(CO 2)y]−, … Metal Target Lens λ=355 nm 35– 40 m. J pulse− 1 Even-Lavie Valve 5 Weber, Rev. Sci. Instrum. 2005, 76, 043301.
![Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 1. Mass Selection 2. Photodissociation [MOx(CO 2)y]− Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 1. Mass Selection 2. Photodissociation [MOx(CO 2)y]−](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-6.jpg)
Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 1. Mass Selection 2. Photodissociation [MOx(CO 2)y]− - Laser ablation - CO 2 expansion M CO 2 [M(CO 2)y]− + hν 6
![Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 hν hot cluster [M(CO 2)y]− + hν Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 hν hot cluster [M(CO 2)y]− + hν](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-7.jpg)
Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 hν hot cluster [M(CO 2)y]− + hν [M(CO 2)y-1]− + CO 2 7
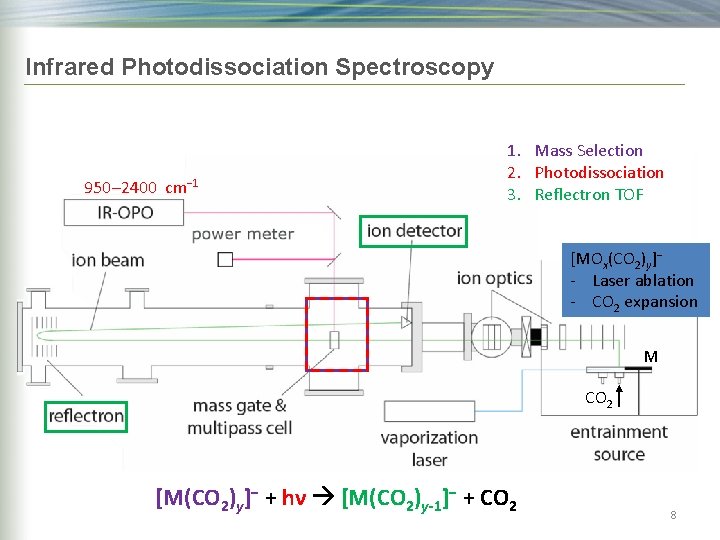
Infrared Photodissociation Spectroscopy 950– 2400 cm− 1 1. Mass Selection 2. Photodissociation 3. Reflectron TOF [MOx(CO 2)y]− - Laser ablation - CO 2 expansion M CO 2 [M(CO [Ti(CO 22))4 y]]−− + hν [M(CO 2)y-1]− + CO 2 8
![Photodissociation Spectroscopy of [Ti(CO 2)y]− [Ti(CO 2)4]− Ligand modes Solvent CO 2 asym str Photodissociation Spectroscopy of [Ti(CO 2)y]− [Ti(CO 2)4]− Ligand modes Solvent CO 2 asym str](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-9.jpg)
Photodissociation Spectroscopy of [Ti(CO 2)y]− [Ti(CO 2)4]− Ligand modes Solvent CO 2 asym str • [Ti(CO 2)4]− • Smallest cluster with measured CO 2 photodissociation • One weakly-bound solvent molecule • Three strongly-bound ligands 9 L. G. D. , Thompson, Weber, 2018, J. Phys. Chem. A 2018, 122, 2983.
![Assignment of [Ti(CO 2)4]− Clusters [Ti(CO 2)4]− Experiment + 0 k. J mol− 1 Assignment of [Ti(CO 2)4]− Clusters [Ti(CO 2)4]− Experiment + 0 k. J mol− 1](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-10.jpg)
Assignment of [Ti(CO 2)4]− Clusters [Ti(CO 2)4]− Experiment + 0 k. J mol− 1 Simulation Metal Carbonyl Region + 38 k. J mol− 1 + 71 k. J mol− 1 10 B 3 LYP-disp/def 2 -tzvpp. ZPE-corrected. Ti (purple), C (black), O (red).
![Assignment of [Fe(CO 2)3]− Clusters [Fe(CO 2)3]− Experiment + 0 k. J mol− 1 Assignment of [Fe(CO 2)3]− Clusters [Fe(CO 2)3]− Experiment + 0 k. J mol− 1](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-11.jpg)
Assignment of [Fe(CO 2)3]− Clusters [Fe(CO 2)3]− Experiment + 0 k. J mol− 1 Simulation Metal Carbonyl Region + 85 k. J mol− 1 + 88 k. J mol− 1 Thompson, L. G. D. , Weber, J. Phys. Chem. A 2017, 121, 4132. 11 Fe (yellow), C (black), O (red).
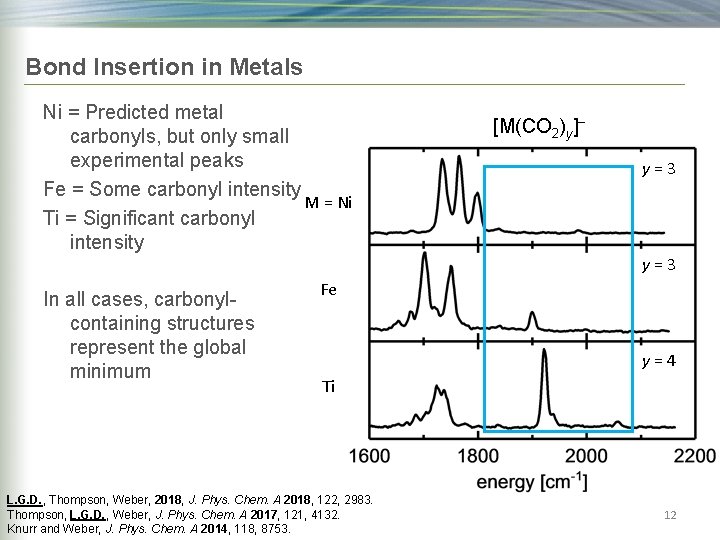
Bond Insertion in Metals Ni = Predicted metal carbonyls, but only small experimental peaks Fe = Some carbonyl intensity M = Ni Ti = Significant carbonyl intensity [M(CO 2)y]− y=3 In all cases, carbonylcontaining structures represent the global minimum Fe y=4 Ti L. G. D. , Thompson, Weber, 2018, J. Phys. Chem. A 2018, 122, 2983. Thompson, L. G. D. , Weber, J. Phys. Chem. A 2017, 121, 4132. Knurr and Weber, J. Phys. Chem. A 2014, 118, 8753. 12
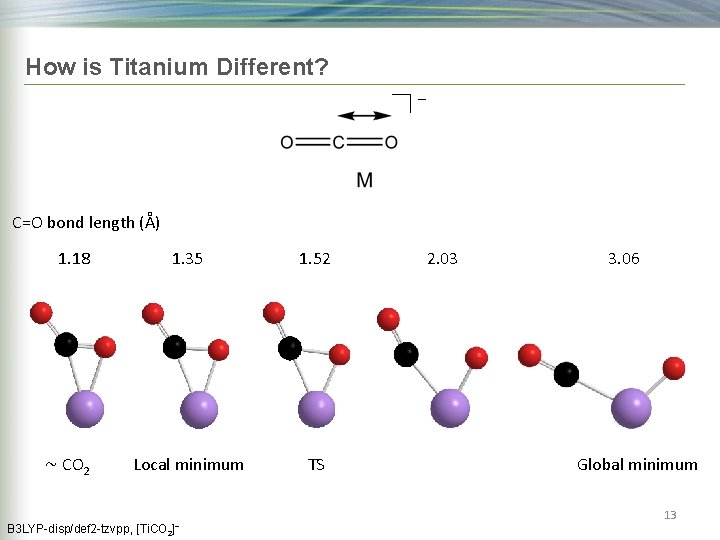
How is Titanium Different? − C=O bond length (Å) 1. 18 ~ CO 2 B 3 LYP-disp/def 2 -tzvpp, 1. 35 1. 52 Local minimum TS [Ti. CO 2]− 2. 03 3. 06 Global minimum 13
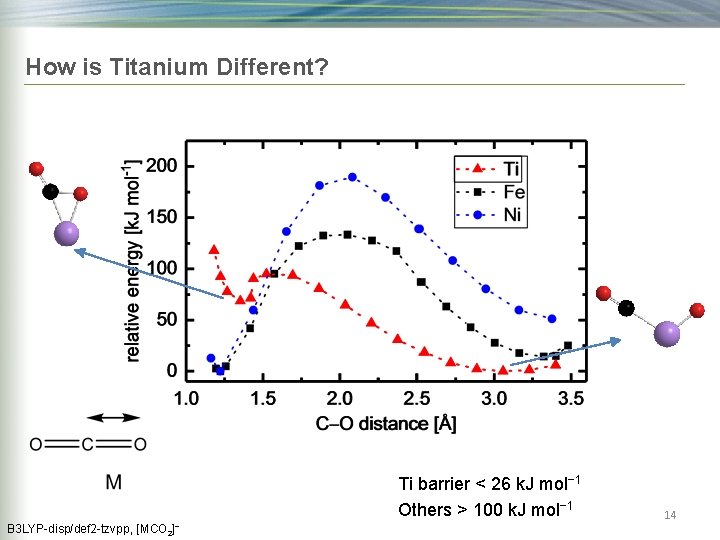
How is Titanium Different? Ti barrier < 26 k. J mol− 1 Others > 100 k. J mol− 1 B 3 LYP-disp/def 2 -tzvpp, [MCO 2]− 14
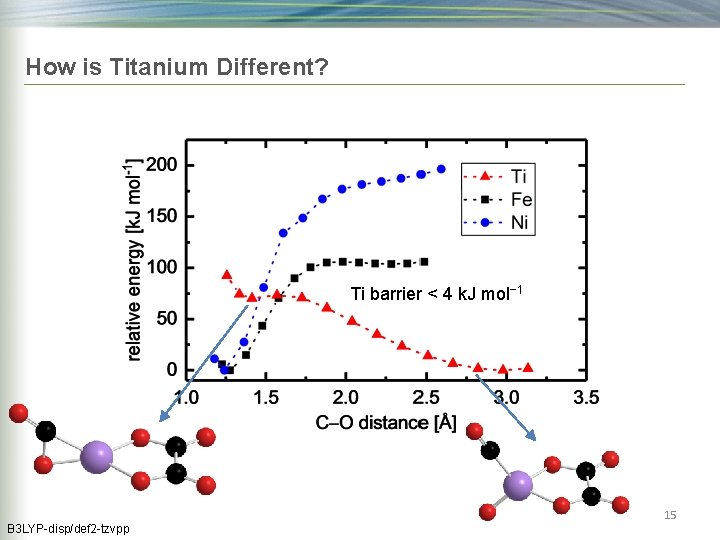
How is Titanium Different? Ti barrier < 4 k. J mol− 1 B 3 LYP-disp/def 2 -tzvpp 15

Metal Comparison Ni Oxophilicity (θ(M)): Ni: 0. 2 Fe Ti Fe: 0. 4 Ti: 1. 0 16 Oxophilicities quantified by: Kepp, Inorg. Chem. , 2016, 55, 9461.
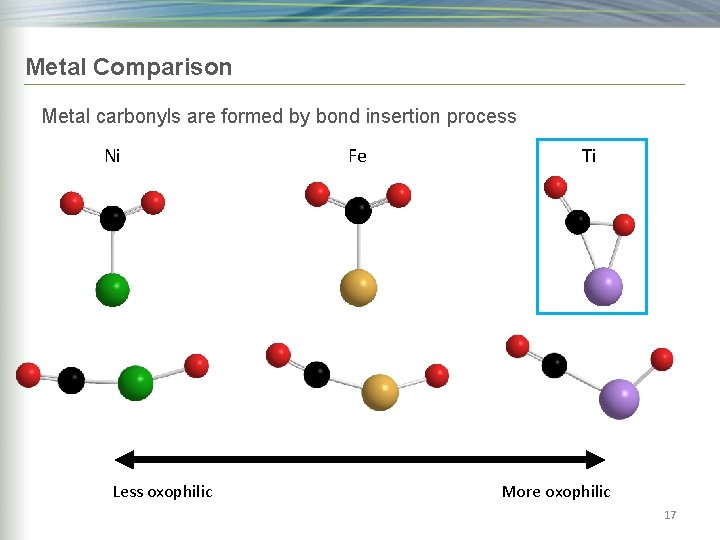
Metal Comparison Metal carbonyls are formed by bond insertion process Ni Less oxophilic Fe Ti More oxophilic 17
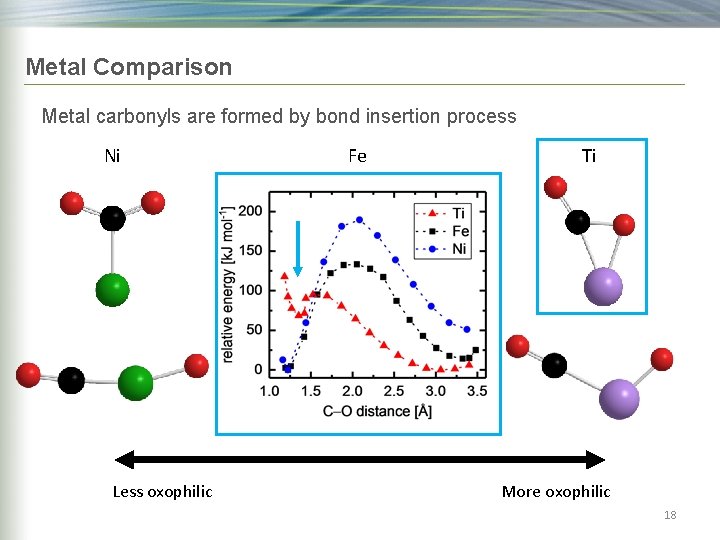
Metal Comparison Metal carbonyls are formed by bond insertion process Ni Less oxophilic Fe Ti More oxophilic 18
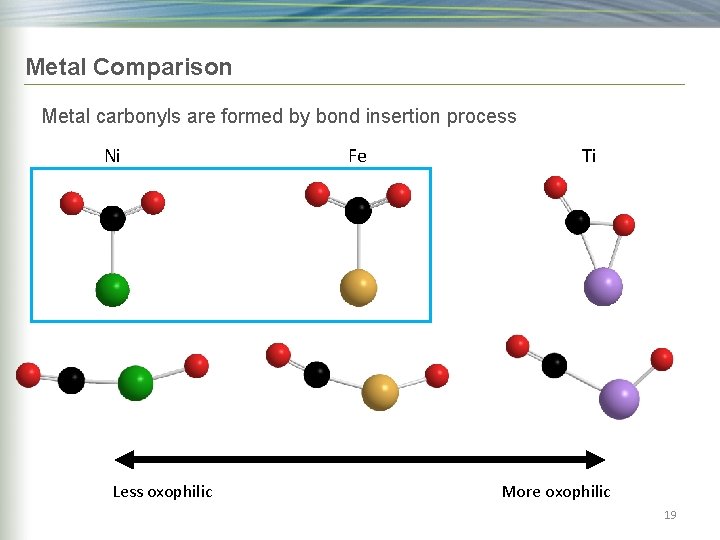
Metal Comparison Metal carbonyls are formed by bond insertion process Ni Less oxophilic Fe Ti More oxophilic 19

Metal Comparison Ni Oxophilicity: Mulliken-Mayer Bond Order: Fe Ti Ni: 0. 2 Fe: 0. 4 Ti: 1. 0 Ni–O: 1. 29 Fe–O: 1. 52 Ti–O: 2. 11 Oxophilicities quantified by: Kepp, Inorg. Chem. , 2016, 55, 9461. Bond orders calculated at B 3 LYP-disp/def 2 -TZVPP using a Natural Population Analysis. 20
![[M(CO 2)y]−: Oxophilicity Drives Bond Insertion [M(CO 2)y]− y=3 M = Ni y=3 Fe [M(CO 2)y]−: Oxophilicity Drives Bond Insertion [M(CO 2)y]− y=3 M = Ni y=3 Fe](http://slidetodoc.com/presentation_image_h2/8497e7e18f7053d3ba6ac32ac1cc8b07/image-21.jpg)
[M(CO 2)y]−: Oxophilicity Drives Bond Insertion [M(CO 2)y]− y=3 M = Ni y=3 Fe y=4 Ti L. G. D. , Thompson, Weber, J. Phys. Chem. A 2018, 122, 2983. Thompson, L. G. D. , Weber, J. Phys. Chem. A 2017, 121, 4132. Knurr and Weber, J. Phys. Chem. A 2014, 118, 8753. 21

Summary • Oxophilicity differences in metal atoms drive C=O bond insertion in CO 2 • Titanium has the highest oxophilicity • Observe significant metal carbonyl intensity in infrared experimental spectra • Low barriers found computationally for bond insertion • Iron and nickel both have lower oxophilicities • Little or no intensity for metal carbonyl signatures • Higher barriers to bond insertion • Oxalato ligands decrease the barrier to insertion for titanium • Oxalato ligands increase barrier for insertion in iron and nickel 22

Acknowledgements NIST NRC Research Associateship NSF AMO PFC 23

Acknowledgements Cold and Ultracold Molecules – Roger Adams Lab 116 RG 01 – Electronic photodissociation spectroscopy of cold nitrophenolate ions. Part I. Ortho- and para-nitrophenolate RG 02 – Electronic photodissociation spectroscopy of cold nitrophenolate ions. Part II. Meta-nitrophenolate NIST NRC Research Associateship NSF AMO PFC 24
- Slides: 24