Bond Energies Bond energy the energy required to

Bond Energies • Bond energy: the energy required to break a given chemical bond. • To break bonds, energy must be added to the system (endothermic). • To form bonds, energy must be released (exothermic).
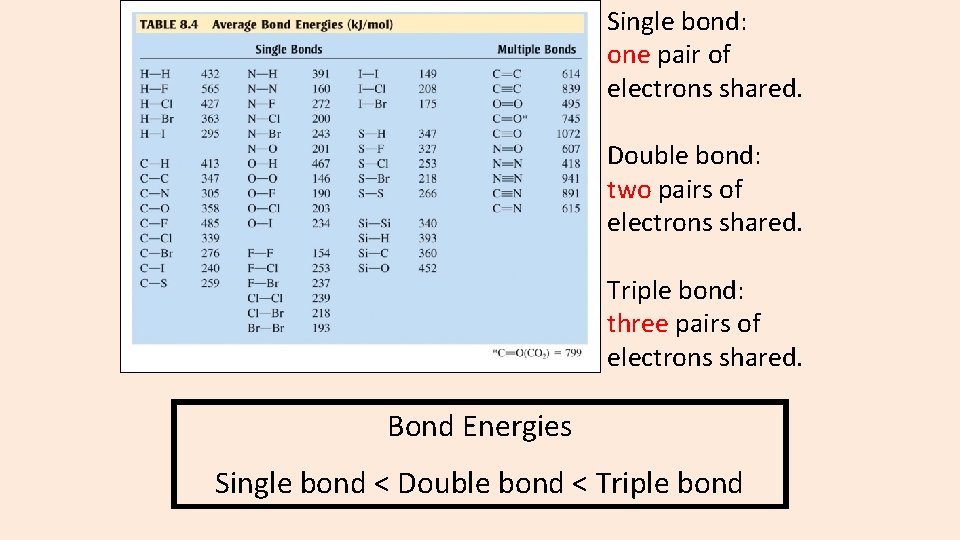
Single bond: one pair of electrons shared. Double bond: two pairs of electrons shared. Triple bond: three pairs of electrons shared. Bond Energies Single bond < Double bond < Triple bond
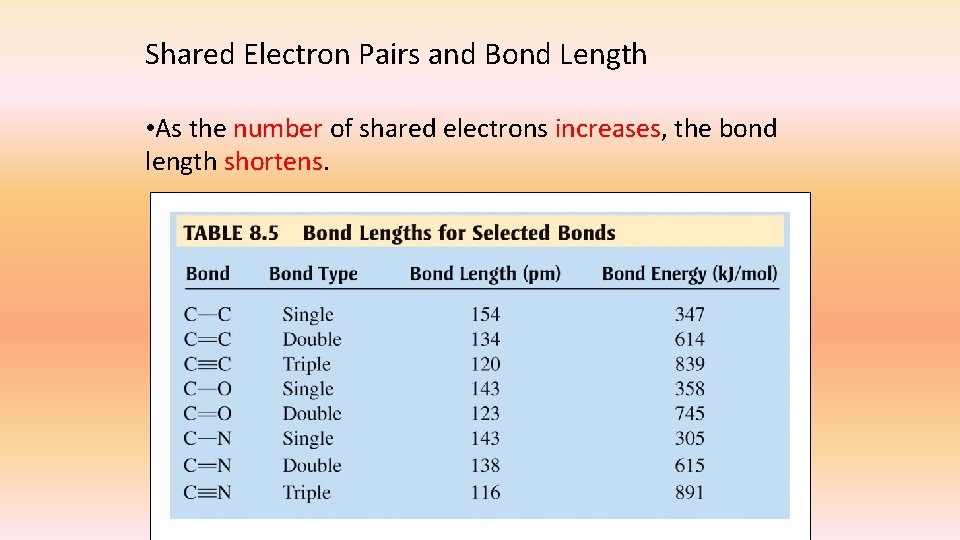
Shared Electron Pairs and Bond Length • As the number of shared electrons increases, the bond length shortens.

• Bond energy values can be used to calculate approximate energies for reactions. • Example: calculate the change in energy that accompanies the following reaction: H 2 (g) + F 2 (g) → 2 HF (g) • To form HF, one H-H bond and one F-F bond must be broken and two H-F bonds must be formed.

• Remember for bonds to be broken energy must be added to the system – an endothermic process – and carries a positive sign. • Formation of a bond releases energy – an exothermic process – and carries a negative sign. Enthalpy change: H = n×D(bonds broken) – n×D(bonds formed) where represents the sum of terms and D represents the bond energy per mole (n) of bonds. D always has a positive sign.

In the case of the formation of HF, ∆H = DH-H + DF-F – 2 DH-F = (1 mol x 432 k. J/mol) + (1 mol x 154 k. J/mol) - (2 mol x 565 k. J/mol) = -544 k. J Thus, when 1 mol H 2 (g) and 1 mol F 2 (g) react to form mol HF (g), 544 k. J of energy should be released. 2 When this result is compared to the result for the reaction when using the standard enthalpy of formation for HF (-542 k. J) the use of bond energies works well.
- Slides: 6