Bond Chemical Bond What is Chemical Bonding A
Bond … Chemical Bond

What is Chemical Bonding? ? ? ü A mutual electrical attraction between the nuclei and valence electrons of different atoms that binds the atoms together ü ok, so… ü it’s the attraction due to charges (+ and -) ü Forces that link atoms together ü Involves the valence electrons

So why do atoms bond? üTO BECOME MORE STABLE!!!!

Which electrons are involved in bonding? ü It’s the VALANCE ELECTRONS ü The outer most electrons ü Maybe they’re shared, ü Maybe they’re lost or gained ü These are the electrons you have to worry about!!!

Types of Chemical Bonds ü 1. Covalent Bond ü 2. Ionic Bond ü 3. Metallic Bond ü And… ü James Bond

What Rule Is It? ü Why is Ne, Ar, Kr, Xe, Rn so stable? ü Complete outer shell ü How many e- in the outermost shell ü 8 ü Bonding results because atoms want to be stable ~ they want to have a complete outer shell

And the rule is… ü Octet Rule ü chemical cmpds tend to form so that each atom, by gaining, losing or sharing e-, has an octet of e- in its highest E-level ü aka – YOU WANT 8 IN THE OUTER SHELL

ü Don’t Write This! ü There are some exceptions to the octet rule, however we will cover these when we get into writing Lewis Dot Structures.

1. Covalent Bonding ü The SHARING of electron pairs ü Valance electrons ü Bond between 2 or more non-metals resulting in a molecule ü Properties: low melting and boiling points, do not conduct electricity, typically liquids or gases.
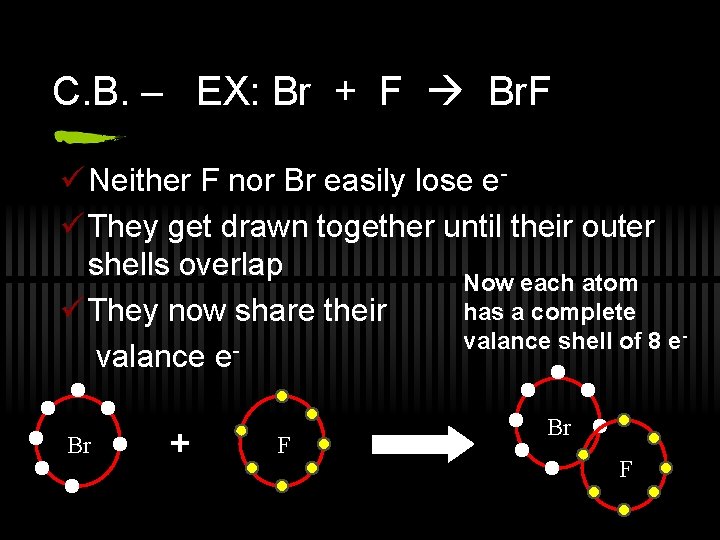
C. B. – EX: Br + F Br. F ü Neither F nor Br easily lose eü They get drawn together until their outer shells overlap Now each atom has a complete ü They now share their valance shell of 8 e valance e. Br + F Br F
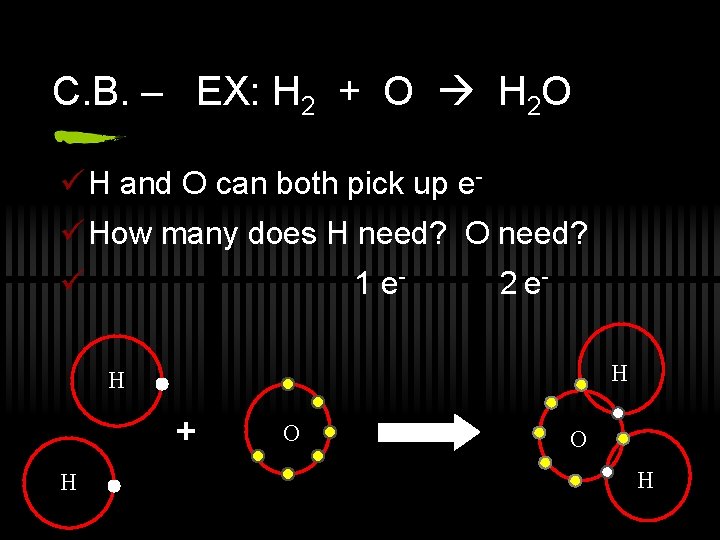
C. B. – EX: H 2 + O H 2 O ü H and O can both pick up eü How many does H need? O need? ü 1 e- 2 e. H H + H O O H

Multiple Bonds (double + triple) ü The past two examples showed a single covalent bond. ü This is not the only type ü There also double bonds and triple bonds ü How many e- are shared in a double? In a triple?
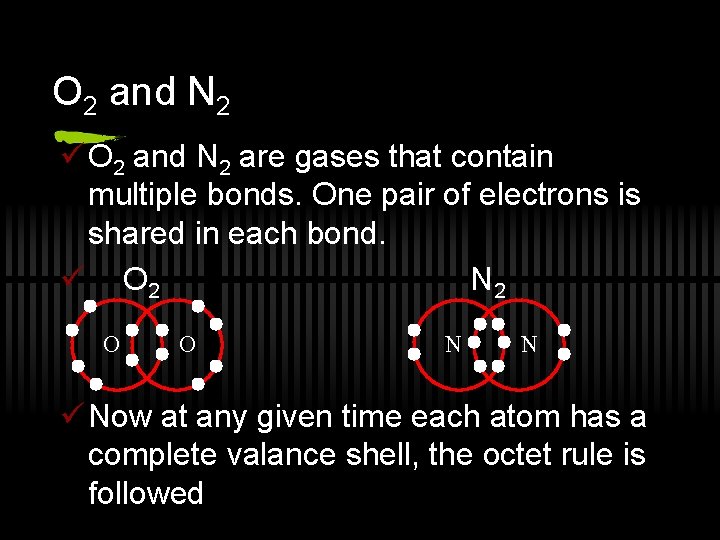
O 2 and N 2 ü O 2 and N 2 are gases that contain multiple bonds. One pair of electrons is shared in each bond. ü O 2 N 2 O O N N ü Now at any given time each atom has a complete valance shell, the octet rule is followed

Lewis Diagrams ü Lewis and Langmuir developed a way to represent molecules ü Pairs of dots used to represent valence electron pairs Irving Langmuir was the first to use the term “Covalent”

Lewis Dot Structures ü Step 1: The element symbol represents the nucleus and all e- except valence. ü Step 2: Determine the # of valence electrons. These e- are the ones with the largest principal quantum numbers in the electron configurations. ü Quick practice

Determine the nbr. of Valance e. Li Be B C N O F Ne ü Where are each of these elements located on the P. T. (pat attention to their group) 1 2 3 4 5 6 7 8 ü Remember what is the max. nbr. of valance ethat an atom can hold? ü Octet Rule

Lewis Dot Structures ü EX: carbon step 1: C step 2: 4 valance es step 3: pz C py px >One more note: when filling the orbitals around the symbol there an “s” and (3) “p” >So you fill them just like filling e- config.
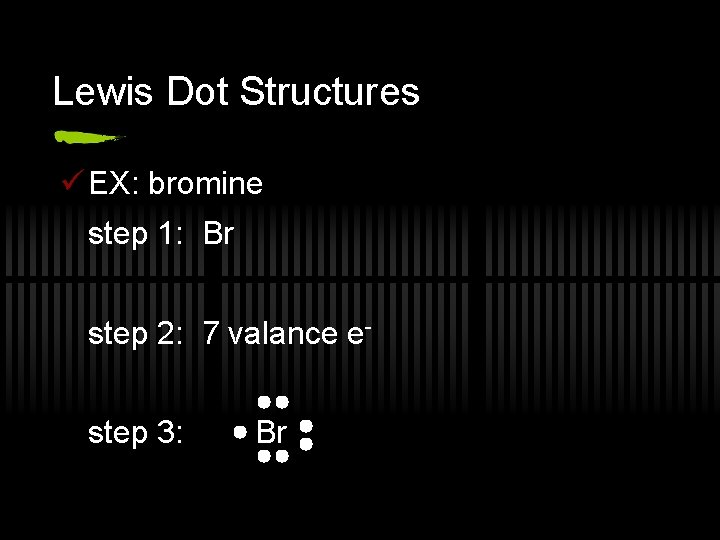
Lewis Dot Structures ü EX: bromine step 1: Br step 2: 7 valance estep 3: Br
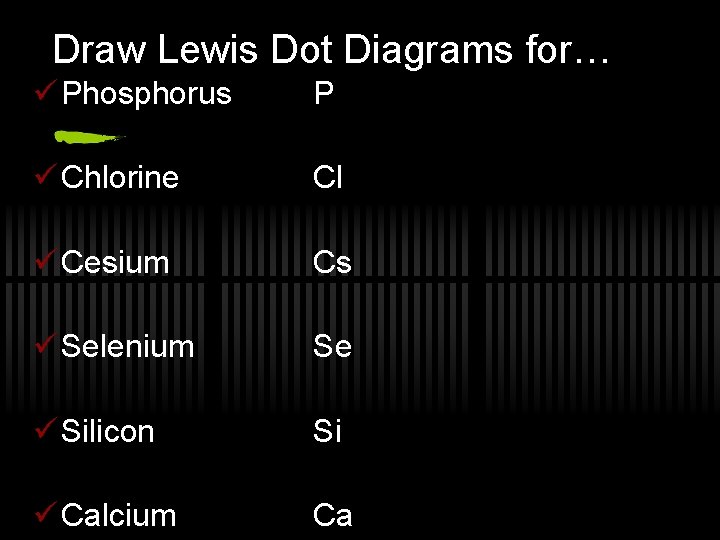
Draw Lewis Dot Diagrams for… ü Phosphorus P ü Chlorine Cl ü Cesium Cs ü Selenium Se ü Silicon Si ü Calcium Ca

Lewis Structures for molecules 1. Determine the total number of valence electrons in the molecule. SF 2 Total = 20 Valence e 2. Arrange atoms to form a skeleton structure. Atom that needs the most e- to complete their octet goes in the center. F S F

Lewis structures cont. 3. Use pairs of electrons (dots) to bond atoms. Put (2) dots in between each element, this represents a single bond. F S F
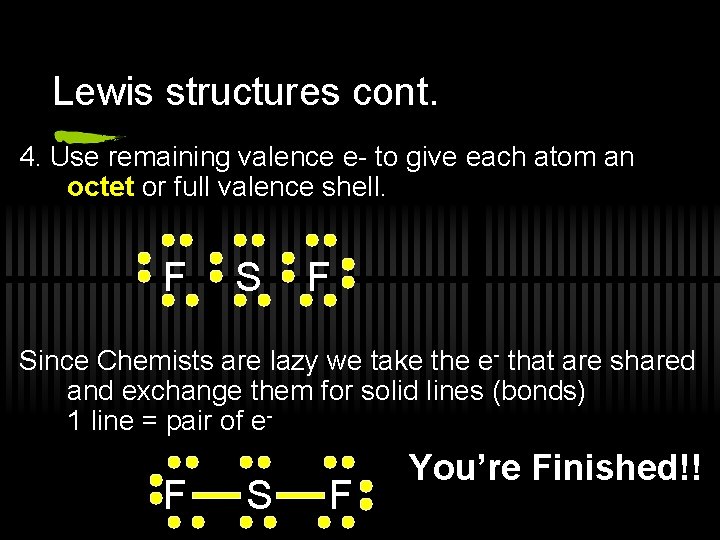
Lewis structures cont. 4. Use remaining valence e- to give each atom an octet or full valence shell. F Since Chemists are lazy we take the e- that are shared and exchange them for solid lines (bonds) 1 line = pair of e- F S F You’re Finished!!

Exceptions ü Now that you’ve mastered the Lewis dot diagrams and the octet rule you should know that there are exceptions to the octet rule ü They involve the atoms of H, B, etc.

Exceptions 1. If you have extra e- and all of the atoms have an octet, put the extra e- on the central atom in pairs. (If the central atom has an atomic number greater than fifteen, you are allowed to have more than eight e- around it). 2. Boron tends to be stable with only 6 e(rather than 8) in some compounds. Ex. BF 3 3. Hydrogen does not follow the octet rule, instead it follows the duet rule, it only needs 2 e- to satisfy its outer energy level
More examples ü HBr ü H 2 S ü CF 4 ü O 2 ü CO 2

Try the following examples… ü HF ü CH 4 ü Si. F 4 ü C 2 H 6

A little trick, that almost always works ü If you didn’t already notice certain atoms like to form specific bonds, for example… ü Carbon (and family) – likes to be a central atom and form 4 bonds ü Nitrogen (and family) – likes to form 3 bonds ü Oxygen (and family) – likes to form 2 bonds ü Halogens – like to form 1 bond

Ions ü These are very simple ü How do you form a cation ü Loose an electron ü This mean you will subtract an e- ü How do you form a anion ü Gain an electron ü This means you will add an e-

Lets try some ü How many e- in NO 3ü 24 ü How many e- in PO 33ü 26 ü How many e- in NH 4+ ü 8

How to draw them… ü Draw them just like a Lewis structure ü Add in brackets and the charge ü

Practice ü NH 4+ ü Cl. Oü CNü Si. O 32ü As. O 33 -

2. Ionic Bonding ü Transfer of an e- from a metal to a nonmetal resulting in oppositely charged ions ü Atoms lose an e- and become a… ü Cation ü Atoms gain an e- and become a… ü Anion ü Prop. of ionic cmpds: brittle, crystalline solids, good conductors of electricity when molten or dissolved in water, high melting and boiling points.
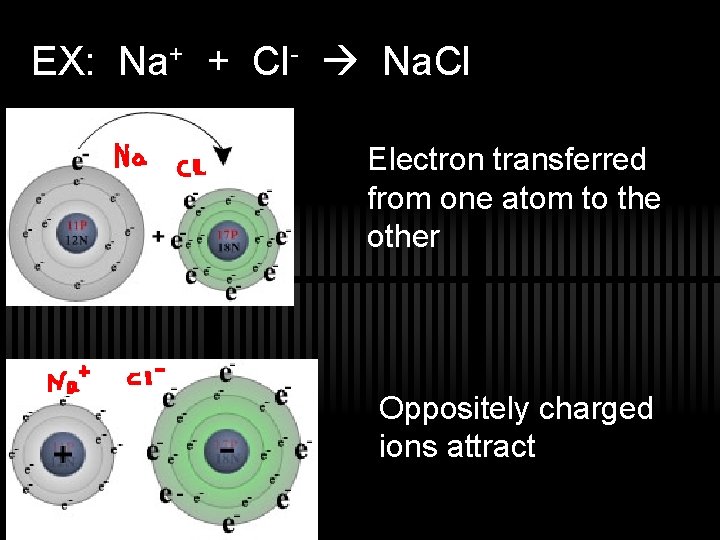
EX: Na+ + Cl- Na. Cl Electron transferred from one atom to the other Oppositely charged ions attract

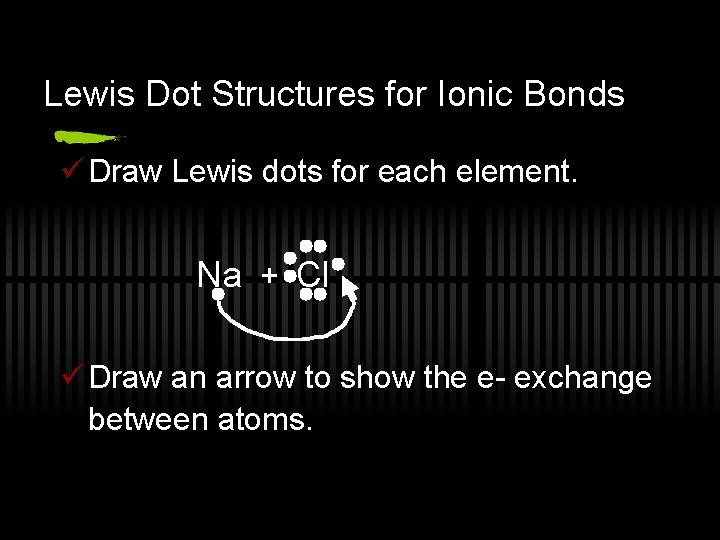
Lewis Dot Structures for Ionic Bonds ü Draw Lewis dots for each element. Na + Cl ü Draw an arrow to show the e- exchange between atoms.
![ü Now draw them together. [Na] [ Cl ] + - ü Ionic bonds ü Now draw them together. [Na] [ Cl ] + - ü Ionic bonds](http://slidetodoc.com/presentation_image_h/d40a885c9ea99171e34111a209cac742/image-36.jpg)
ü Now draw them together. [Na] [ Cl ] + - ü Ionic bonds form because of the attraction between the pos. and neg. charges.

What Type of Bond? ? ? ü How can you tell what bond will form ü Indicators ü Metal and nonmetal will form a ionic ü Usually (2)+ nonmetals will form a covalent ü However the one determining factor is that of Electronegativity ü Covalent can either be polar or non-polar

ü Before we talk about EN, what is this polar/non-polar thing? ü A molec. can have an overall charge ü We can use EN to tell if a covalent bond is either polar or non-polar ü We can also figure out in which part of the molec. is pos. and which part is neg. ü Remember that electronegativity is the ability of an atom to attract an e- when bonded to another atom.

Bond Type – told by diff. in EN I >1. 7 = IONIC N C 0. 3 -1. 7 = POLAR COVALENT E N <0. 3 = NON-POLAR COVALENT

Bond Type üM – O ü |3. 50 – 1. 23| = 2. 27 IONIC üB – P ü |2. 01 – 2. 06| = 0. 05 NON-POLAR COVALENT ü Mg – N ü |1. 23 – 3. 07| = 1. 84 IONIC ü C – Na ü |2. 50 – 1. 01| =1. 49 POLAR COVALENT

3. Metallic Bond ü Metals bonding with other metals DO NOT gain, lose e- or share e-. ü Bonds are created from the delocalized ethat hold metallic atoms together. ü Electrons flow like “a sea of charge”. Properties: good conductors of electricity and heat, malleable and ductile, “shiny” reflective appearance

4. James Bond
- Slides: 42