BOMB CALORIMETRY Heat Internal Energy and Enthalpy Heat

BOMB CALORIMETRY

Heat, Internal Energy, and Enthalpy Heat (q) is the energy transferred between two systems initially at different temperatures when placed in thermal contact. From the first law of thermodynamics U = q + w where w = work and U = internal energy. For a system where only mechanical (pressure-volume) work occurs, and under conditions where V = constant, we can show U = q. V If we define enthalpy (H) as H = U + p. V we can then show that under conditions where p = constant H = qp

Significance of U = q. V and H = qp There at least two important reasons why the relationship between q and either U or H is significant. 1) There are standard methods for measuring q for processes carried out in the laboratory (calorimetry). 2) The relationship between q and either U (for constant volume processes) or H (for constant pressure processes) relates q to a change in a state function. Since changes in state functions are path independent, this allows us to tabulate thermochemical data for pure chemical substances.

Formation Reaction One of the most important general types of reactions in chemistry is the formation reaction. By definition, the formation reaction is the chemical reaction that forms one mole of a single product from elements in their standard (most stable) state. Standard state for elements (at T= 25. C) Gases – noble gases (He, Ne, Ar, Kr, Xe, Rn) diatomic gases (H 2, N 2, O 2, F 2, Cl 2) Liquids – Br 2, Hg All other elements exist as solids in their standard state.

Formation Reaction (Examples) Consider the formation reactions for the following pure chemical substances – CH 4(g), C 6 H 5 Cl( ), H 2 O( ), and H 2 O(g) CH 4(g) C(s) + 2 H 2(g) CH 4(g) C 6 H 5 Cl( ) 6 C(s) + 5/2 H 2(g) + ½ Cl 2(g) C 6 H 5 Cl( ) H 2 O(g) H 2(g) + ½ O 2(g) H 2 O(g) Note that in some cases fractional coefficients must be used for one or more of the reactants to write the correctly balanced formation reaction. Also note that the formation reaction depends on both the identity and state of the product being formed.

Enthalpy of Formation ( H f) By definition, the enthalpy of formation ( H f) for a pure chemical substance (for standard conditions) is the change in enthalpy when one mole of the formation reaction is carried out under conditions of constant pressure. For example, H f(CH 4(g)) is the change in enthalpy when one mole of the following reaction is carried out: C(s) + 2 H 2(g) CH 4(g) Note that when the above reaction takes place under conditions of constant pressure, H f(CH 4(g)) = qp. Since qp can be measured experimentally this gives a laboratory method for finding H f(CH 4(g)). Values for the enthalpy of formation for other substances can also be found less directly using other reactions and Hess’ Law.

Usefulness of H f The usefulness of knowing values for H f is related to the fact that enthalpy is a state function. Because of this, there is a general formula for finding the enthalpy change for any chemical reaction for standard conditions. H rxn = [ H f(products) ] - [ H f(reactants) ] For example, for the reaction 4 Fe 3 O 4(s) + O 2(g) 6 Fe 2 O 3(s) the change in enthalpy per mole of reaction is H rxn = [6 H f(Fe 2 O 3(s))] - [4 H f(Fe 3 O 4(s)) + H f(O 2(g)) ] = [6 (- 824. 2 k. J/mol)] - [4 (- 1118. 4 k. J/mol) + 0. 00] = - 471. 6. k. J/mol

Combustion Reaction Another important general type of reaction is the combustion reaction. By definition, the combustion reaction is the chemical reaction that reacts one mole of a single substance with molecular oxygen (O 2(g)) to form combustion products. Combustion products are defined as follows. C CO 2(g) H H 2 O( ) Combustion products are sometimes also defined for other elements. N N 2(g) Cl 2(g) Br 2( ) Note that unlike the formation reaction, the combustion reaction is not defined for all pure chemical substances.

Combustion Reaction (Examples) Consider the combustion reaction for the following pure chemical substances – CH 4(g), C 6 H 5 COOH(s). CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O( ) C 6 H 5 COOH(s) + 15/2 O 2(g) 7 CO 2(g) + 3 H 2 O( ) Note that as is the case formation reactions, we sometimes must use fractional coefficients to write a correctly balanced combustion reaction. The enthalpy change when one mole of a combustion reaction is carried out under standard conditions is written as H c, and is called the enthalpy of combustion. For example H c(CH 4(g)) is the change in enthalpy when one mole of the combustion reaction for methane is carried out under standard conditions.

Usefulness Of H c The usefulness of knowing values for H c is related to the fact that enthalpy is a state function. Because of this, the enthalpy of combustion for a substance can be related to the enthalpy of formation for the substance. For example, for the combustion reaction for methane CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O( ) H c = [ H f(CO 2(g)) + 2 H f(H 2 O( ) ] - [ H f(CH 4(g)) + 2 H f(O 2(g)) ] Since precise values for H f(CO 2(g)) and H f(H 2 O( )) are known (and H f(O 2(g)) = 0. 00 k. J/mol), an experimental measurement of H c(CH 4(g)) makes it possible to determine H f(CH 4(g)).

Bomb Calorimetry One experimental method for determining the enthalpy of combustion for a solid compound in bomb calorimetry. In bomb calorimetry a sample of a pure solid compound is burned in excess oxygen to form combustion products. Because high pressures of O 2 are used, the combustion takes place inside of a bomb, a thick walled metal container. Because of this, bomb calorimetry is a constant volume process, and so bomb calorimetry measures a value for U c, the energy of combustion. This can be used to find H c, the enthalpy of combustion, as discussed below.

Bomb Parts In bomb calorimetry the combustion reaction is carried out in a bomb, a thick walled metal container. Inside of the bomb there is a cup for holding a compressed solid pellet of the compound being burned. After a fuse wire is connected between the two electrodes of the bomb and in contact with the pellet, the bomb is assembled and then pressurized to ~ 20 atm of oxygen.

Assembling the Calorimeter The bomb where the combustion reaction takes place is placed inside of a bucket containing an exact amount of water. The bucket, containing the bomb, is then placed inside of an insulated container. A propeller is used to stir the water to obtain a uniform temperature, and a thermometer is used to measure temperature.

Experimental Procedure 1) Compress ~ 1 gram of the compound to be burned into a pellet. Measure the mass of the pellet, and also the initial mass of the fuse wire used to initiate combustion. 2) Assemble the bomb. 3) Carefully pressurize the bomb to ~ 20 atm O 2. 4) Place the assembled bomb into the metal bucket. Fill the bucket with exactly 2. 000 L of water (at a temperature near room temperature). 5) Place the bomb and bucket inside the insulated calorimeter housing. Attach the electrode connections, and then replace the top of the insulated container. Attach the drive belt for the stirrer, and begin stirring the water inside the bucket.

Experimental Procedure (continued) 6) Begin taking temperature measurements at 30 s intervals. Measure temperature to the nearest 0. 01 C. 7) After at least four measurements of temperature where the temperature is either constant or only changing by a small amount, detonate the bomb. If combustion has taken place, the temperature should begin to increase after ~ 20 s. Continue taking temperature measurements at 30 s intervals until a final stable temperature has been reached (four consecutive measurements where the temperature is approximately constant). 8) Release the pressure and disassemble the bomb. Check to make sure complete combustion has occurred. Find the mass of any unburnt fuse wire to determine the final mass of wire.
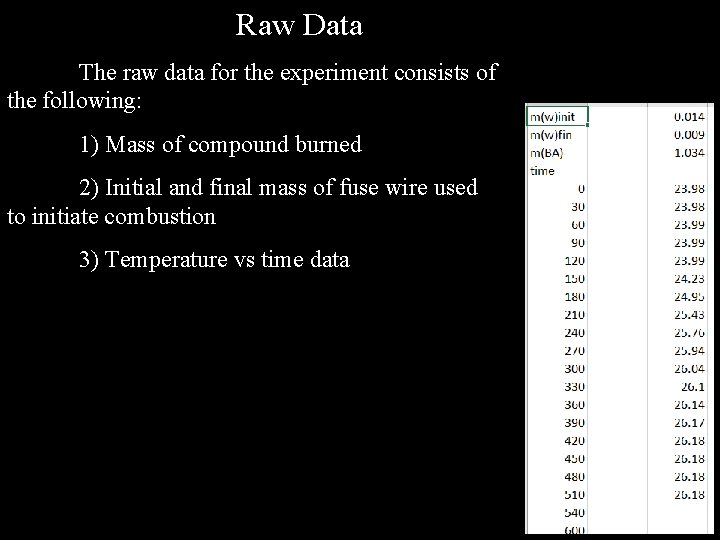
Raw Data The raw data for the experiment consists of the following: 1) Mass of compound burned 2) Initial and final mass of fuse wire used to initiate combustion 3) Temperature vs time data
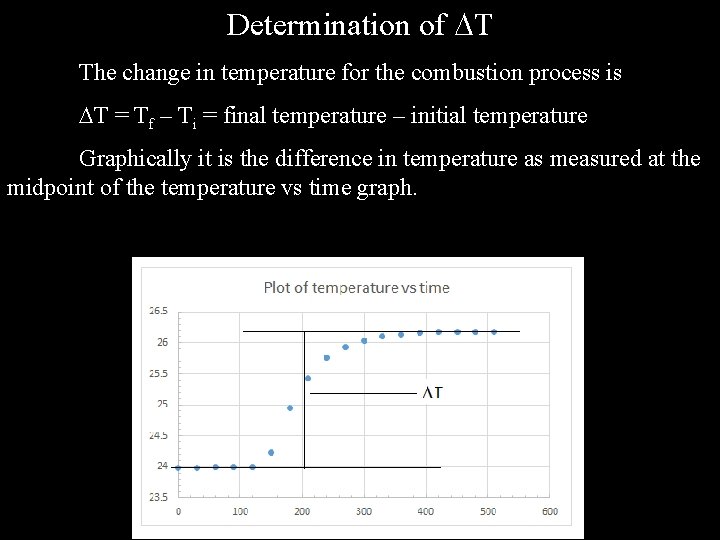
Determination of T The change in temperature for the combustion process is T = Tf – Ti = final temperature – initial temperature Graphically it is the difference in temperature as measured at the midpoint of the temperature vs time graph.

Determination of T (continued) While it is possible to find the value for T graphically, it is uaually more accurate (when you have a stable initial and final temperature) to find the value by examination of the experimental data. For example, for the sample data I get Tf = 26. 18 C Ti = 23. 99 C T = 26. 18 C – 23. 99 C = 2. 19 C

Determination of CB (heat capacity of calorimeter) There are two equations that apply for the combustion of benzoic acid. q = m. BA Um, c(BA) + mw Um, w(wire) where m. BA = mass benzoic acid Um, c(BA) = energy of combustion of benzoic acid = - 26410. J/g mw = mass of wire burned = mfin – minit Um, c(wire) = energy of combustion of wire = - 5850. J/g However, q is also given by the relationship q = - CB T The minus sign is because we measure the temperature change for the water (surroundings) and qsyst = - qsurr.

Determination of CB (continued) Since we know Um, c(BA) Um, c(wire) m. BA, mw, and since we measure T, the two equations on the previous page are sufficient to find the value for CB. For the present example, using the first equation for q gives q = (1. 034 g)( - 26410. J/g) + (0. 005 g)( - 5850. J/g) = - 27337. J From the second equation q = - CB T CB = - q = - (- 27337. J) = 12483. J/ C T (2. 19 C) NOTE – This is not the value for CB for our calorimeter, since the data being analyzed was made up to be used as an example.

Determination of UN The same two equations that apply for the combustion of benzoic acid also apply to the combustion of naphthalene. q = m. N Um, c(N) + mw Uc, w where m. N = mass naphthalene Um, N(N) = energy of combustion of naphthalene and all other terms are as previously defined, and q = - CB T Since we can assume that CB is the same for the combustion of naphthalene and benzoic acid, we have enough information to determine an experimental value for Um, c(N) from combustion data for naphthalene.

Determination of H c(N) To convert the experimental value of Um, c(N), the energy of combustion of naphthalene on a per mass basis, to H c(N), the molar enthalpy of combustion of naphthalene, two further steps are required. 1) To convert Um, c(N) to U c(N), the molar energy of combustion for naphthalene, we must multiply Um, c(N) by the molecular mass of naphthalene U c(N) = MW(N) Um, c(N) where MW(N) = molecular mass of naphthalene = 128. 17 g/mol

Determination of H c(N) (continued) 2) To convert U c(N) to H c(N), we use the following approximate relationship H c(N) U c(N) + ng. RT where ng = change in moles of gas per mole of combustion. For example, for the combustion of benzoic acid C 6 H 5 COOH(s) + 15/2 O 2(g) 7 CO 2(g) + 3 H 2 O( ) ng = 7 – 15/2 = - ½ To find the corresponding value for ng for naphthalene you will need to use the correctly balanced combustion reaction. Since the term ng. RT is much smaller than U c(N) (less than 1% in magnitude) you can choose T = 25. C = 298. K in calculating ng. RT without introducing significant error in your final result.

Lab Report Your lab report should include the following: 1) The determination of CB from the benzoic acid data. 2) The determination of Um, c(N) from the naphthalene data and the value for CB. 3) The conversion of Um, c(N) to H c(N). This will require that you give the correctly balanced combustion reaction for naphthalene. 4) A comparison of your value for H c(N) to a literature value. 5) A discussion of the major sources of random and systematic error in the experiment, their estimated magnitude, and their expected effect on the results of your experiment.
- Slides: 24