Bomb Calorimetry Constant Volume Calorimetry Reaction carried out

Bomb Calorimetry (Constant Volume Calorimetry) • Reaction carried out under constant volume. • Use a bomb calorimeter. • Usually study combustion.

Example A 0. 5269 g sample of octane is placed in a bomb calorimeter with a heat capacity of 11. 3 k. J °C-1. The original temperature of the water in the calorimeter is 21. 93°C. After combustion, the temperature increases to 24. 21°C. Determine the enthalpy of combustion per mole and per gram of octane.

Example A 0. 5269 g sample of octane is placed in a bomb calorimeter with a heat capacity of 11. 3 k. J °C-1. The original temperature of the water in the calorimeter is 21. 93°C. After combustion, the temperature increases to 24. 21°C. Determine the enthalpy of combustion per mole and per gram of octane. -48. 9 k. J g-1 -5590 k. J mol-1

• Hess’s law: if a reaction is carried out in a number of steps, H for the overall reaction is the sum of H for each individual step. • For example: CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) H = -802 k. J 2 H 2 O(g) 2 H 2 O(l) H = -88 k. J CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) H = -890 k. J
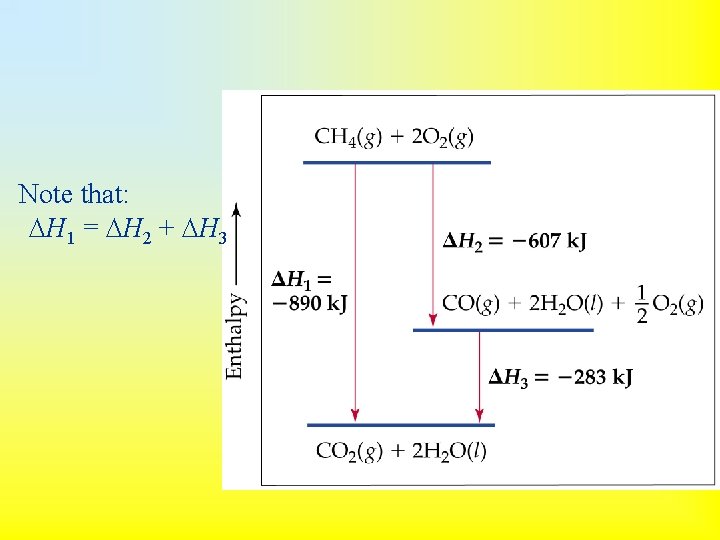
Note that: H 1 = H 2 + H 3

Example Given: Fe 2 O 3 (s) + 3 CO (g) 2 Fe (s) + 3 CO 2 (g) ΔH = -23 k. J 3 Fe 2 O 3 (s) + CO (g) 2 Fe 3 O 4 (s) + CO 2 (g) ΔH = -39 k. J Fe 3 O 4 (s) + CO (g) 3 Fe. O (s) + CO 2 (g) What is ΔH for the following? Fe. O (s) + CO (g) Fe (s) + CO 2 (g) ΔH =+18 k. J

Example Given: Fe 2 O 3 (s) + 3 CO (g) 2 Fe (s) + 3 CO 2 (g) ΔH = -23 k. J 3 Fe 2 O 3 (s) + CO (g) 2 Fe 3 O 4 (s) + CO 2 (g) ΔH = -39 k. J Fe 3 O 4 (s) + CO (g) 3 Fe. O (s) + CO 2 (g) What is ΔH for the following? Fe. O (s) + CO (g) Fe (s) + CO 2 (g) -11 k. J ΔH =+18 k. J

Example The bombardier beetle uses an explosive discharge as a defensive measure. The chemical reaction involved is the oxidation of hydroquinone by hydrogen peroxide to produce quinone and water: C 6 H 4(OH)2 (aq) + H 2 O 2 (aq) C 6 H 4 O 2 (aq) + 2 H 2 O (l)

C 6 H 4(OH)2 (aq) C 6 H 4 O 2 (aq) + H 2 (g) ΔH = +177. 4 k. J H 2 (g) + O 2 (g) H 2 O 2 (aq) ΔH = -191. 2 k. J H 2 (g) + ½ O 2 (g) H 2 O (g) ΔH = -241. 8 k. J H 2 O (g) H 2 O (l) ΔH = -43. 8 k. J C 6 H 4(OH)2 (aq) + H 2 O 2 (aq) C 6 H 4 O 2 (aq) + 2 H 2 O (l) ΔH = ? ? ?

C 6 H 4(OH)2 (aq) C 6 H 4 O 2 (aq) + H 2 (g) ΔH = +177. 4 k. J H 2 (g) + O 2 (g) H 2 O 2 (aq) ΔH = -191. 2 k. J H 2 (g) + ½ O 2 (g) H 2 O (g) ΔH = -241. 8 k. J H 2 O (g) H 2 O (l) ΔH = -43. 8 k. J C 6 H 4(OH)2 (aq) + H 2 O 2 (aq) C 6 H 4 O 2 (aq) + 2 H 2 O (l) ΔH = ? ? ? -202. 6 k. J

• If 1 mol of compound is formed from its constituent elements, then the enthalpy change for the reaction is called the enthalpy of formation, Hof. • Standard conditions (standard state): 1 atm and 25 o. C (298 K). • Standard enthalpy, Ho, is the enthalpy measured when everything is in its standard state. • Standard enthalpy of formation: 1 mol of compound is formed from substances in their standard states. • If there is more than one state for a substance under standard conditions, the more stable one is used. • Standard enthalpy of formation of the most stable form of an element is zero.

Examples – write formation reactions for each: 1. H 2 SO 4 2. C 2 H 5 OH 3. NH 4 NO 3

Examples – write formation reactions for each: 1. H 2 SO 4 H 2 (g) + S (s) + 2 O 2 (g) → H 2 SO 4 2. C 2 H 5 OH 2 C (s) + 3 H 2 (g) + 1/2 O 2 (g) → C 2 H 5 OH 3. NH 4 NO 3 N 2 (g) + 2 H 2 (g) + 3/2 O 2 (g) → NH 4 NO 3
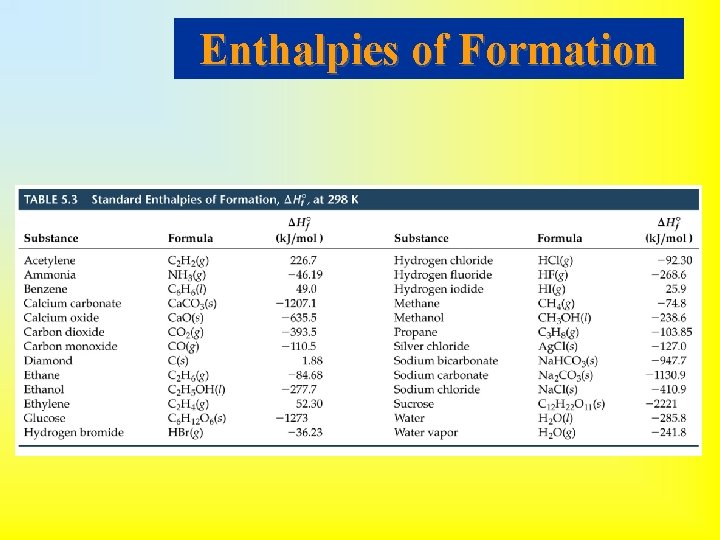
Enthalpies of Formation

Using Enthalpies of Formation of Calculate Enthalpies of Reaction • For a reaction

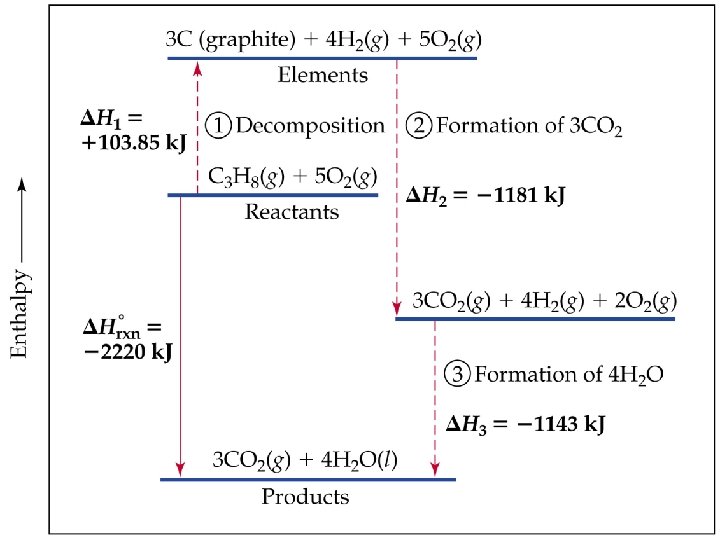
Examples Use the tables in the back of your book to calculate ΔH for the following reactions: 1. 4 Cu. O (s) 2 Cu 2 O (s) + O 2 (g) 2. C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (l) 3. NH 3 (g) + HCl (g) NH 4 Cl (s)

Examples Use the tables in the back of your book to calculate ΔH for the following reactions: 1. 4 Cu. O (s) 2 Cu 2 O (s) + O 2 (g) 283 k. J 2. 2. C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (l) -2220 k. J 3. NH 3 (g) + HCl (g) NH 4 Cl (s) -175. 9 k. J

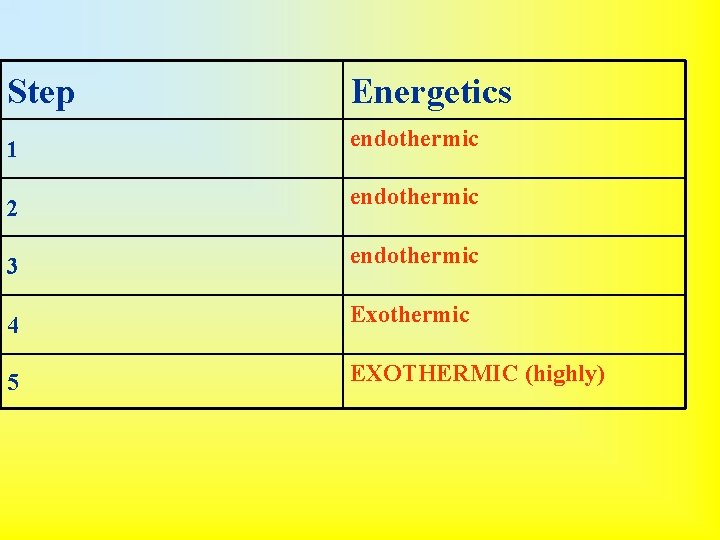
Born-Haber Cycle Application of Hess’s Law 1. Vaporize the metal (enthalpy of vaporization) • Na (s) Na (g) 2. Break diatomic nonmetal molecules (if applicable) (bond enthalpy) • ½ Cl 2 (g) Cl- 3. Remove electron(s) from metal (ionization energy) • Na (g) Na+ (g) + e- 4. Add electron(s) to nonmetal (electron affinity) • Cl (g) + e- Cl- (g) 5. Put ions together to form compound (lattice energy) • Na+ + Cl- Na. Cl (s)

• Overall Reaction: – Na (s) + ½ Cl 2 (g) Na. Cl (s) • This is useful because all quantities are directly measurable except lattice energy. The Born-Haber cycle can be used to calculate lattice energy from the other values.
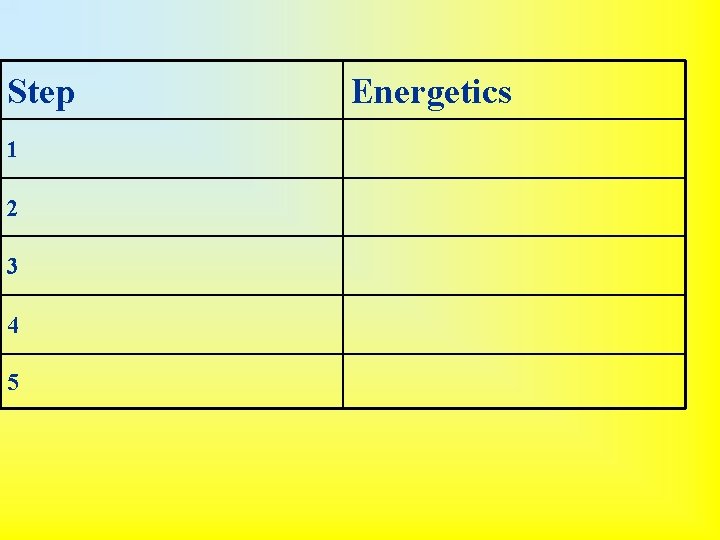
Step 1 2 3 4 5 Energetics
Step Energetics 1 endothermic 2 endothermic 3 endothermic 4 Exothermic 5 EXOTHERMIC (highly)
- Slides: 22