Boiler water treatment Rajan Bhandari Question Severe pitting

Boiler water treatment Rajan Bhandari

Question • • Severe pitting has been detected in way of the tube to tube plate connection on a waste heat boiler. State the reasons for such pitting State, with reasons, the action to be taken in order to enable safe operation of the boiler to continue. Explain how the incidence of future pitting can be minimised

Corrosion & Scale • Boilers • Feed System • Condensate system
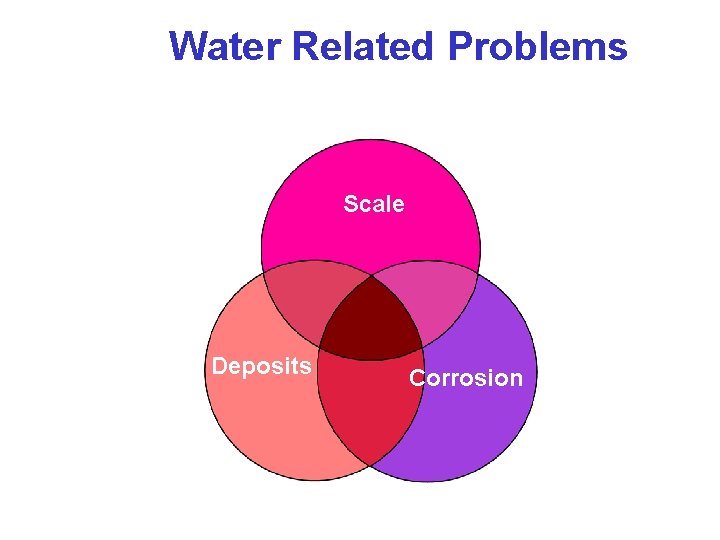
Water Related Problems Scale Deposits Corrosion
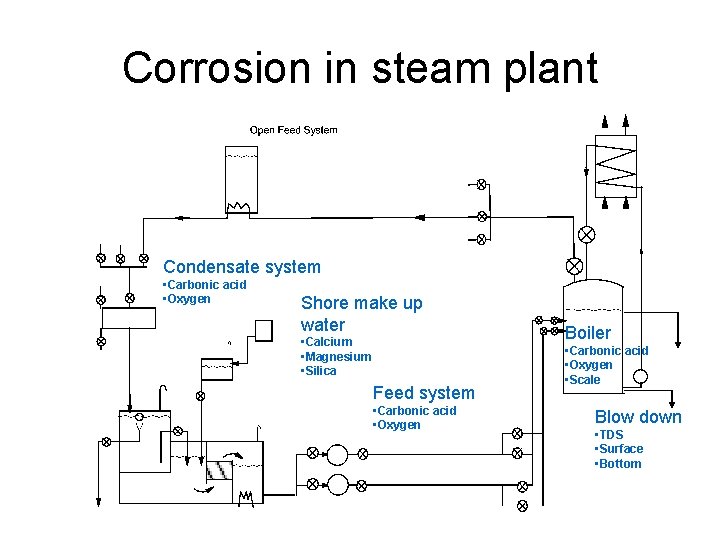
Corrosion in steam plant Condensate system • Carbonic acid • Oxygen Shore make up water • Calcium • Magnesium • Silica Feed system • Carbonic acid • Oxygen Boiler • Carbonic acid • Oxygen • Scale Blow down • TDS • Surface • Bottom

Boiler • Dissolved Oxygen: --Causing localized pitting • Scale: Mineral or chemical deposits)--Can result in underdeposit corrosion • Deposits Pitting
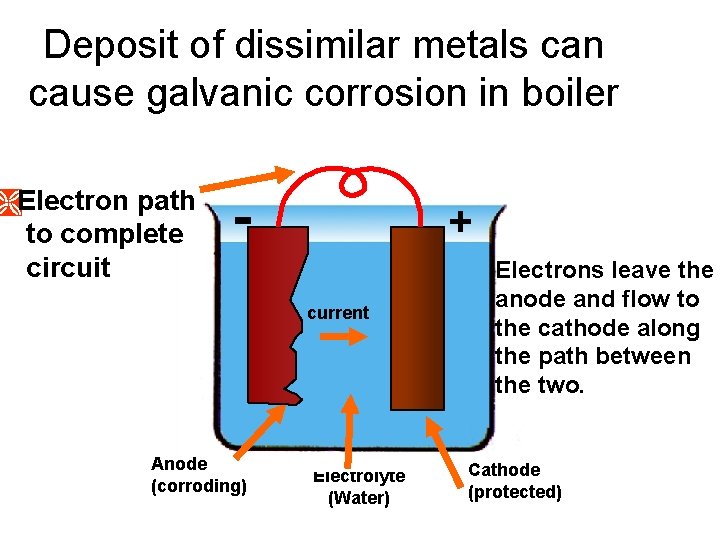
Deposit of dissimilar metals can cause galvanic corrosion in boiler ÌElectron path to complete circuit - + current Anode (corroding) Electrolyte (Water) Electrons leave the anode and flow to the cathode along the path between the two. Cathode (protected)
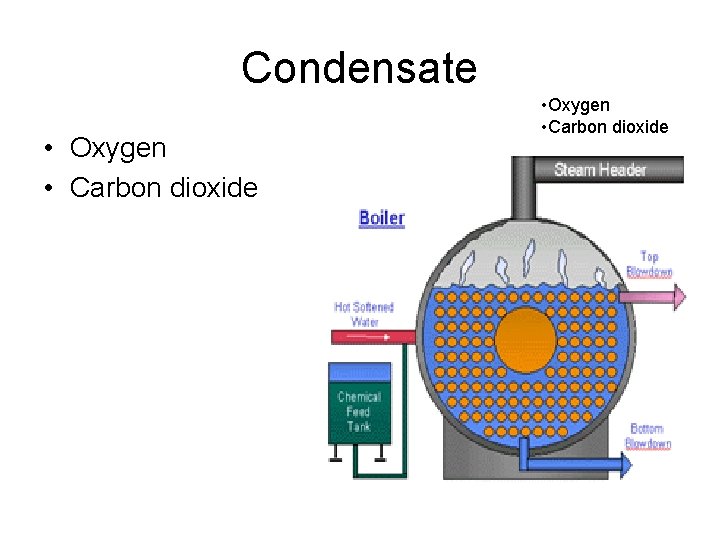
Condensate • Oxygen • Carbon dioxide

Results of Low Condensate p. H Condensate System Corrosion Grooving Carbonic Acid Attack

Results of Low Condensate p. H Condensate System Corrosion Notice Thinning Of The Tube

Under Deposit Corrosion Oxygen starved area covered by deposit is anodic High oxygen area is cathodic u Local Corrosion äPotential differences between areas on the same metal äResults from variation in oxygen content

Under Deposit Corrosion Dissolving The Metal From The Inside
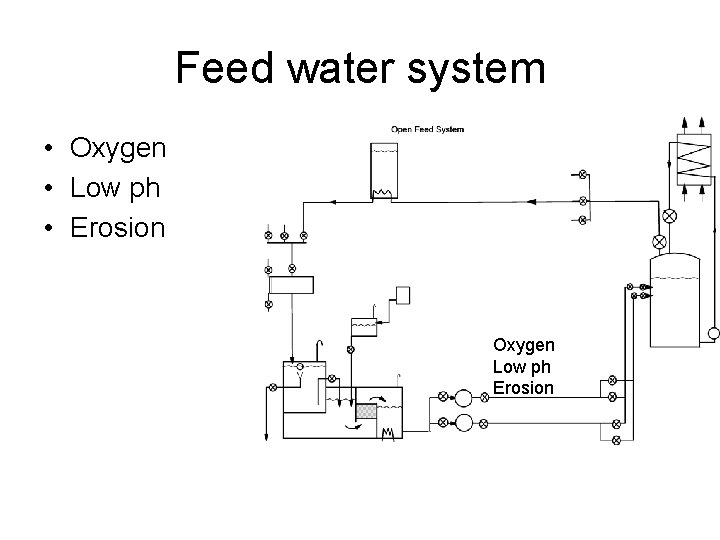
Feed water system • Oxygen • Low ph • Erosion Oxygen Low ph Erosion

Oxygen Corrosion Control u Consider Where Oxygen May Enter The System u Provide Optimum Effectiveness of Oxygen Scavenger

Common Source Of Oxygen is Feedwater u Deaerators remove most oxygen by thermalmechanical means u Efficient deaerators reduce oxygen to < 0. 007 ppm u Use chemical oxygen scavenger to remove remaining oxygen

Air In-Leakage can be a Source of Oxygen u u Piping threads Packing glands Tank vents Low pressure areas ä suction side of feedwater pump ä cross-over between high and low pressure stages of turbines in high pressure systems ä condenser ¾ particularly when there is low-load
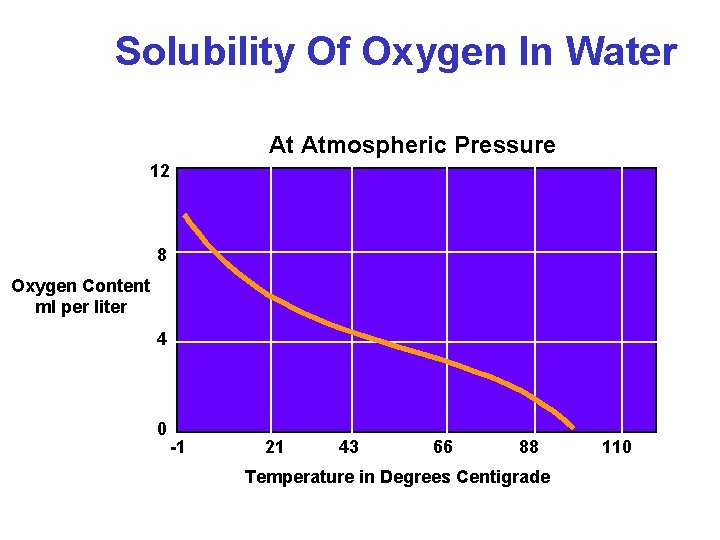
Solubility Of Oxygen In Water At Atmospheric Pressure 12 8 Oxygen Content ml per liter 4 0 -1 21 43 66 88 Temperature in Degrees Centigrade 110

Systems Without Deaeration u. Heat feedwater to 90ºC u. Dose chemical oxygen scavenger u. Sodium sulphite u. Hydrazine u. DEHA

Sodium Sulfite Advantages u u u Disadvantages Excellent oxygen scavenger Easy test for residual Economical u ä Less expensive where oxygen is excessive u Adds dissolved solids ä Increased blowdown u Non-volatile ä No after boiler protection u No metal passivation Not for high pressure systems ä Breakdown to acidic gases

Sodium Sulfite Reaction with Dissolved Oxygen 2 Na 2 SO 3 + sodium sulfite O 2 oxygen 2 Na 2 SO 4 sodium sulfate Stoichiometry with oxygen is 8: 1

Sodium Sulfite Decomposes above 532°F (278°C) Na 2 SO 3 + H 2 O + sodium water sulfite Heat 2 Na. OH + sodium hydroxide SO 2 sulfur dioxide (acidic gas)

Diethylhydroxylamine DEHA Advantages u u Good oxygen scavenger Good volatility ä Similar to morpholine ä Contributes to condensate p. H neutralization ä Contributes to oxygen scavenging in after boiler section u u Does not add dissolved solids Good metal passivation ä Pre-boiler, boiler and after boiler sections u u Low toxicity Much less breakdown to ammonia than hydrazine

DEHA Reaction with Dissolved Oxygen 4(C 2 H 5)2 NOH DEHA + 9 O 2 oxygen 8 CH 3 COOH + N 2 + acetic acid nitrogen 6 H 2 O water The sodium hydroxide in the boiler water, neutralizes the acetic acid which is removed by blowdown as sodium acetate. Thermally stable up to 550°F (288°C). Stoichiometry with oxygen is 1. 24: 1

DEHA Forms Protective Oxide Films with Ferric and Cupric Oxide 2(C 2 H 5)2 NOH DEHA + 27 Fe 2 O 3 18 Fe 3 O 4 + ferric ferrous oxide (hematite) (magnetite) 2(C 2 H 5)2 NOH + 18 Cu. O DEHA cupric oxide 9 Cu 2 O + cuprous oxide 4 CH 3 COOH acetic acid + 3 H 2 O water 4 CH 3 COOH + N 2 + 3 H 2 O acetic acid nitrogen water

Factors Influencing Scale Formation u Solubility of a compound in water äAffected by concentration of compound and temperature of water u Precipitation äWater can no longer hold compound in solution

Factors That Accelerate Scale Formation u High Concentration of Dissolved Solids u High Water Temperature u Areas of Evaporation in Water Lines

Consequences of Scale Buildup u u u u Insulates Metal Surfaces Inefficient Heat Transfer Wasted Fuel Lost Production Capacity Potential for Under Deposit Corrosion Equipment Damage Localized overheating and metal failure

Calcium Formed Scales u Calcium Carbonate äUnder treatment with phosphate u Calcium Sulfate u Calcium Phosphate äHydroxyapatite is normal fluid sludge

Magnesium Formed Scales u Magnesium Silicate äNormal sludge u Magnesium Hydroxide äInsufficient phosphate and high alkalinity u Magnesium Carbonate äIf high Mg to Ca ratio exists

Silica Formed Scales u Silicon Dioxide äQuartz ¾ Carried into boiler system as sand or precipitates from boiler water äAmorphous ¾ Non-crystallized but aging can crystallize and form quartz

Iron Deposits in Boiler Systems u Oxides äMagnetite - ideal protection äHematite - occurs at neutral to low p. H in presence of oxygen

Copper Deposits In Boiler Systems u Metal äImproper acid cleaning äOverfeed of strong reducing agent äErosion in condensate or pre-boiler u Oxide äCorrosion product from alloys in system
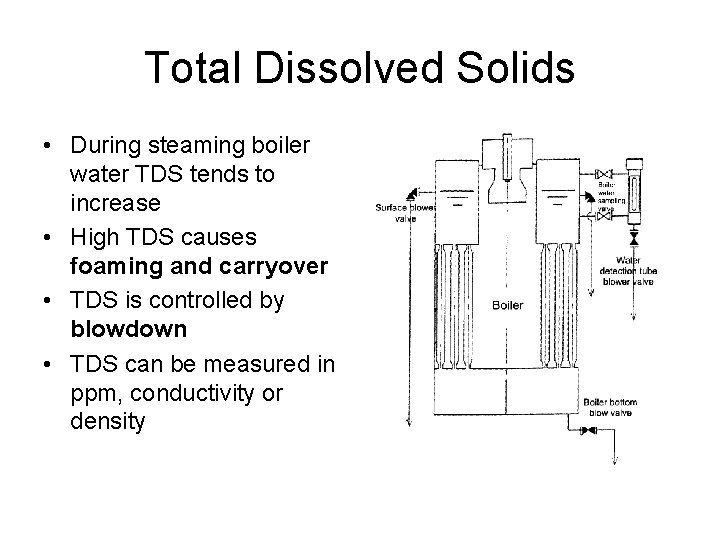
Total Dissolved Solids • During steaming boiler water TDS tends to increase • High TDS causes foaming and carryover • TDS is controlled by blowdown • TDS can be measured in ppm, conductivity or density

Total Dissolved Solids • Daily – Surface blow – Bottom blow • Measurement – PPM – Conductivity – Relative density • Sample cooler
Total Dissolved Solids

Sampling and Testing Procedures • Inaccurate gathering and analysis of water samples can result in losing control of the treatment program. • This can lead to a higher cost of chemical treatment, misapplication of treatment, reduced efficiency, and even boiler failure. • Sample point • Sampling Technique • Sampling Interval • Testing procedure

Record keeping • Well documented logs of water testing results are necessary to indicate the current status and trends of chemical treatment and general boiler operations. Records are particularly valuable for preventing boiler failures or determining the cause of failures that do occur. Well maintained records can predict the condition of the boiler before inspections are performed.

Survey & Inspections • Boiler inspections are necessary to document the effectiveness of the water treatment program. • Proper chemical treatment application and record keeping can allow one to predict the condition of the boiler, however the inspection documents the condition. • Proper documentation allows for comparison to previous inspections to see if the condition of the boiler has changed for the better or worse. • Photos and videos should be used wherever practical.

Summary Minimizing Water Treatment Problems Three Step Approach Ê Good Quality Makeup Water Ë Properly Applied Boiler Water System Treatment Program Ì Good Sampling, Monitoring and Control Program
- Slides: 39