Bohrs Model of the Atom Bohrs Model Why

Bohr’s Model of the Atom

Bohr’s Model Why don’t the electrons fall into the nucleus? e- move like planets around the sun. They move in circular orbits at different levels. Amounts of energy separate one level from another.

Bohr postulated that: Fixed energy related to the orbit Electrons cannot exist between orbits The higher the energy level, the further it is away from the nucleus An atom with maximum number of electrons in the outermost orbital energy level is stable (unreactive)
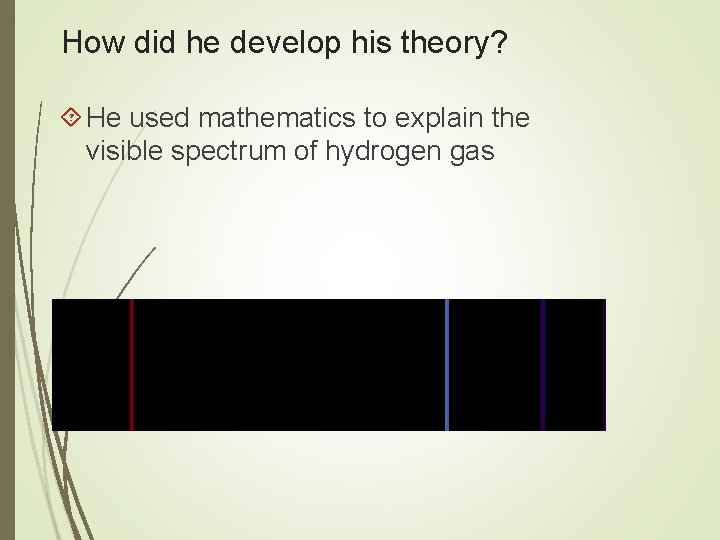
How did he develop his theory? He used mathematics to explain the visible spectrum of hydrogen gas
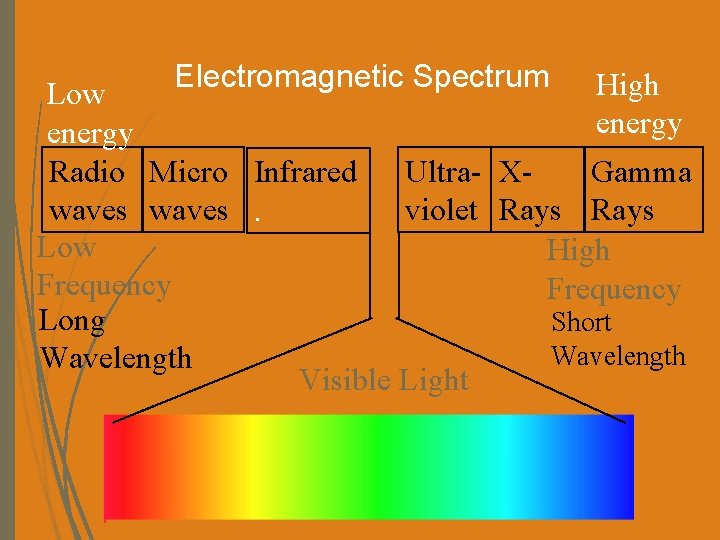
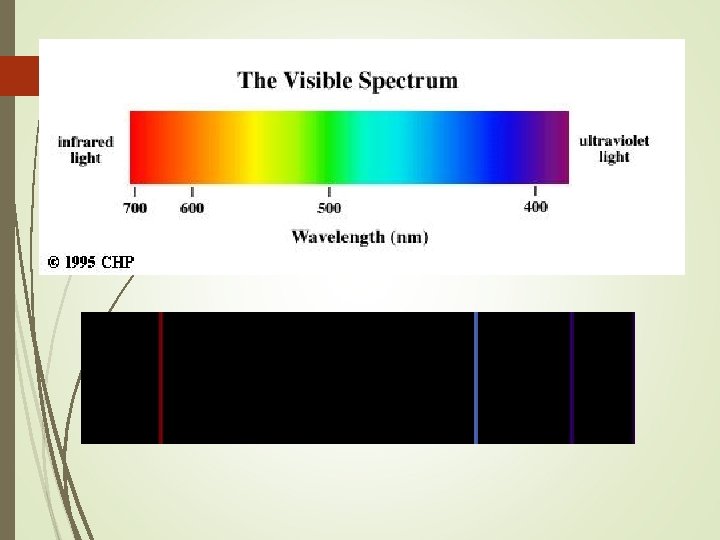
Electromagnetic Spectrum High Low energy Radio Micro Infrared Ultra- XGamma waves. violet Rays Low High Frequency Long Short Wavelength Visible Light
The line spectrum Electricity passed through a gaseous element emits light at a certain wavelength The colors can be seen when passed through a prism Every gas has a unique pattern (color)
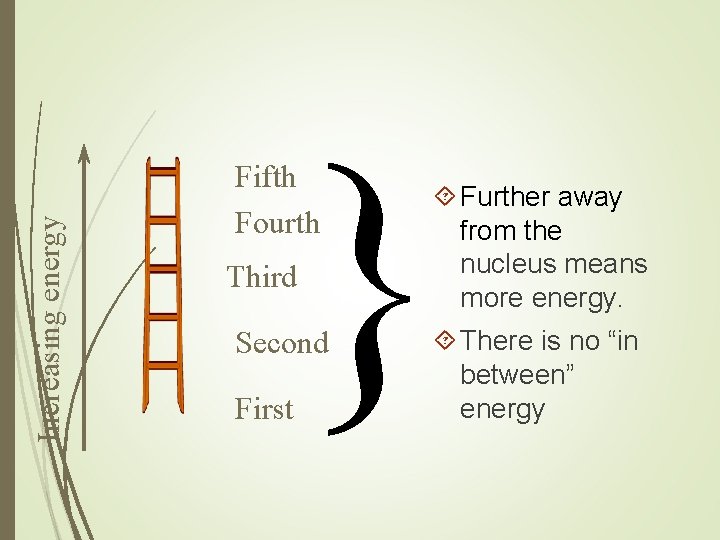
Increasing energy } Fifth Fourth Third Second First Further away from the nucleus means more energy. There is no “in between” energy
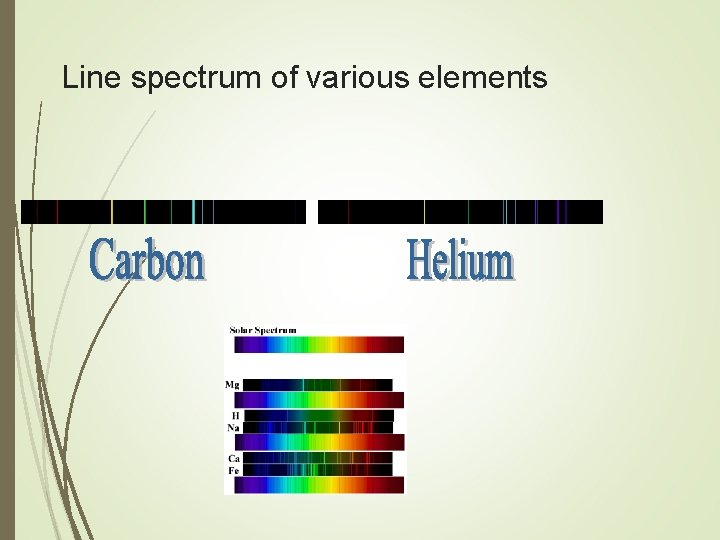
Line spectrum of various elements

Electrons orbiting closest to the nucleus are said to be in the lowest energy state called the ground state Atoms can absorb an amount of energy This promotes an electron to a higher energy level called the excited state This energy level is unstable and so the electron will fall back to its ground state When it does this, the excess energy will be emitted as light

When the e- falls from one energy level to another, an amount of energy is emitted as light This light emitted at specific wavelengths, which corresponds to our atomic spectra Each atom will have different electron “jumps” therefore emitting different amounts of energy as light This creates different line spectra for various elements

Let’s watch this video http: //www. mhhe. com/physsci/chemist ry/chang 7/esp/folder_structure/pe/m 1/s 3/

More videos (view OYO) http: //www. mhhe. com/physsci/astronomy/applets/Bohr/applet_files/B ohr. html http: //highered. mcgrawhill. com/sites/0072482621/student_view 0/interactives. html#

Bohr’s Triumph His theory helped to explain periodic law Halogens are so reactive because it has one e- less than a full outer orbital Alkali metals are also reactive because they have only one e- in outer orbital
Drawback Bohr’s theory did not explain or show the shape or the path traveled by the e-. His theory could only explain hydrogen and not the more complex atoms

The Quantum Mechanical Model Energy is quantized – meaning it comes in chunks. A quanta is the amount of energy needed to move from one energy level to another. Since the energy of an atom is never “in between” there must be a quantum leap in energy. An equation has been developed that described the energy and position of the electrons in an atom

Atomic Orbitals Principal Quantum Number (n) = the energy level of the electron. Within each energy level the complex math equation describes several shapes. These are called atomic orbitals Orbitals are regions where there is a high probability of finding an e-
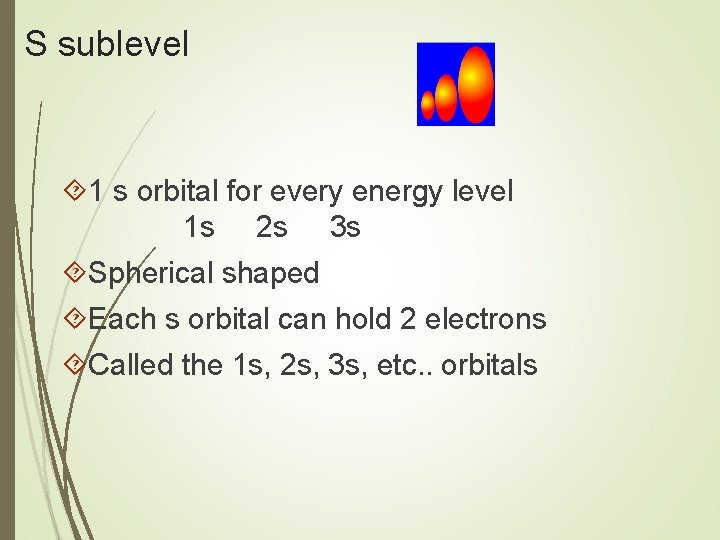
S sublevel 1 s orbital for every energy level 1 s 2 s 3 s Spherical shaped Each s orbital can hold 2 electrons Called the 1 s, 2 s, 3 s, etc. . orbitals
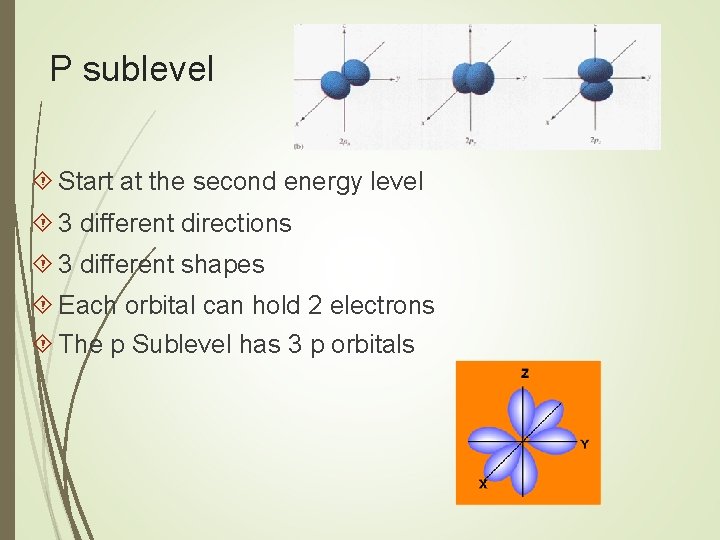
P sublevel Start at the second energy level 3 different directions 3 different shapes Each orbital can hold 2 electrons The p Sublevel has 3 p orbitals
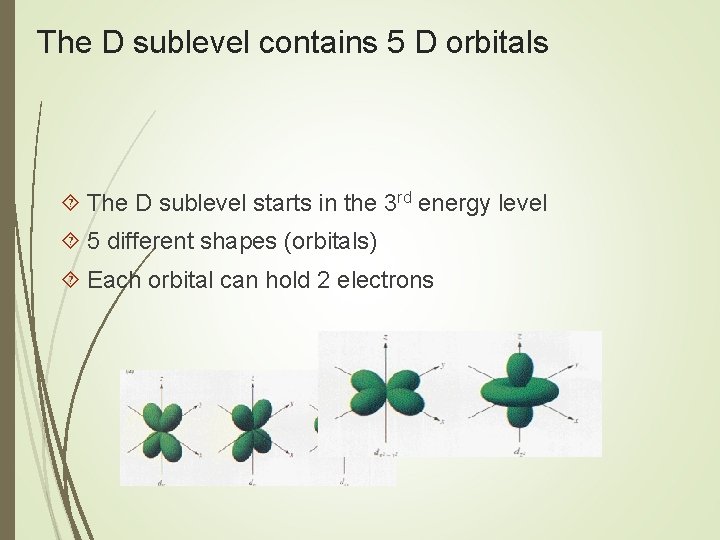
The D sublevel contains 5 D orbitals The D sublevel starts in the 3 rd energy level 5 different shapes (orbitals) Each orbital can hold 2 electrons
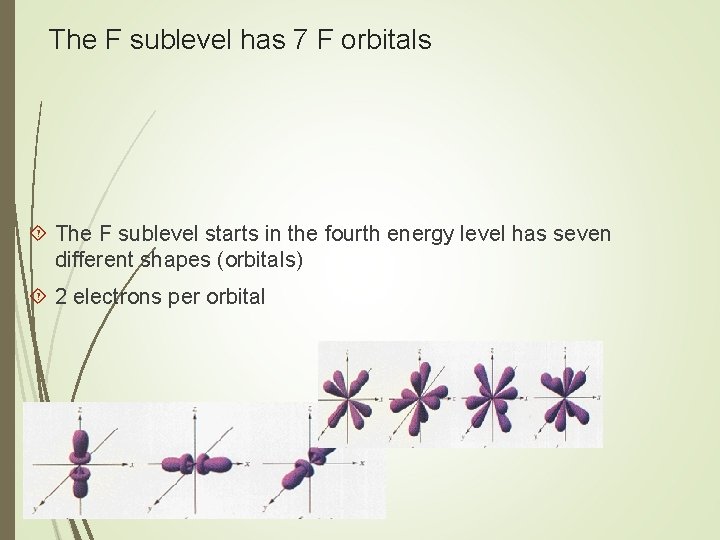
The F sublevel has 7 F orbitals The F sublevel starts in the fourth energy level has seven different shapes (orbitals) 2 electrons per orbital
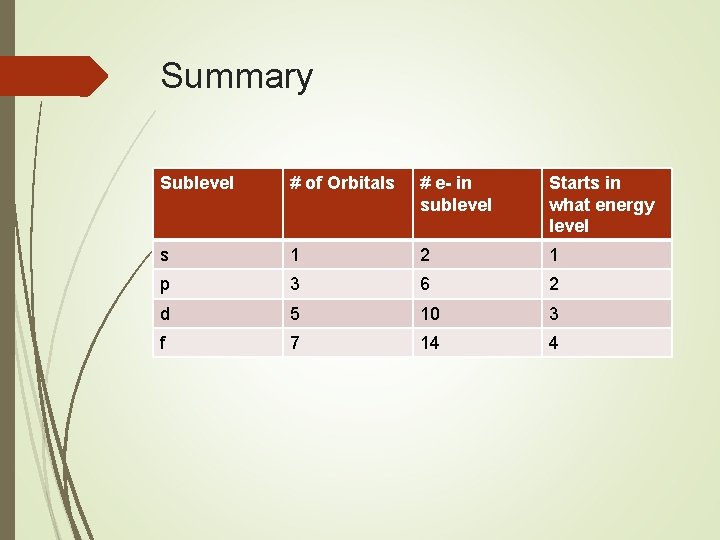
Summary Sublevel # of Orbitals # e- in sublevel Starts in what energy level s 1 2 1 p 3 6 2 d 5 10 3 f 7 14 4

Electron Configurations The way electrons are arranged in atoms. Aufbau principle- e- enter the lowest energy first. This causes difficulties because of the overlap of orbitals of different energies. Pauli Exclusion Principle- at most 2 e-per orbital with different spins Hund’s Rule- When e- occupy orbitals of equal energy they don’t pair up until they have to.
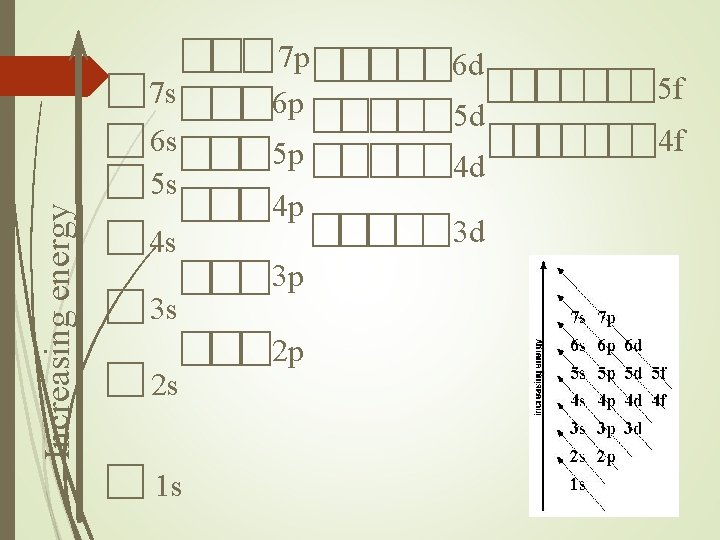
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 3 p 2 p 6 d 5 d 4 d 3 d 5 f 4 f

Orbitals fill in order Lowest energy to higher energy. Adding electrons can change the energy of the orbital. Half filled orbitals have a lower energy. Makes them more stable. Changes the filling order
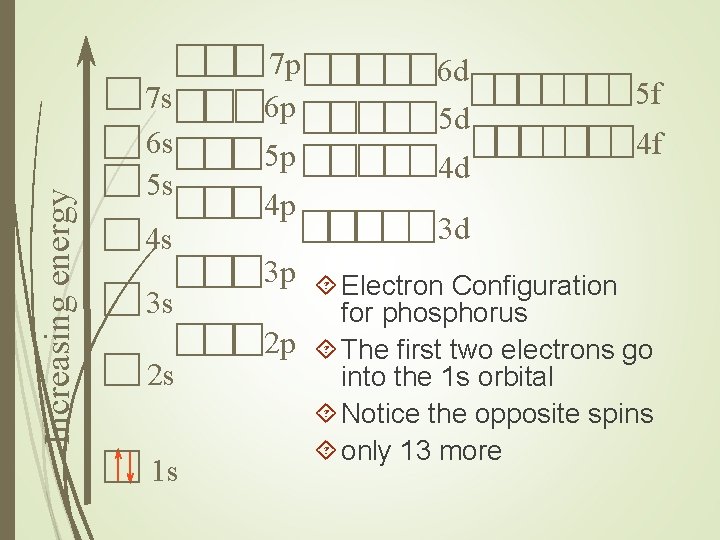
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 6 d 5 d 4 d 5 f 4 f 3 d 3 p Electron Configuration for phosphorus 2 p The first two electrons go into the 1 s orbital Notice the opposite spins only 13 more
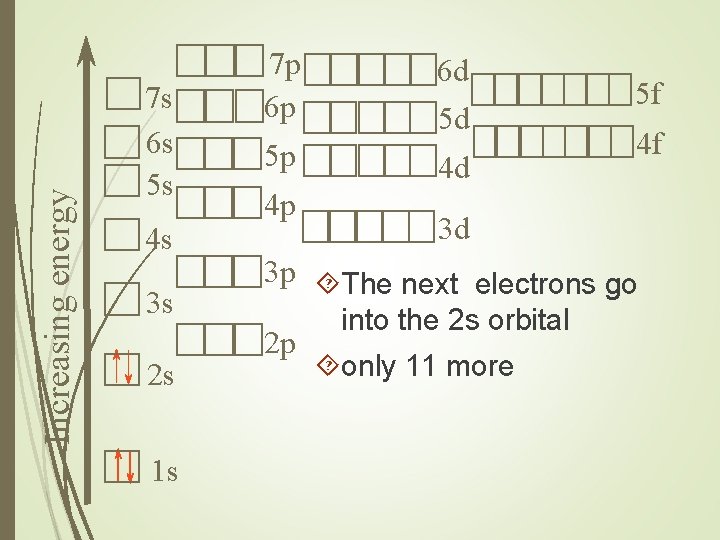
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 6 d 5 d 4 d 5 f 4 f 3 d 3 p The next electrons go 2 p into the 2 s orbital only 11 more
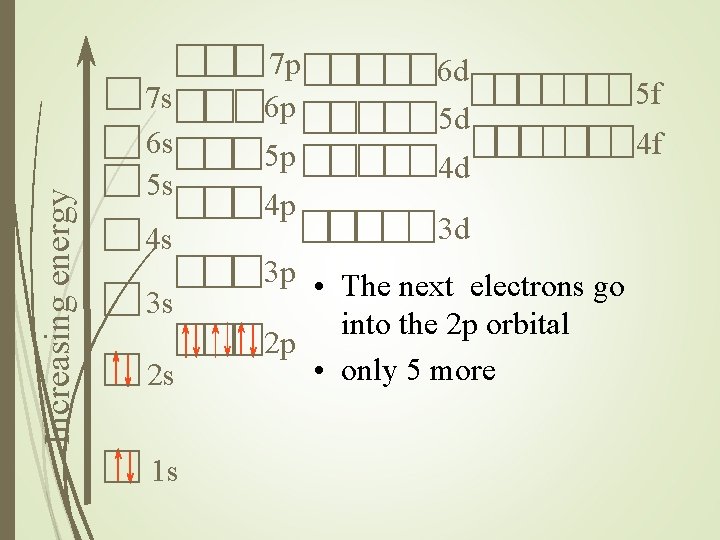
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 6 d 5 d 4 d 3 d 3 p • The next electrons go into the 2 p orbital 2 p • only 5 more 5 f 4 f
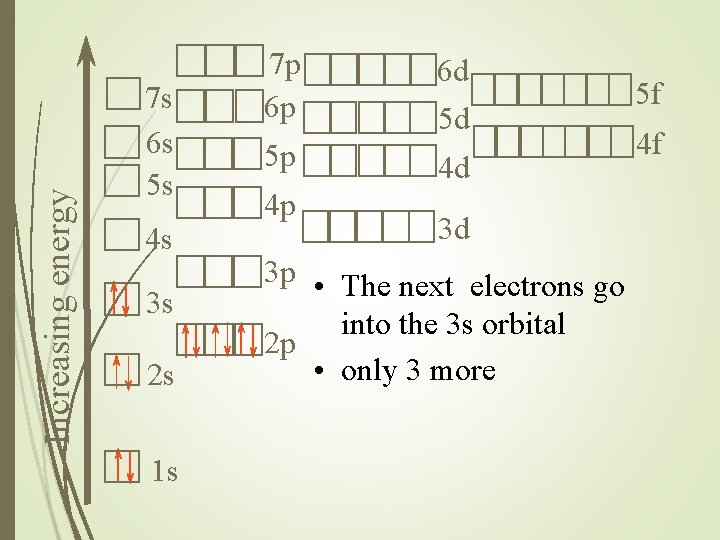
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 6 d 5 d 4 d 3 d 3 p • The next electrons go into the 3 s orbital 2 p • only 3 more 5 f 4 f
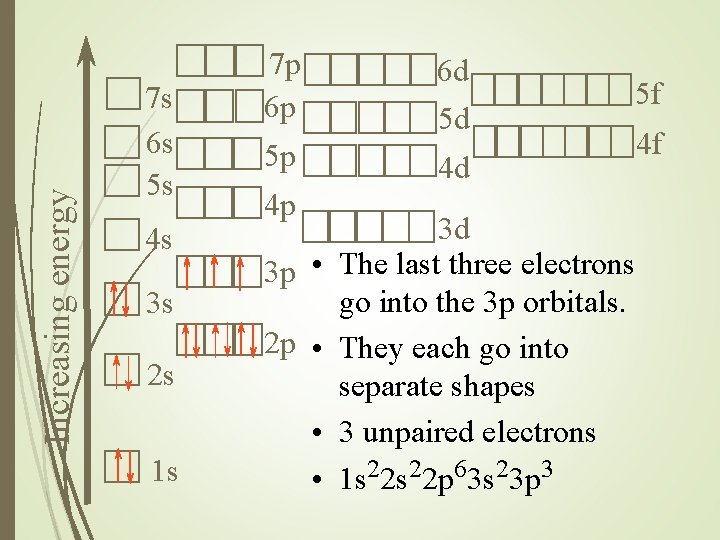
Increasing energy 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 6 d 5 d 5 p 5 f 4 d 4 p 3 p • 2 p • • • 3 d The last three electrons go into the 3 p orbitals. They each go into separate shapes 3 unpaired electrons 1 s 22 p 63 s 23 p 3 4 f

Write these electron onfigurations Titanium Vanadium Chromium Copper

Electron Configurations 2 2 6 2 3 Vanadium - 23 electrons 1 s 2 s 2 p 3 s 3 p 4 s 3 d 2 2 6 2 4 Chromium - 24 electrons 1 s 2 s 2 p 3 s 3 p 4 s 3 d Titanium - 22 electrons 1 s 2 s 2 p 3 s 3 p 4 s 3 d Expected. But, this is wrong!! 2 2 6 1 5 1 s 2 s 2 p 3 s 3 p 4 s 3 d 2 2 6 2 9 Copper – 29 electrons 1 s 2 s 2 p 3 s 3 p 4 s 3 d Actual configuration is 2 2 6 1 10 1 s 2 s 2 p 3 s 3 p 4 s 3 d Why are chromium and copper configurations different? This gives two half filled orbitals for chromium and one completely full and one half filled orbital for copper. Slightly lower in energy. Remember these exceptions
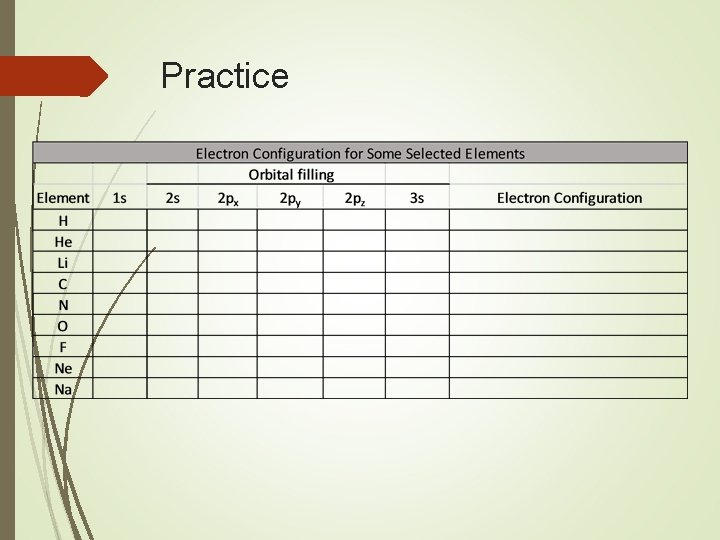
Practice
- Slides: 33