Bohrs model of H atom PHY 123 9302020

Bohr’s model of H atom PHY 123 9/30/2020 Lecture XIII 1

Niels Bohr (1885 -1962) 9/30/2020 Lecture XIII 2 By Iutta Waloschek

Hydrogen atom • Positively charged nucleus inside (+Ze), negatively charged electron (-e) around • Electron is attracted to nucleus • Electron is trapped in a potential well created by nucleus (“a box”) • Energy levels in atom 9/30/2020 Lecture XIII 3

Standing electron waves in Hydrogen atom • Standing waves: • 2 prn=nl l=h/mv mvrn=nh/2 p • Angular momentum L=mvrn is quantized L=nh/2 p • n – orbital quantum number 9/30/2020 Lecture XIII 4

Disclaimer • Though Bohr’s model was able to predict many properties of H atom and correctly calculate some of its characteristics, this model is incomplete and it is not advisable to think of an atom as a miniature “solar system”. • Orbits make sense only as average radii of electron position (not the same as electron slightly smeared around circular orbits!!!) 9/30/2020 Lecture XIII 5

Hydrogen atom • mvrn=nh/2 p v=nh/(2 pmrn) • Newton’s 2 nd law for circular motion: Bohr radius 9/30/2020 Lecture XIII 6

Hydrogen atom • Energy in H atom KE+U 9/30/2020 Lecture XIII 7

Hydrogen atom • What did we learn? • For high n energy approaches -0, radius approaches infinity • Energy proportional to Z 2 e 4 – Ze 2 from the potential, Ze 2 from 1/r (smaller orbits around larger charges) • Radius inversely proportional to mass (F=mv 2/r) Energy proportional to electron mass • Energy does not depend on nucleus mass – assume infinitely heavy • In classical case (h 0) rn 0 – electron falls on nucleus. 9/30/2020 Lecture XIII 8
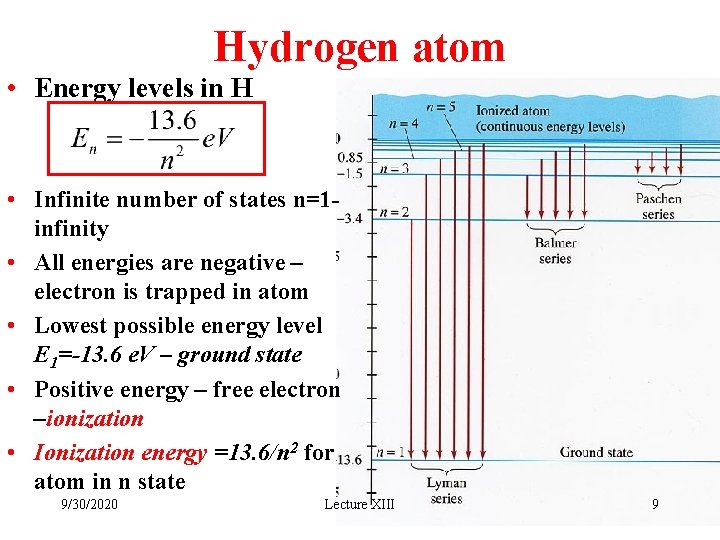
Hydrogen atom • Energy levels in H • Infinite number of states n=1 infinity • All energies are negative – electron is trapped in atom • Lowest possible energy level E 1=-13. 6 e. V – ground state • Positive energy – free electron –ionization • Ionization energy =13. 6/n 2 for atom in n state 9/30/2020 Lecture XIII 9

Some parameters of the H atom • Ground state n=1, Z=1 • Electron’s “orbit” radius • Wavelength • Momentum • v/c 9/30/2020 Lecture XIII 10

Some parameters of the H atom • Ground state n=1, Z=1 • Kinetic Energy • Potential Energy • Total energy • Longest wavelength absorbed (l – max E =hc/l – min) n=1 n=2 E 1=-13. 6 e. V E 2=-13. 6/4=-3. 4 e. V Eg=13. 6 -3. 4=10. 2 e. V l=hc/Eg=1243 e. Vnm/10. 2 e. V=122 nm 9/30/2020 Lecture XIII 11
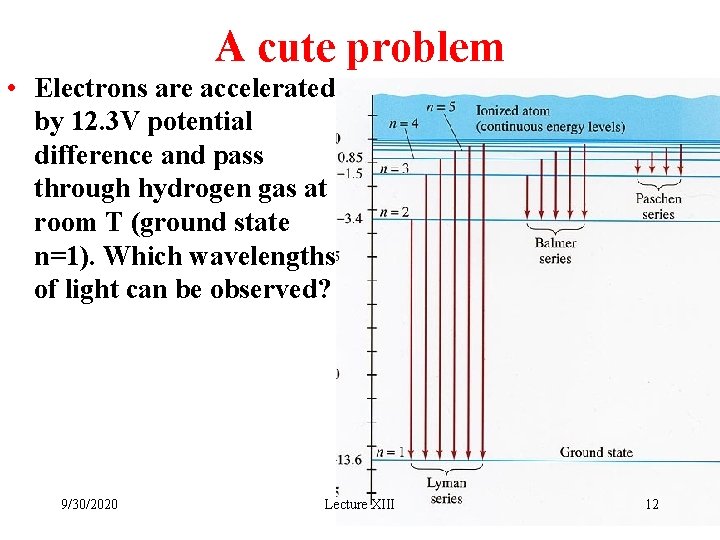
A cute problem • Electrons are accelerated by 12. 3 V potential difference and pass through hydrogen gas at room T (ground state n=1). Which wavelengths of light can be observed? 9/30/2020 Lecture XIII 12
- Slides: 12