Bohrs Atomic Model In 1913 the physicist Niels

Bohr's Atomic Model In 1913, the physicist Niels Bohr introduced a model of the atom that contributed a greater understanding to its structure and quantum mechanics. The Bohr Modelwas the first to propose quantum energy levels, where electrons orbit the nucleus at predefined distances and must overcome an energy barrier to move into a new orbital. Bohr was awarded a Nobel prize in 1922 for his investigations into atomic structure. 1. The electrons revolve around the nucleus in special orbits called discrete orbits to overcome the loss of energy. When an electron revolves around the nucleus in this orbit, it does not radiate energy. This proved that the electrons need not lose energy and fall into the nucleus. 2. Each orbit is called a shell or energy level, and each level contains a specific amount of energy. 3. An electron will absorb energy when moving from a lower energy level to a higher energy level. This is called an excited state. 4. An electron will radiate energy when moving from a higher energy level to a lower energy level. 5. When electrons move from one orbit to another, they emit photons, producing light in characteristic absorption and emission spectra. Since each element has its own signature, the spectra can be used to determine the composition of a material. 6. Orbits closer to the nucleus (those that have lower energy levels) are more stable. (An electron in its orbit with the lowest possible energy is said to be in its ground state. )

Limitations of Bohr's Atomic Model 1. Bohr's model only explains the spectra of species that have a single electron, such as the hydrogen atom , etc. 2. Bohr's theory predicts the origin of only one spectral line from an electron between any two given energy states. Under a spectroscope of strong resolution, a single line is found to split into a number of very closely related lines. Bohr's theory could not explain this multiple or fine structure of spectral lines. The appearance of the several lines implies that there are several sub energy levels of nearly similar energy for each principal quantum number, n. This necessitates the existence of new quantum numbers. 3. It does not explain the splitting of spectral lines under the influence of a magnetic field or under the influence of an electric field. 4. The pictorial concept of electrons jumping from one orbit to another orbit is not justified because of the uncertainty in their positions and velocities. Also, This theory is unrealistic in the sense that periodic motion around a central body usually follows an elliptical path rather than a circular path which has been assumed in the case of Bohr theory. If electrons follow elliptical path, the velocity along the path does not remain constant.
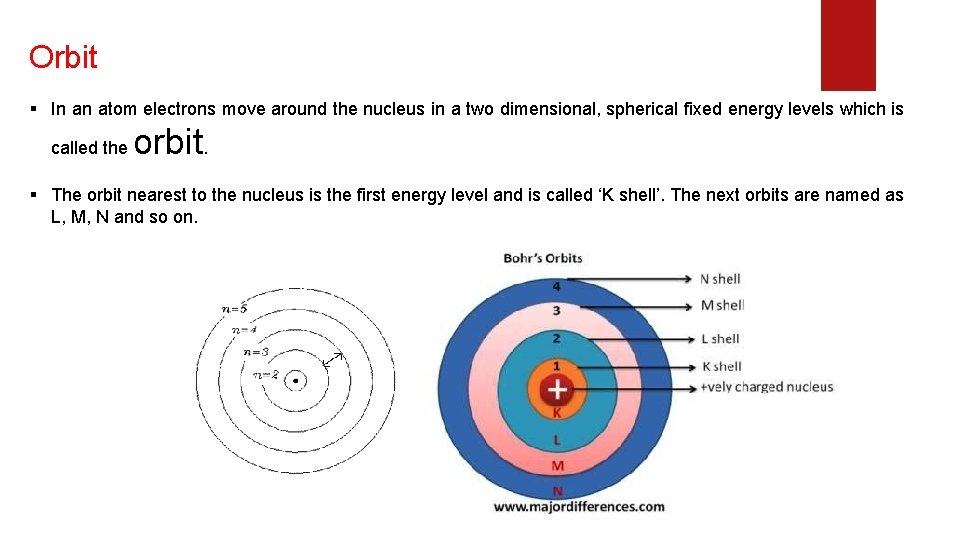
Orbit § In an atom electrons move around the nucleus in a two dimensional, spherical fixed energy levels which is called the orbit. § The orbit nearest to the nucleus is the first energy level and is called ‘K shell’. The next orbits are named as L, M, N and so on.
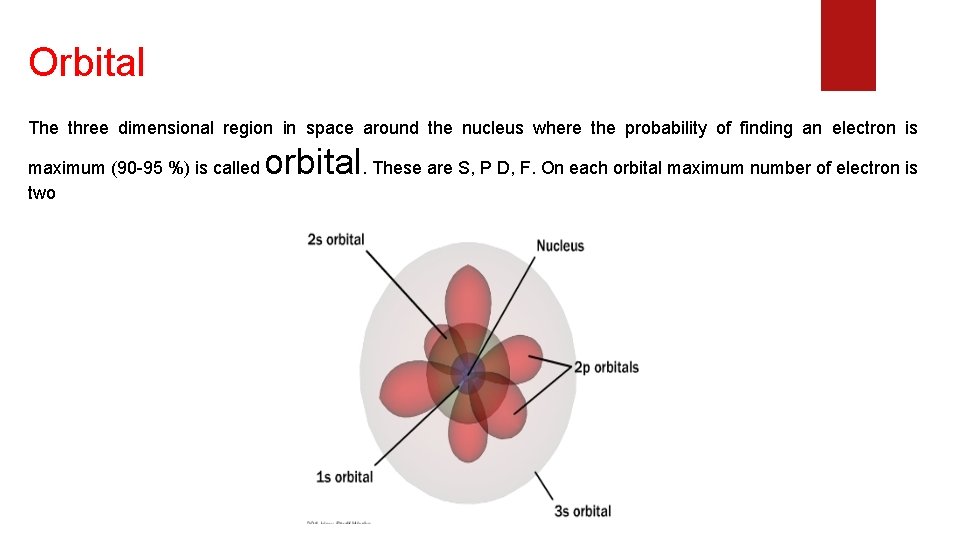
Orbital The three dimensional region in space around the nucleus where the probability of finding an electron is maximum (90 -95 %) is called two orbital. These are S, P D, F. On each orbital maximum number of electron is
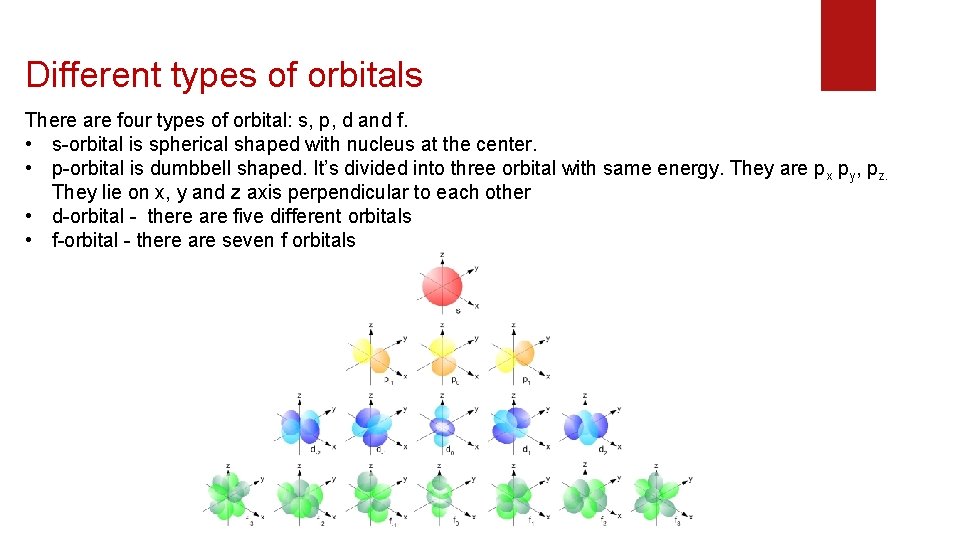
Different types of orbitals There are four types of orbital: s, p, d and f. • s-orbital is spherical shaped with nucleus at the center. • p-orbital is dumbbell shaped. It’s divided into three orbital with same energy. They are px py, pz. They lie on x, y and z axis perpendicular to each other • d-orbital - there are five different orbitals • f-orbital - there are seven f orbitals
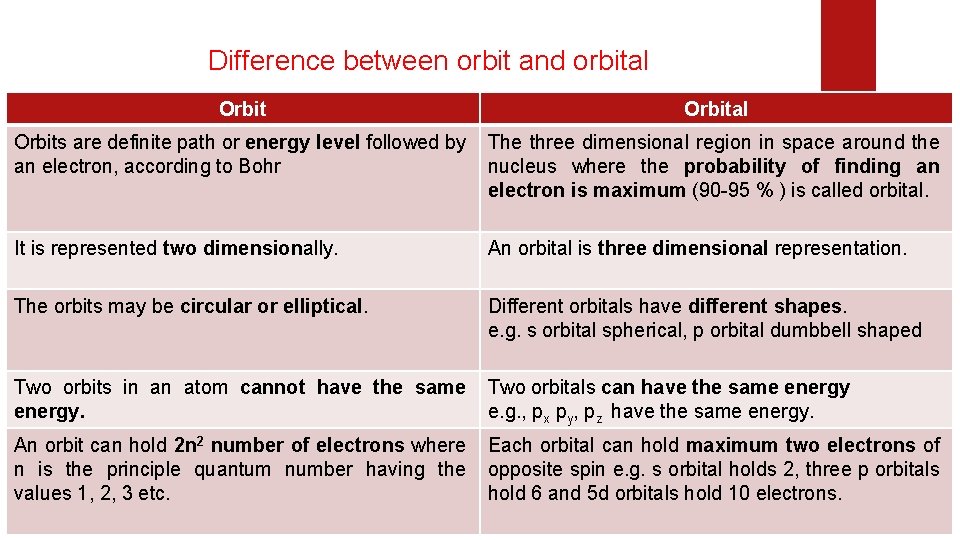
Difference between orbit and orbital Orbital Orbits are definite path or energy level followed by an electron, according to Bohr The three dimensional region in space around the nucleus where the probability of finding an electron is maximum (90 -95 % ) is called orbital. It is represented two dimensionally. An orbital is three dimensional representation. The orbits may be circular or elliptical. Different orbitals have different shapes. e. g. s orbital spherical, p orbital dumbbell shaped Two orbits in an atom cannot have the same energy. Two orbitals can have the same energy e. g. , px py, pz have the same energy. An orbit can hold 2 n 2 number of electrons where n is the principle quantum number having the values 1, 2, 3 etc. Each orbital can hold maximum two electrons of opposite spin e. g. s orbital holds 2, three p orbitals hold 6 and 5 d orbitals hold 10 electrons.

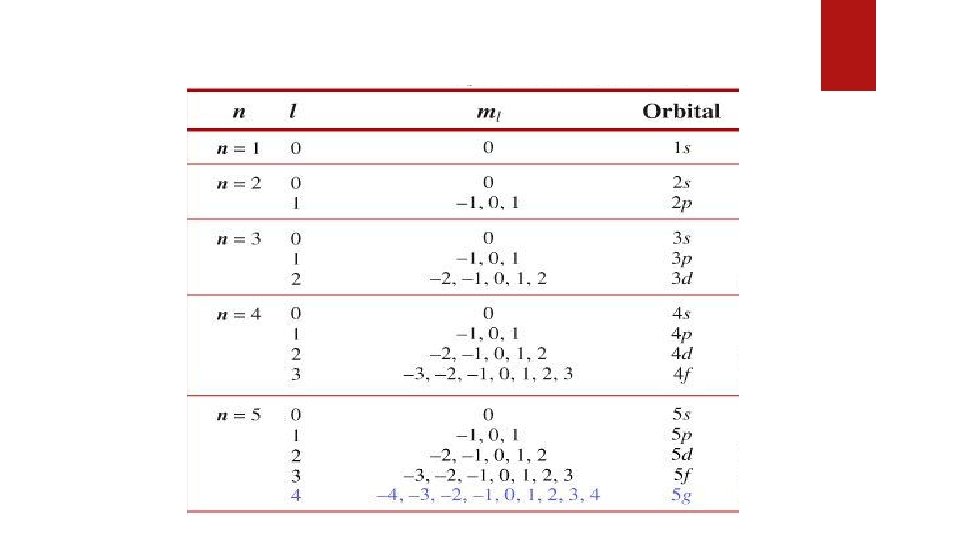
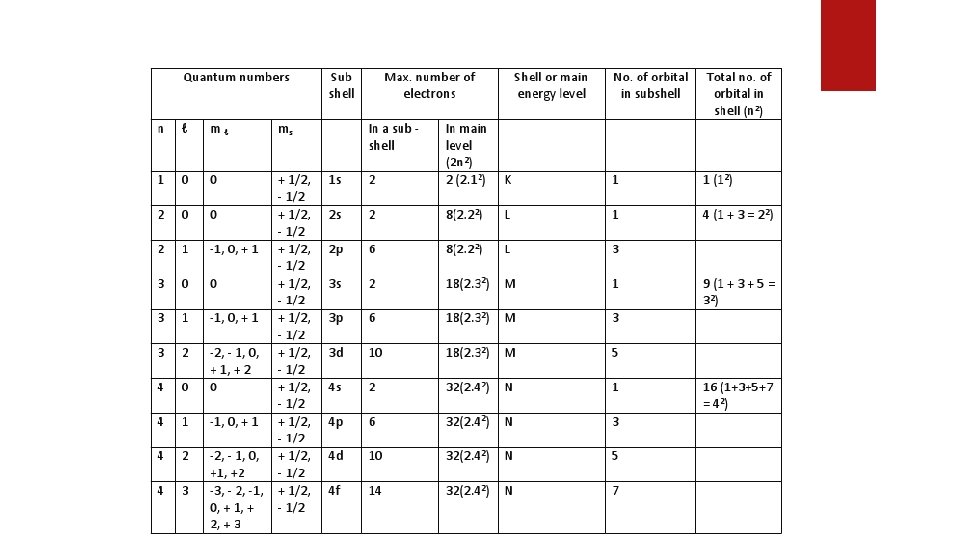
Quantum Numbers Quantum numbers are a set of values that describes the state of an electron including the size of an electron orbit (energy levels), the shape of the orbitals, orientation of orbital in the magnetic field and the spin of electrons about its own axis. The above four characteristics are represented by four Quantum Numbers 1. 2. 3. 4. Principal quantum number (n) Azimuthal or subsidiary quantum number (l) Magnetic quantum number (ml) Spin quantum number (ms)
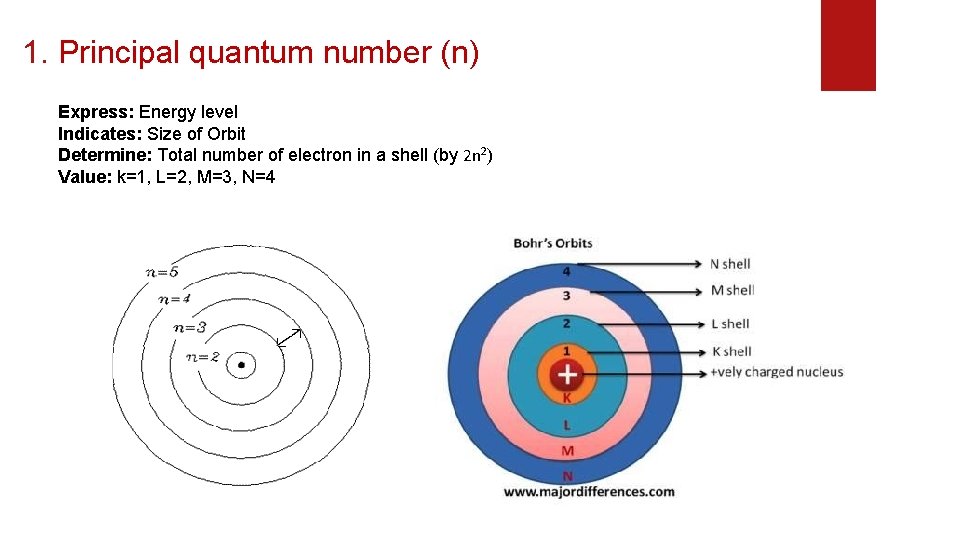
1. Principal quantum number (n) Express: Energy level Indicates: Size of Orbit Determine: Total number of electron in a shell (by 2 n 2) Value: k=1, L=2, M=3, N=4

2. Azimuthal or subsidiary quantum number (l) Express: Which orbitals are present in orbit & Concepts of orbitals of different shapes. Letter s p d f g > 0 1 2 3 4 Indicates: shapes and numbers of orbitals in a shell Determine: total number of electrons in an orbital as 2(2 l+1) Value: 0 to (n-1); e. g. n = 3, l = 0, 1, 2
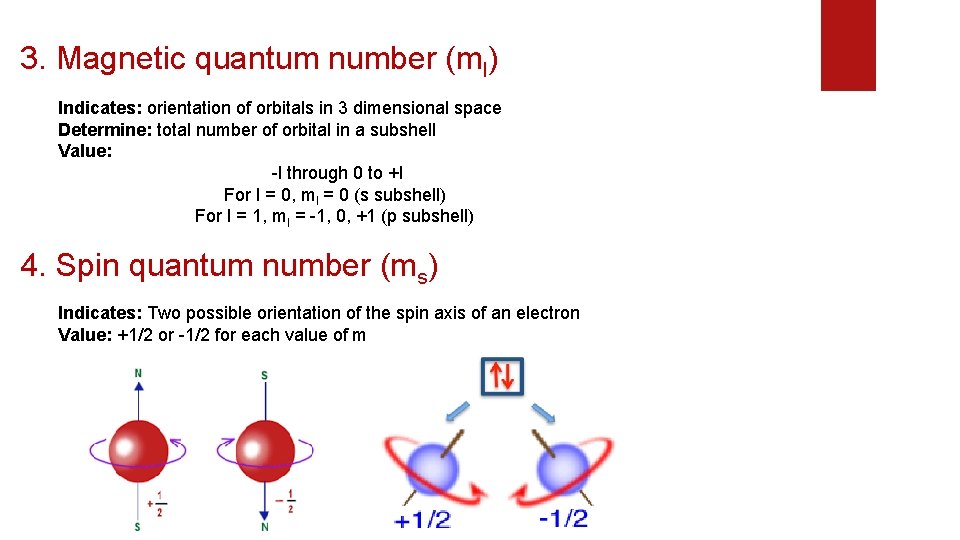
3. Magnetic quantum number (ml) Indicates: orientation of orbitals in 3 dimensional space Determine: total number of orbital in a subshell Value: -l through 0 to +l For l = 0, ml = 0 (s subshell) For l = 1, ml = -1, 0, +1 (p subshell) 4. Spin quantum number (ms) Indicates: Two possible orientation of the spin axis of an electron Value: +1/2 or -1/2 for each value of m


Electronic Configuration of Atoms In an atom the electrons are held in different orbits, sub-shells and orbital in a regular way. This arrangement of electrons in an atom is called the electronic configuration. Principles that influence the electronic configuration of atom: Ø Pauli’s exclusion principle Ø Aufbau principle Ø Hund’s rule

Wolfgang Pauli 1900 -1958 Nobel Prize in Physics 1945 "for the discovery of the Exclusion Principle, also called the Pauli Principle. "

Friedrich Hund 1896 -1997 Max Planck Medal in Physics 1943

Ø Pauli’s exclusion principle According to this principle, ‘no two electrons in the same atom can have the same values for the four quantum numbers’. For example, He (helium) had two electrons in 1 s orbital and their electronic configuration is 1 s 2, that means they had two electrons in the 1 s orbital. This two electrons can have same values for three of their quantum numbers, but the fourth quantum number must be different for the two electrons. Electronic configuration of helium atom is: He (2) = 1 s 2 For the first electron, n = 1, l = 0, ms = +1/2 And for the second electron, n = 1, l = 0, ms = -1/2 So, these two electrons have different values of their spin quantum number.

Ø Aufbau principle According to this principle, ‘the electrons will first occupy the lowest energy levels’. v. The energy of the levels are determined by their (n + l) values. v The electron will first occupy the orbital having the lowest value of (n + l) 1 s < 2 p < 3 s < 3 p < 4 s < 3 d < 4 p < 5 s…………
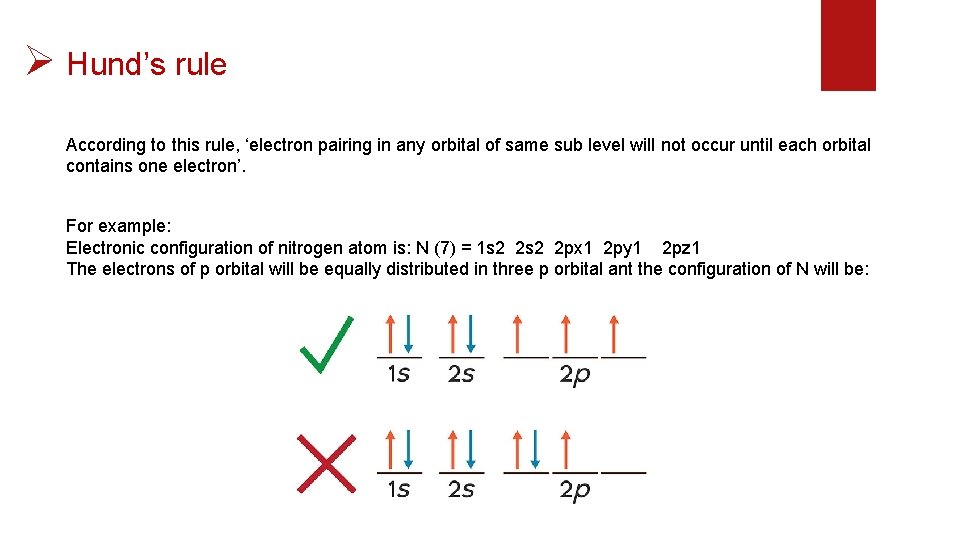
Ø Hund’s rule According to this rule, ‘electron pairing in any orbital of same sub level will not occur until each orbital contains one electron’. For example: Electronic configuration of nitrogen atom is: N (7) = 1 s 2 2 px 1 2 py 1 2 pz 1 The electrons of p orbital will be equally distributed in three p orbital ant the configuration of N will be:
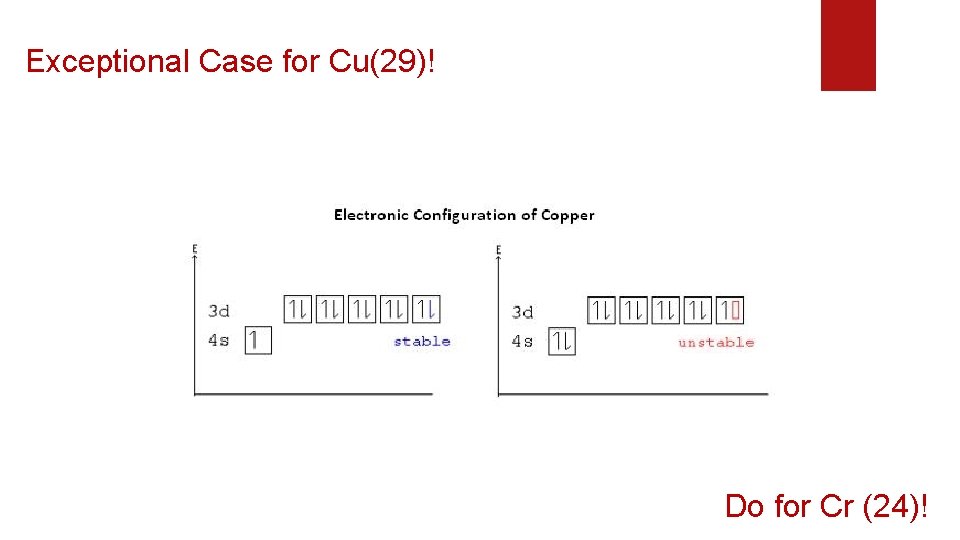
Exceptional Case for Cu(29)! Do for Cr (24)!
- Slides: 20