Bohrs Atomic Model electrons can only be in
Bohr’s Atomic Model electrons can only be in discrete orbitals Suggested________________ and that the electrons Absorb or emit energy as they move between orbitals ____________________

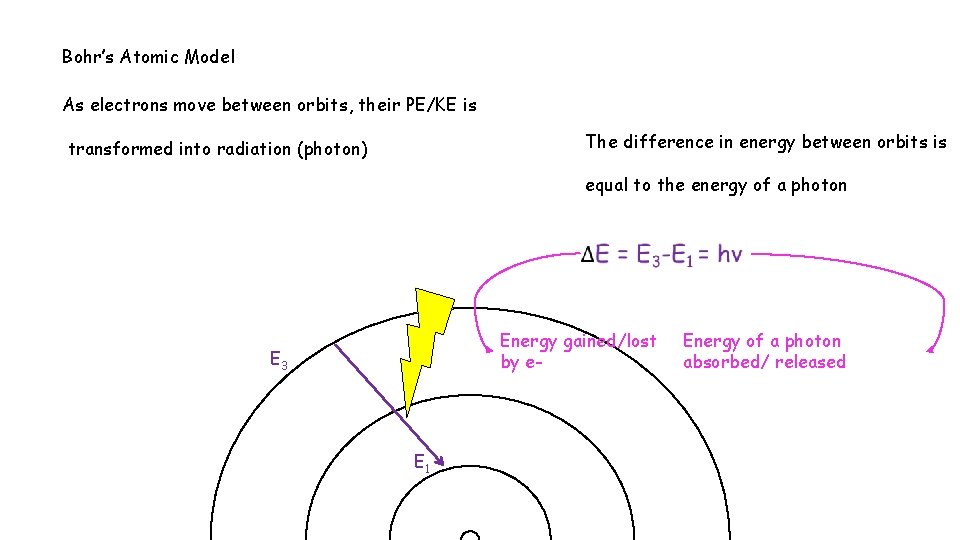
Bohr’s Atomic Model As electrons move between orbits, their PE/KE is The difference in energy between orbits is transformed into radiation (photon) equal to the energy of a photon Energy gained/lost by e- E 3 E 1 Energy of a photon absorbed/ released

Bohr’s Atomic Model Where can the electrons be? ? ? Bohr modeled the electrons as orbiting the nucleus, in contrast to the classical physics description of charged particles. Condition of Stability……. The electron must move fast enough so that v e The________ centripetal force equals the electromagnetic force between the electron and nucleus Z: number of protons e: fundamental charge (1. 60 x 10 -19) coulombs r: radius v: speed (Ze) r
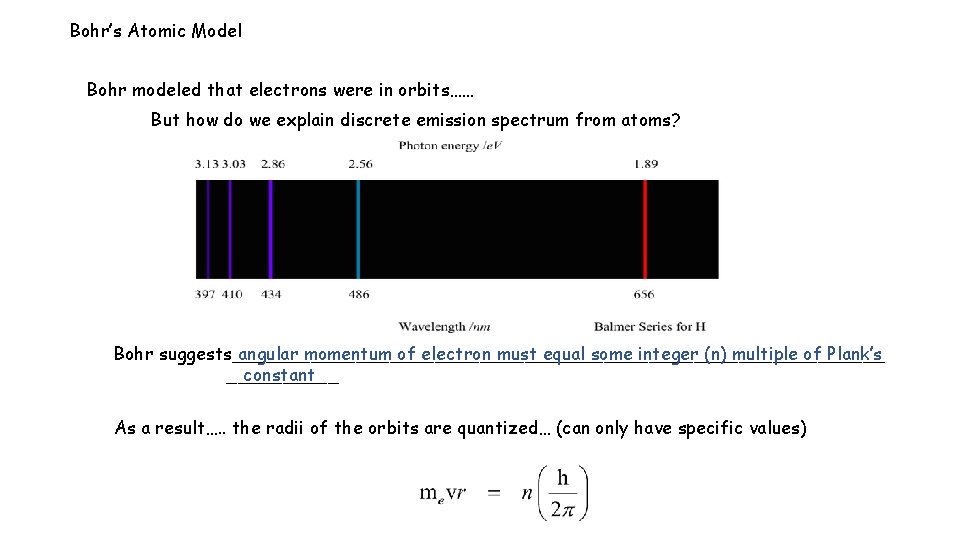
Bohr’s Atomic Model Bohr modeled that electrons were in orbits…… But how do we explain discrete emission spectrum from atoms? Bohr suggests_____________________________ angular momentum of electron must equal some integer (n) multiple of Plank’s _____ constant As a result…. . the radii of the orbits are quantized… (can only have specific values)
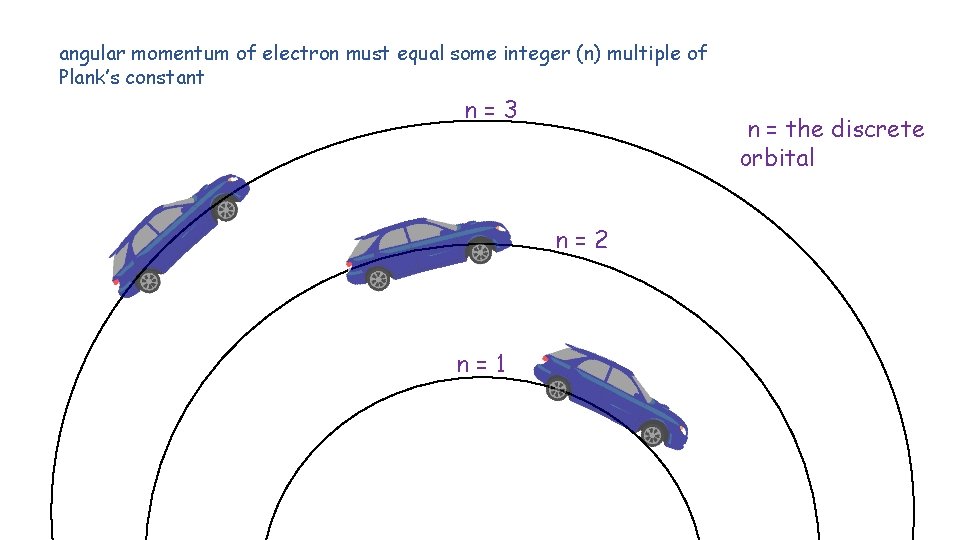
angular momentum of electron must equal some integer (n) multiple of Plank’s constant n=3 n = the discrete orbital n=2 n=1
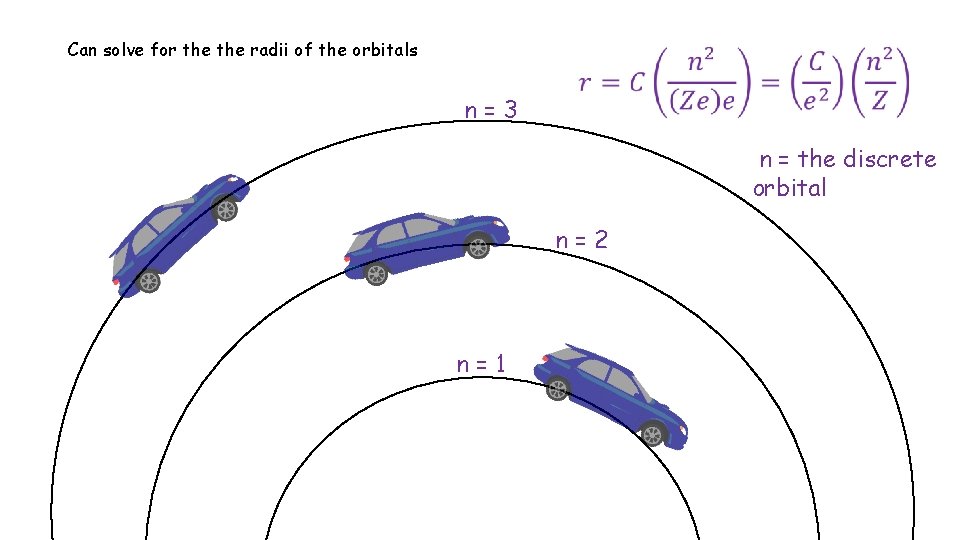
Can solve for the radii of the orbitals n=3 n = the discrete orbital n=2 n=1
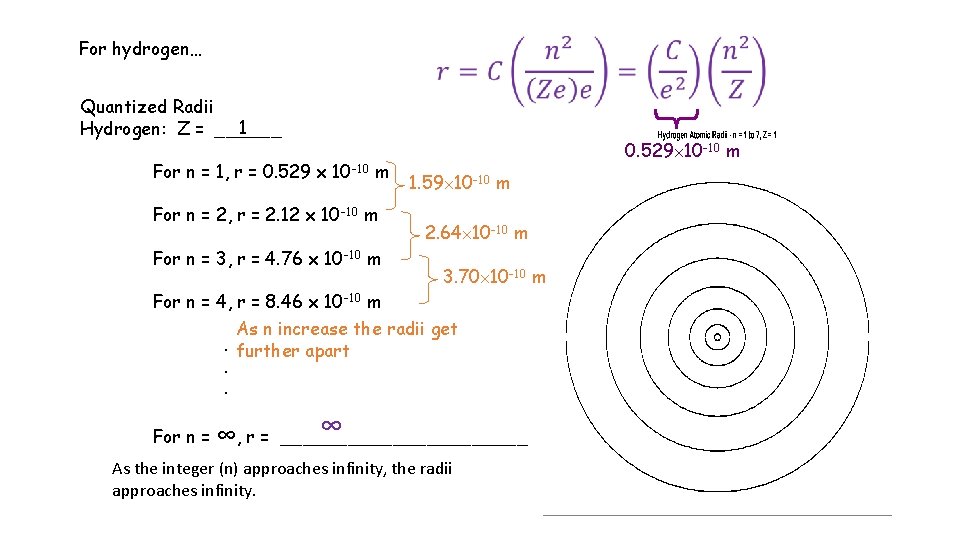
For hydrogen… Quantized Radii 1 Hydrogen: Z = ______ For n = 1, r = 0. 529 x 10 -10 m For n = 2, r = 2. 12 x 10 -10 m For n = 3, r = 4. 76 x 10 -10 m For n = 4, r = 8. 46 x 10 -10 m 0. 529 10 -10 m 1. 59 10 -10 m 2. 64 10 -10 m 3. 70 10 -10 m As n increase the radii get. further apart. . For n = ∞, r = ∞ ___________ As the integer (n) approaches infinity, the radii approaches infinity.
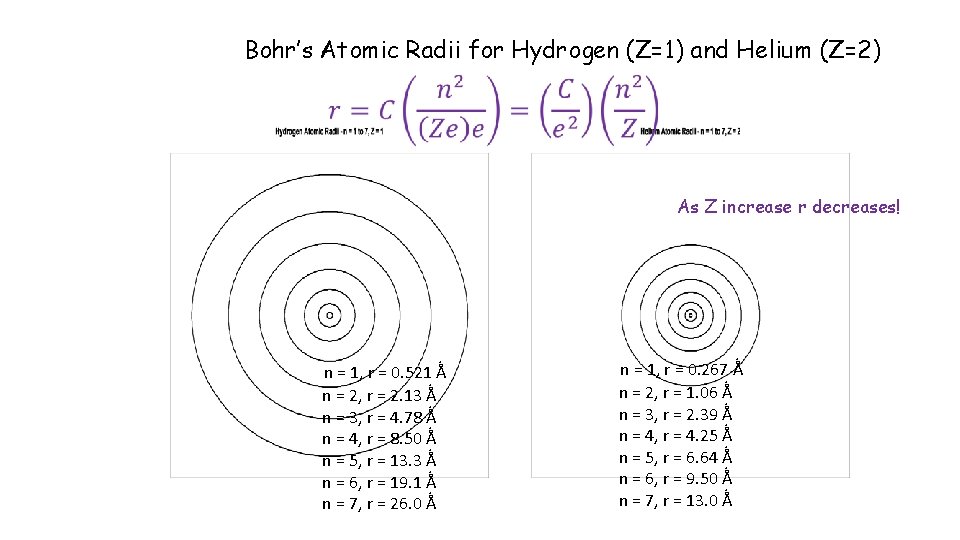
Bohr’s Atomic Radii for Hydrogen (Z=1) and Helium (Z=2) As Z increase r decreases! n = 1, r = 0. 521 Ǻ n = 2, r = 2. 13 Ǻ n = 3, r = 4. 78 Ǻ n = 4, r = 8. 50 Ǻ n = 5, r = 13. 3 Ǻ n = 6, r = 19. 1 Ǻ n = 7, r = 26. 0 Ǻ n = 1, r = 0. 267 Ǻ n = 2, r = 1. 06 Ǻ n = 3, r = 2. 39 Ǻ n = 4, r = 4. 25 Ǻ n = 5, r = 6. 64 Ǻ n = 6, r = 9. 50 Ǻ n = 7, r = 13. 0 Ǻ

How do we find the energy of an electron? With the position of the e- known we can determine the energy of it We don’t know how fast the e- is moving (v)…. But we can manipulate the variables and combine terms to get energy in terms of r. get

Quantized energy states The energy depends on where the electron is. If the radii are quantized (discrete) , the energies will be as well get (bunch o’ constants)
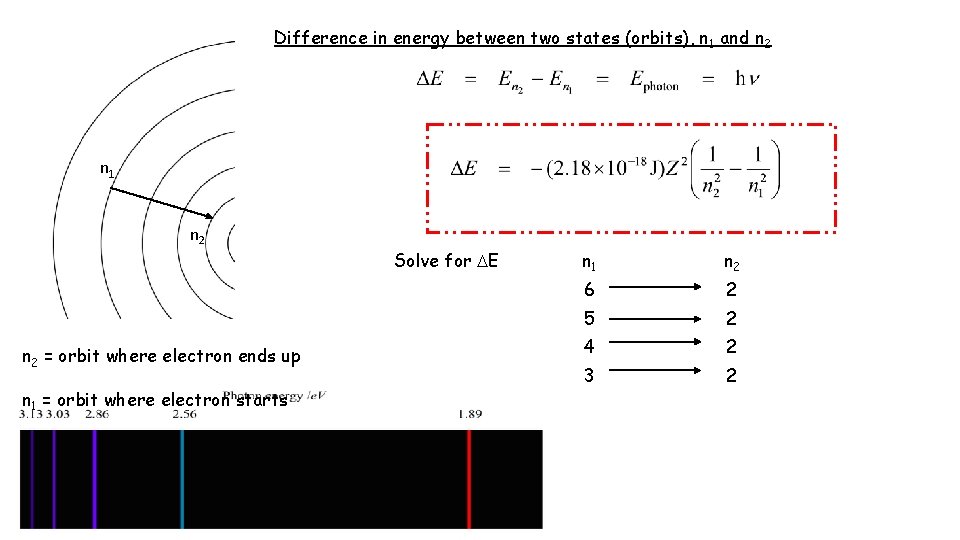
Difference in energy between two states (orbits), n 1 and n 2 n 1 n 2 = orbit where electron ends up n 1 = orbit where electron starts Solve for E n 1 n 2 6 2 5 2 4 2 3 2
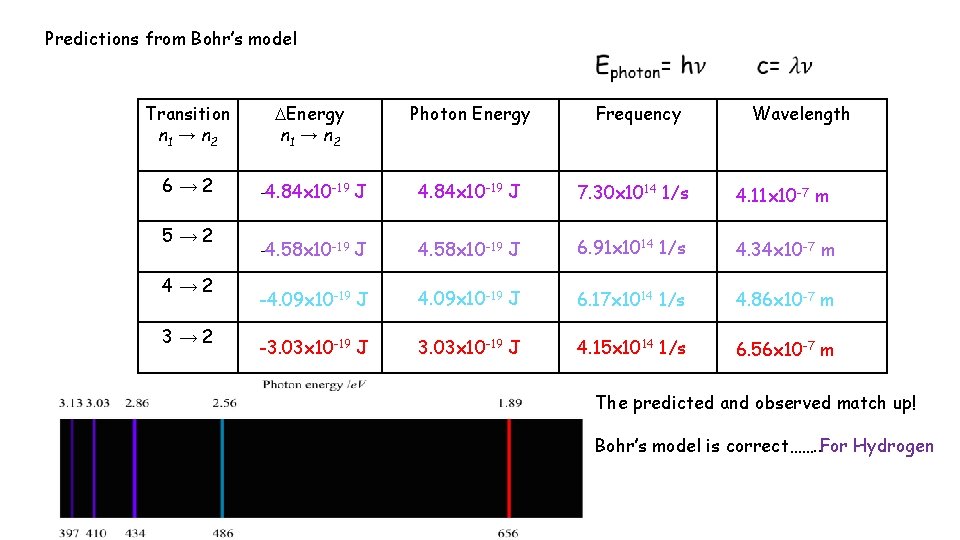
Predictions from Bohr’s model Transition n 1 → n 2 Energy n 1 → n 2 Photon Energy Frequency 6→ 2 -4. 84 x 10 -19 J 7. 30 x 1014 1/s 4. 11 x 10 -7 m -4. 58 x 10 -19 J 6. 91 x 1014 1/s 4. 34 x 10 -7 m -4. 09 x 10 -19 J 6. 17 x 1014 1/s 4. 86 x 10 -7 m -3. 03 x 10 -19 J 4. 15 x 1014 1/s 6. 56 x 10 -7 m 5→ 2 4→ 2 3→ 2 Wavelength The predicted and observed match up! Bohr’s model is correct……. . For Hydrogen

. … m u i l Bohr’s Atomic Radii for Hydrogen (Z=1) and Helium (Z=2) e H r o f a t a d e h t t u o b a t a h W. . n = 1, r = 0. 521 Ǻ n = 2, r = 2. 13 Ǻ n = 3, r = 4. 78 Ǻ n = 4, r = 8. 50 Ǻ n = 5, r = 13. 3 Ǻ n = 6, r = 19. 1 Ǻ n = 7, r = 26. 0 Ǻ n = 1, r = 0. 267 Ǻ n = 2, r = 1. 06 Ǻ n = 3, r = 2. 39 Ǻ n = 4, r = 4. 25 Ǻ n = 5, r = 6. 64 Ǻ n = 6, r = 9. 50 Ǻ n = 7, r = 13. 0 Ǻ
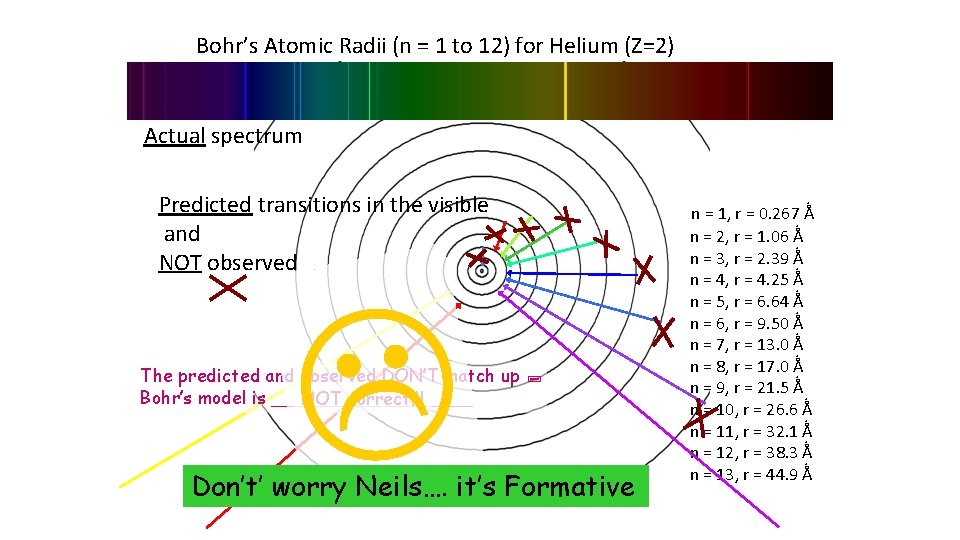
Bohr’s Atomic Radii (n = 1 to 12) for Helium (Z=2) Actual spectrum Predicted transitions in the visible and NOT observed. The predicted and observed DON’T match up Bohr’s model is _________ NOT correct!!! Don’t’ worry Neils…. it’s Formative . n = 1, r = 0. 267 Ǻ n = 2, r = 1. 06 Ǻ n = 3, r = 2. 39 Ǻ n = 4, r = 4. 25 Ǻ n = 5, r = 6. 64 Ǻ n = 6, r = 9. 50 Ǻ n = 7, r = 13. 0 Ǻ n = 8, r = 17. 0 Ǻ n = 9, r = 21. 5 Ǻ n = 10, r = 26. 6 Ǻ n = 11, r = 32. 1 Ǻ n = 12, r = 38. 3 Ǻ n = 13, r = 44. 9 Ǻ
- Slides: 15