Bohrs Atomic Model Bromfield Honors Chemistry Modern Atomic

Bohr’s Atomic Model Bromfield Honors Chemistry
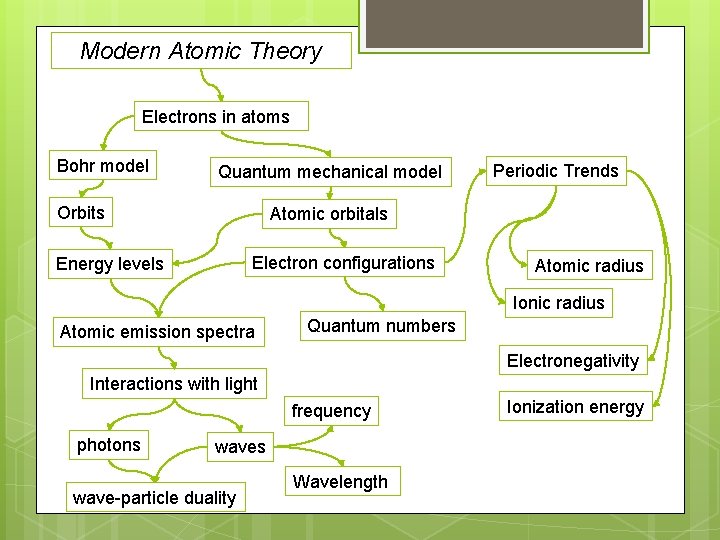
Modern Atomic Theory Electrons in atoms Bohr model Quantum mechanical model Orbits Periodic Trends Atomic orbitals Electron configurations Energy levels Atomic radius Ionic radius Atomic emission spectra Quantum numbers Electronegativity Interactions with light frequency photons wave-particle duality Wavelength Ionization energy

Do now… Do this on the small whiteboards… A ruby laser emits light at a wavelength of 694. 3 nm Calculate the frequency, in s-1, of the light. Calculate the energy of one photon of this light c = 2. 998 x 108 m/s h = 6. 626 x 10 -34 J s

Video overview https: //youtu. be/l. Jh 2 Ra 1 eyg. A

White light source
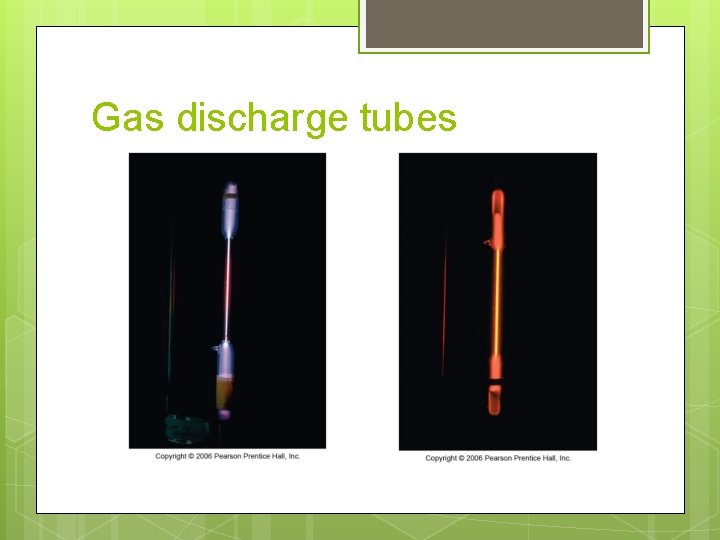
Gas discharge tubes
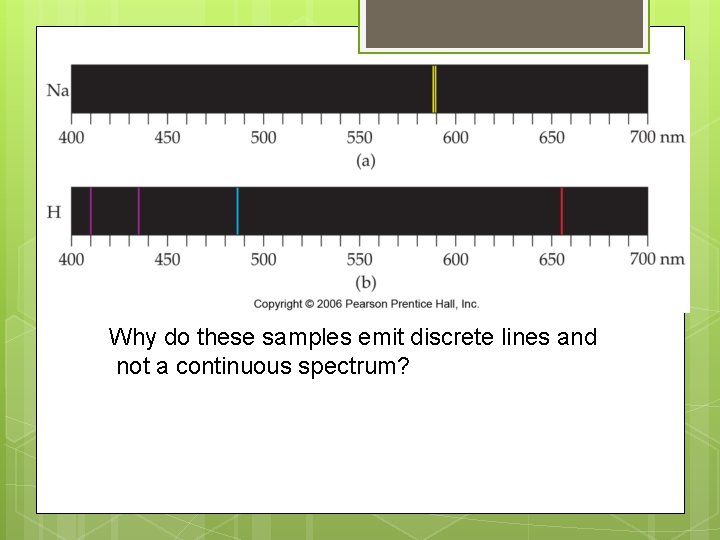
Element emission spectra Why do these samples emit discrete lines and not a continuous spectrum?

Light It exhibits a wave-particle duality can be treated as a wave

Light exhibits a wave-particle duality It can be treated as a wave It can be treated as a particle Photon Spoiler or quantum of light alert: Electrons also exhibit a waveparticle duality!

Max Planck German physicist

Max Planck Energy is QUANTIZED i. e. , only certain (discrete) energies are allowed

Niels Bohr Worked with Rutherford Developed an explanation for atomic emission spectra Refined Rutherford’s atomic model

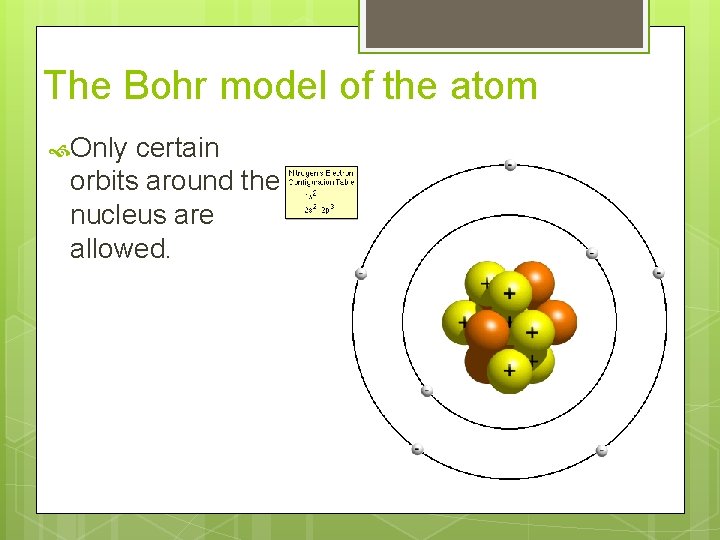
The Bohr model of the atom Only certain orbits around the nucleus are allowed.
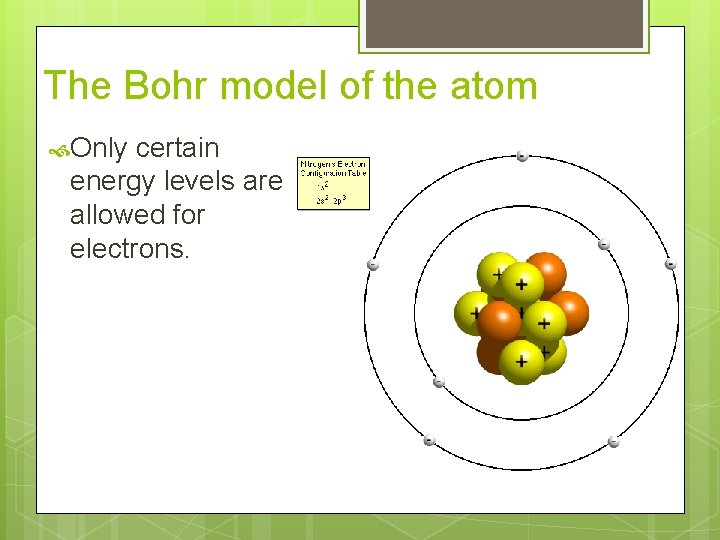
The Bohr model of the atom Only certain energy levels are allowed for electrons.
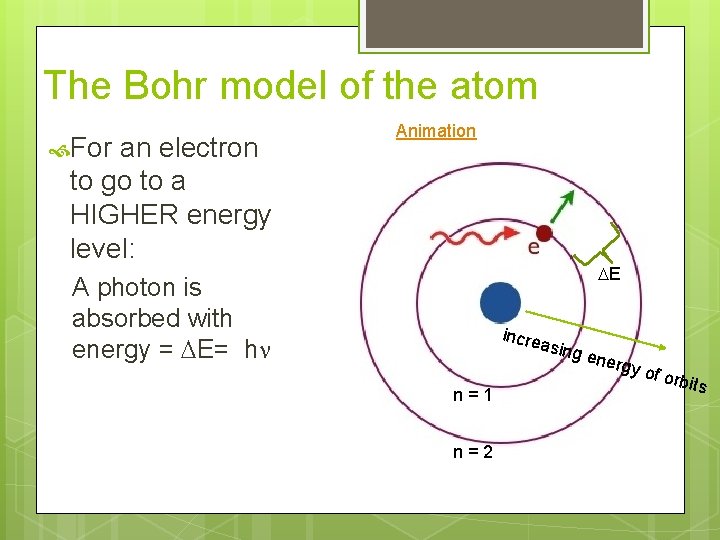
The Bohr model of the atom For an electron to go to a HIGHER energy level: Animation DE A photon is absorbed with energy = DE= hn incre asing n=1 n=2 ener gy of o rbits
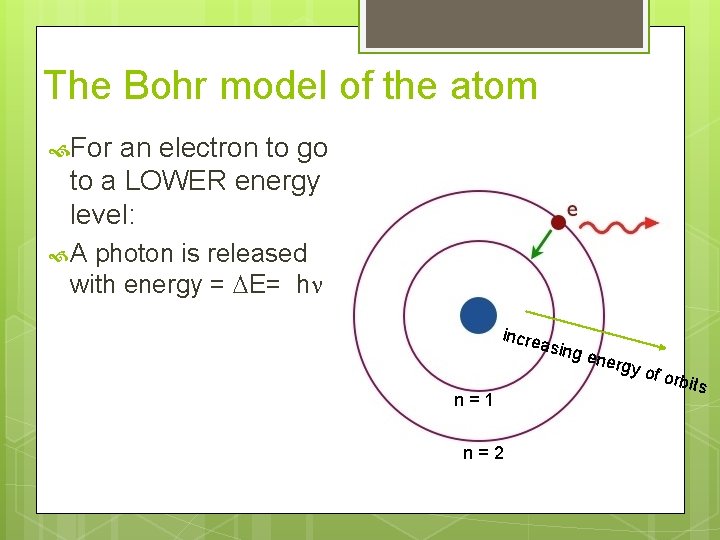
The Bohr model of the atom For an electron to go to a LOWER energy level: A photon is released with energy = DE= hn incre asing n=1 n=2 ener gy of o rbits

Electron energy levels
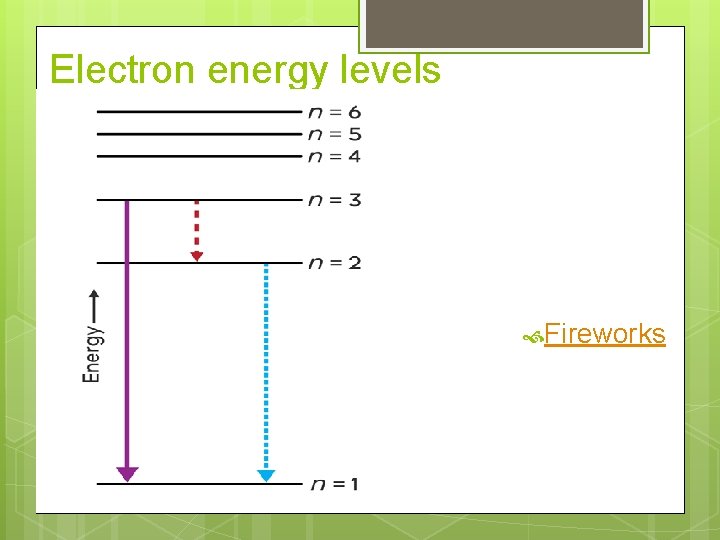
Electron energy levels Fireworks

Problems with the Bohr model Why don’t the electrons just collapse into the nucleus? Electrons traveling in fixed orbits give off xrays Photo from NSLS, Brookhaven National Laboratory

2009 Nobel Prize in Chemistry Structural and functional studies of the ribosome Venkatraman Ramakrishnan Thomas A. Steitz Ada E. Yonath
- Slides: 21