Bohrs atom model Bohrs atom model The neon

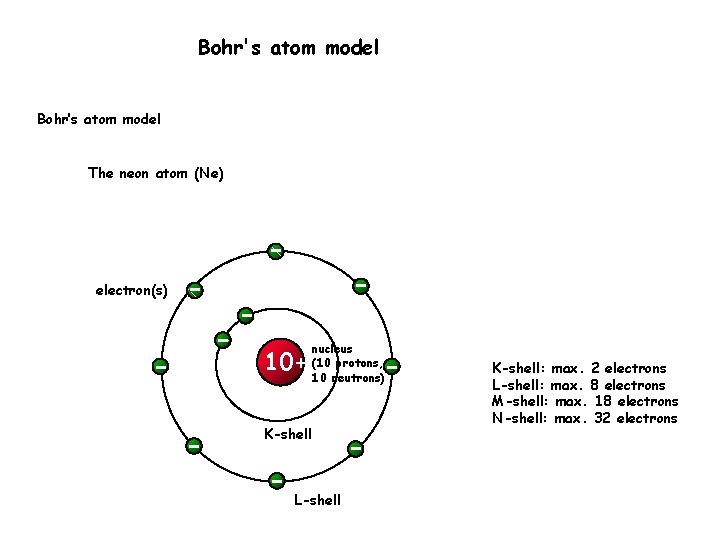
Bohr's atom model Bohr’s atom model The neon atom (Ne) electron(s) 10+ nucleus (10 protons, 10 neutrons) K-shell L-shell K-shell: max. 2 electrons L-shell: max. 8 electrons M-shell: max. 18 electrons N-shell: max. 32 electrons
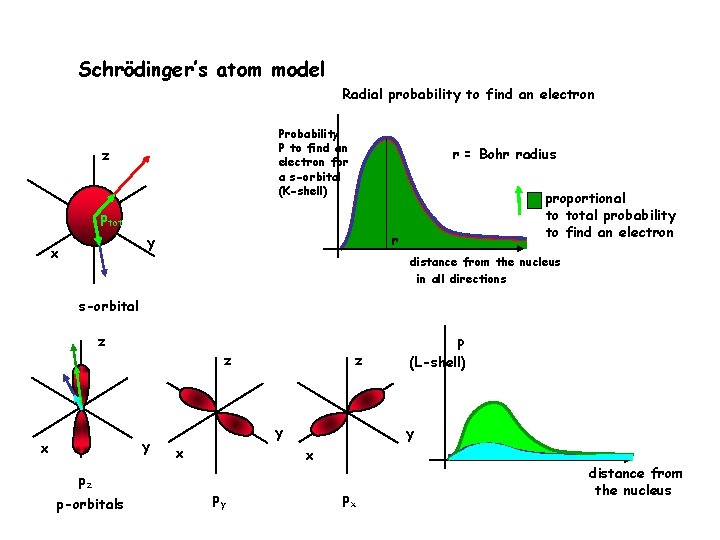
Schrödinger’s atom model Radial probability to find an electron Probability P to find an electron for a s-orbital (K-shell) z r = Bohr radius proportional to total probability to find an electron ptot r y x distance from the nucleus in all directions s-orbital z z y x pz p-orbitals z y x py P (L-shell) px distance from the nucleus
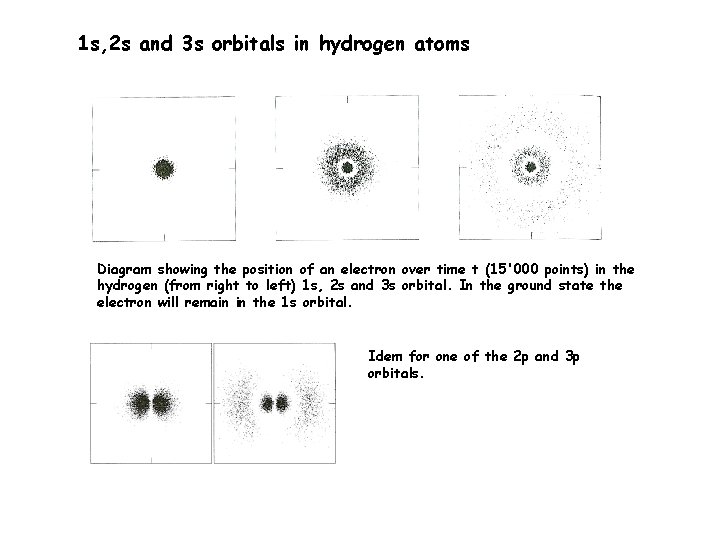
1 s, 2 s and 3 s orbitals in hydrogen atoms Diagram showing the position of an electron over time t (15'000 points) in the hydrogen (from right to left) 1 s, 2 s and 3 s orbital. In the ground state the electron will remain in the 1 s orbital. Idem for one of the 2 p and 3 p orbitals.
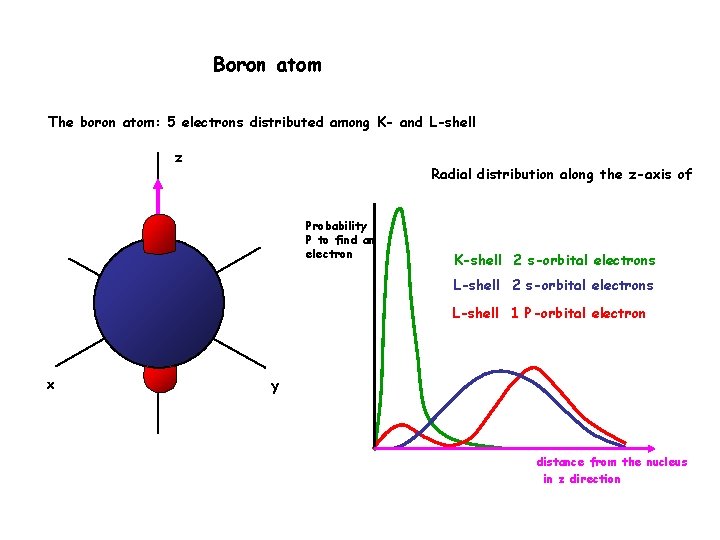
Boron atom The boron atom: 5 electrons distributed among K- and L-shell z Radial distribution along the z-axis of Probability P to find an electron K-shell 2 s-orbital electrons L-shell 1 P-orbital electron x y distance from the nucleus in z direction
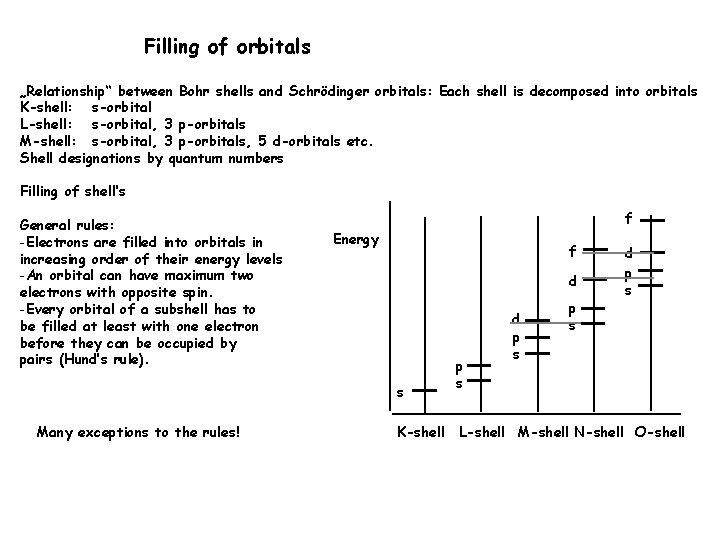
Filling of orbitals „Relationship“ between Bohr shells and Schrödinger orbitals: Each shell is decomposed into orbitals K-shell: s-orbital L-shell: s-orbital, 3 p-orbitals M-shell: s-orbital, 3 p-orbitals, 5 d-orbitals etc. Shell designations by quantum numbers Filling of shell’s General rules: -Electrons are filled into orbitals in increasing order of their energy levels -An orbital can have maximum two electrons with opposite spin. -Every orbital of a subshell has to be filled at least with one electron before they can be occupied by pairs (Hund’s rule). f Energy f d s Many exceptions to the rules! K-shell p s d p s L-shell M-shell N-shell O-shell
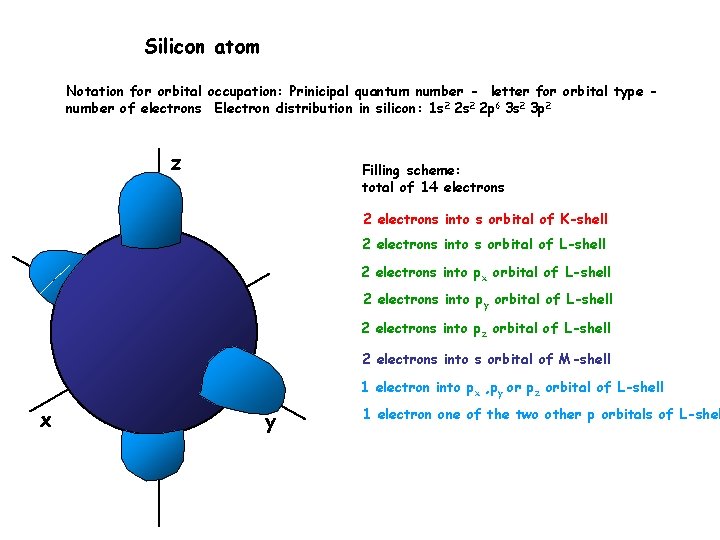
Silicon atom Notation for orbital occupation: Prinicipal quantum number - letter for orbital type number of electrons Electron distribution in silicon: 1 s 2 2 p 6 3 s 2 3 p 2 z Filling scheme: total of 14 electrons 2 electrons into s orbital of K-shell 2 electrons into s orbital of L-shell 2 electrons into px orbital of L-shell 2 electrons into py orbital of L-shell 2 electrons into pz orbital of L-shell 2 electrons into s orbital of M-shell 1 electron into px , py or pz orbital of L-shell x y 1 electron one of the two other p orbitals of L-shel

Electronegativity is a measure of the ability of an atom or molecule to attract respectively to give off an electron. Pauling's electronegativity scale is given in dimensionless units and ranges from 0 to 4: EN < 1. 9: element gives up electrons, metallic character (except noble metals) EN > 2. 1: element acquires electrons, non-metallic character 1. 9 > EN > 2. 1: amphoteric character

Covalent bonds I Covalent bonds: sharing of electrons between atoms = overlap of orbitals. Only between atoms with small �E (Electronegativity difference), perfect covalent bond only when �E = 0 => between like atoms Example: Fluorine electron configuration: 1 s 22 s 2 p 5 2 p - orbitals of fluorine red: occupied by two electrons blue: occupied by one electron Overlapping orbitals: Overlap of the two single occupied 2 p-orbitals (= sharing of the single electrons) molecular orbitals Hybrid orbitals with axial symmetry: �� - bonds Directionality is the major difference of covalent bonds compared to ionic bonds. Most bonds are mixed in character. The covalent contribution is given by: CC(%)= 100 - (16 E + 3. 5 E 2)
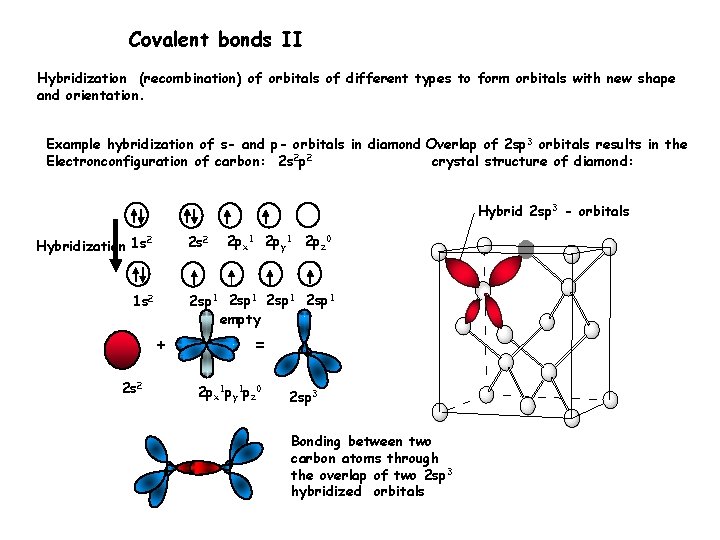
Covalent bonds II Hybridization (recombination) of orbitals of different types to form orbitals with new shape and orientation. Example hybridization of s- and p- orbitals in diamond Overlap of 2 sp 3 orbitals results in the Electronconfiguration of carbon: 2 s 2 p 2 crystal structure of diamond: Hybrid 2 sp 3 - orbitals 2 s 2 2 Hybridization 1 s 2 sp 1 empty 1 s 2 + 2 s 2 2 px 1 2 py 1 2 pz 0 = 2 px 1 py 1 pz 0 2 sp 3 Bonding between two carbon atoms through the overlap of two 2 sp 3 hybridized orbitals
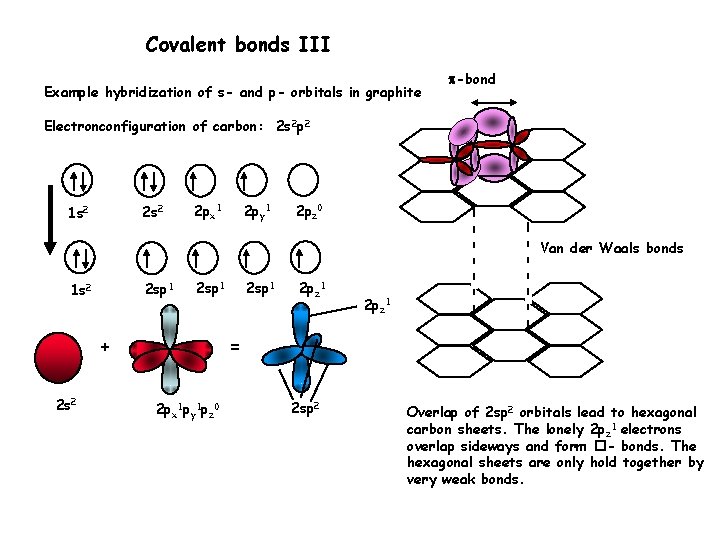
Covalent bonds III Example hybridization of s- and p- orbitals in graphite -bond Electronconfiguration of carbon: 2 s 2 p 2 2 s 2 1 s 2 2 px 1 2 py 1 2 pz 0 Van der Waals bonds 2 sp 1 1 s 2 2 sp 1 + 2 s 2 2 sp 1 2 pz 1 = 2 px 1 py 1 pz 0 2 sp 2 Overlap of 2 sp 2 orbitals lead to hexagonal carbon sheets. The lonely 2 pz 1 electrons overlap sideways and form �- bonds. The hexagonal sheets are only hold together by very weak bonds.
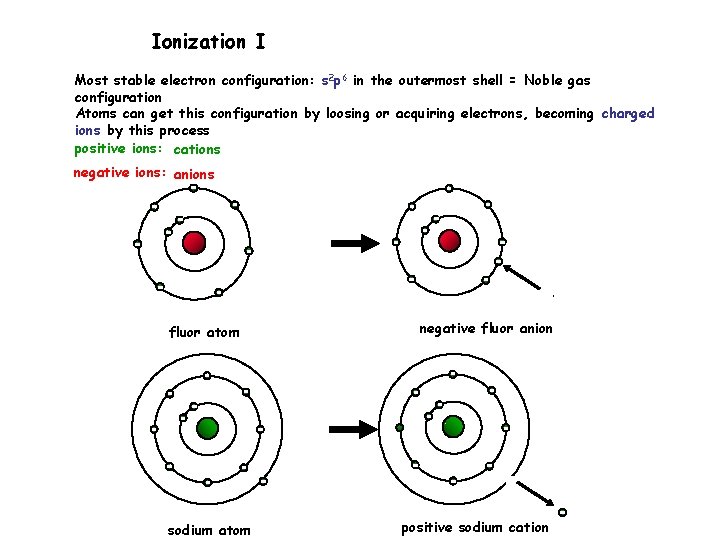
Ionization I Most stable electron configuration: s 2 p 6 in the outermost shell = Noble gas configuration Atoms can get this configuration by loosing or acquiring electrons, becoming charged ions by this process positive ions: cations negative ions: anions fluor atom sodium atom negative fluor anion positive sodium cation

Ionization II Tendency for ionization �� ionization potential �� electron configuration s 2 p 6: most stable configuration, “noble gas” configuration => high ionization potential Tendency to give off an electron increases when: - no other electron was added or subtracted before - the valence shell of the ion gets closer to a s 2 p 6 configuration - the electron is in a low energy orbital, far from the nucleus Examples: Element electron configuration 1 st ionization potential Lithium Caesium 1 s 22 s 1 1 s 22 s 2 p 63 s 2 p 6 d 104 s 2 p 6 d 10 5 s 2 p 66 s 1 5. 39 e. V 3. 89 e. V Neon Krypton 1 s 22 s 2 p 63 s 2 p 6 d 104 s 2 p 6 21. 56 e. V 14. 00 e. V Ability to acquire a valence electron: Electron affinity. Positive affinities = release of energy Example Chlorine Electron affinity 1 s 22 s 2 p 63 s 2 p 5 3. 61 e. V
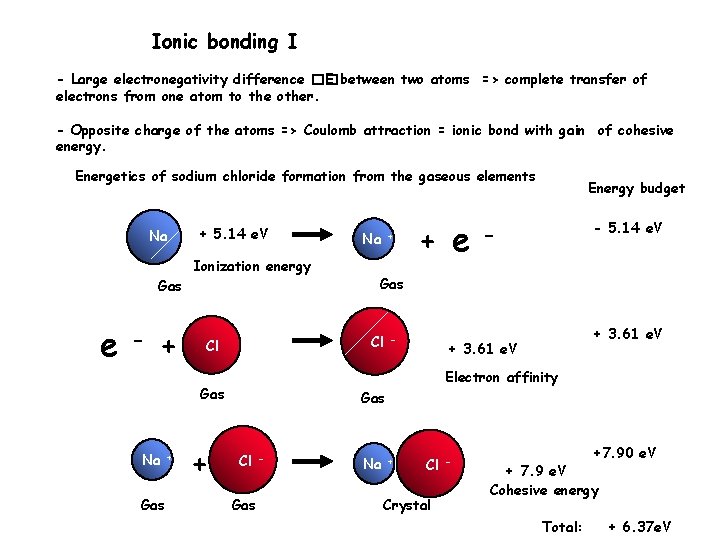
Ionic bonding I - Large electronegativity difference �� E between two atoms => complete transfer of electrons from one atom to the other. - Opposite charge of the atoms => Coulomb attraction = ionic bond with gain of cohesive energy. Energetics of sodium chloride formation from the gaseous elements Na + 5. 14 e. V Ionization energy Gas e - + Gas + + e Na+ - 5. 14 e. V - Gas Cl Cl - + 3. 61 e. V Electron affinity Gas Na Na Energy budget Gas Cl Gas - Na + Cl Crystal - +7. 90 e. V + 7. 9 e. V Cohesive energy Total: + 6. 37 e. V
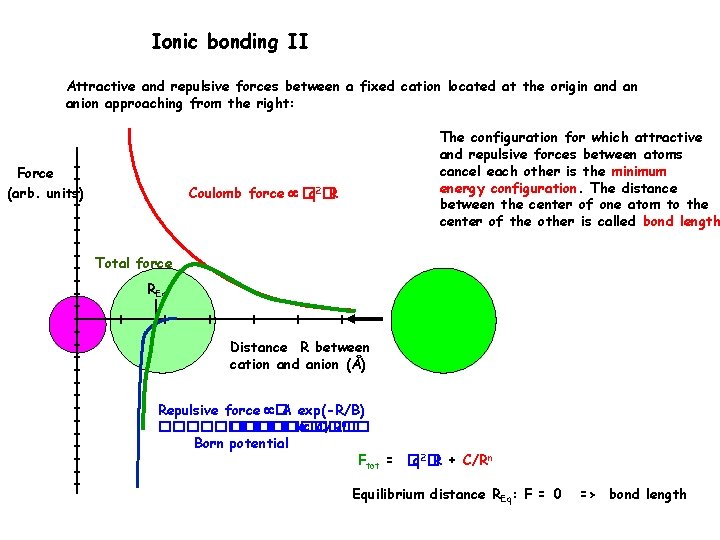
Ionic bonding II Attractive and repulsive forces between a fixed cation located at the origin and an anion approaching from the right: Force (arb. units) The configuration for which attractive and repulsive forces between atoms cancel each other is the minimum energy configuration. The distance between the center of one atom to the center of the other is called bond length Coulomb force �q 2�R Total force REq Distance R between cation and anion (Å) Repulsive force �A exp(-R/B) ���������� �C/Rn� Born potential Ftot = �q 2�R + C/Rn Equilibrium distance REq: F = 0 => bond length
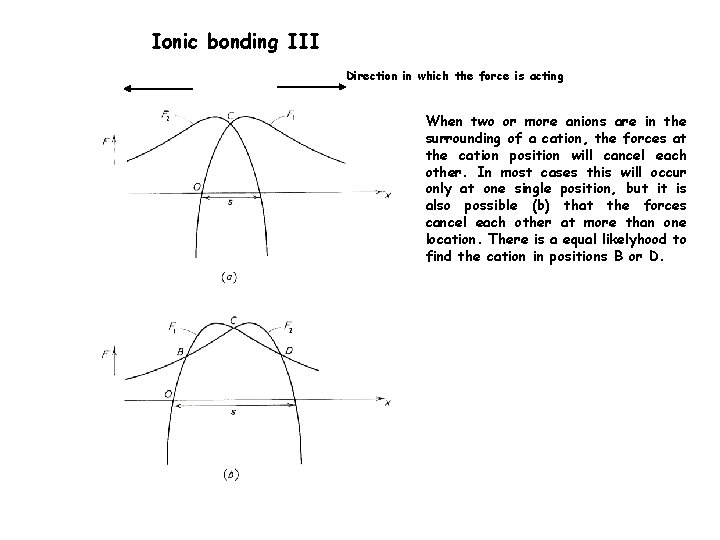
Ionic bonding III Direction in which the force is acting When two or more anions are in the surrounding of a cation, the forces at the cation position will cancel each other. In most cases this will occur only at one single position, but it is also possible (b) that the forces cancel each other at more than one location. There is a equal likelyhood to find the cation in positions B or D.
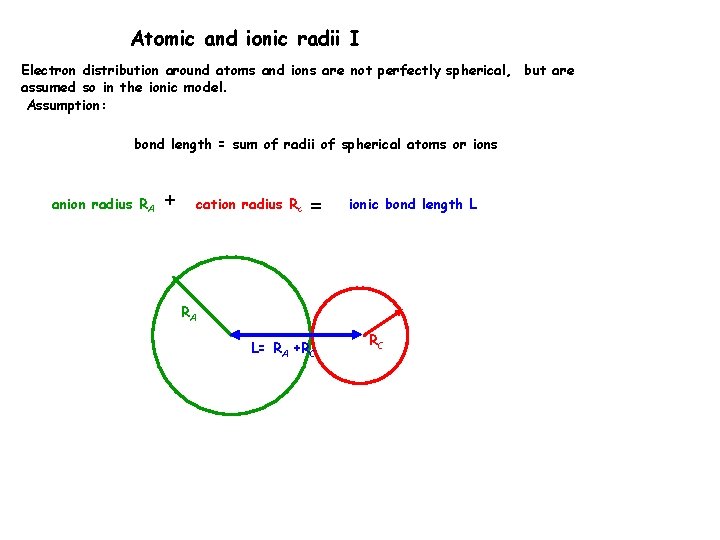
Atomic and ionic radii I Electron distribution around atoms and ions are not perfectly spherical, but are assumed so in the ionic model. Assumption: bond length = sum of radii of spherical atoms or ions anion radius RA + cation radius Rc = ionic bond length L RA L= RA +RC RC
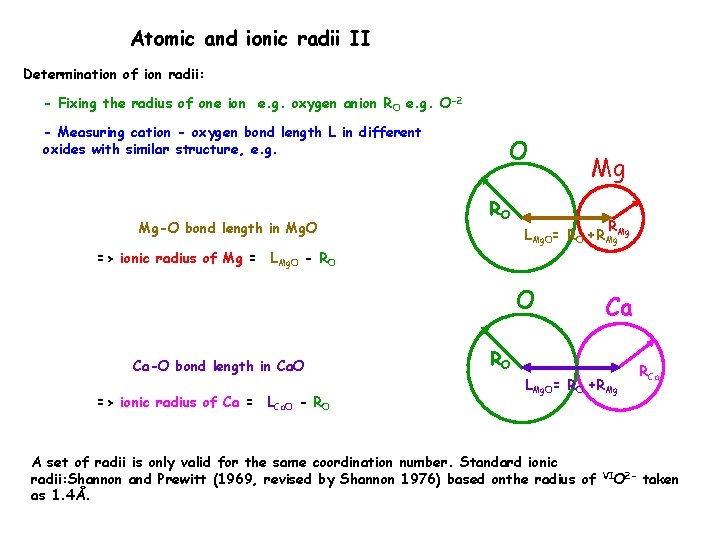
Atomic and ionic radii II Determination of ion radii: - Fixing the radius of one ion e. g. oxygen anion RO e. g. O-2 - Measuring cation - oxygen bond length L in different oxides with similar structure, e. g. Mg-O bond length in Mg. O O RO Mg R LMg. O= RO +RMg. Mg => ionic radius of Mg = LMg. O - RO O Ca-O bond length in Ca. O => ionic radius of Ca = LCa. O - RO Ca RO LMg. O= RO +RMg A set of radii is only valid for the same coordination number. Standard ionic radii: Shannon and Prewitt (1969, revised by Shannon 1976) based onthe radius of as 1. 4Å. VIO 2 - RCa taken
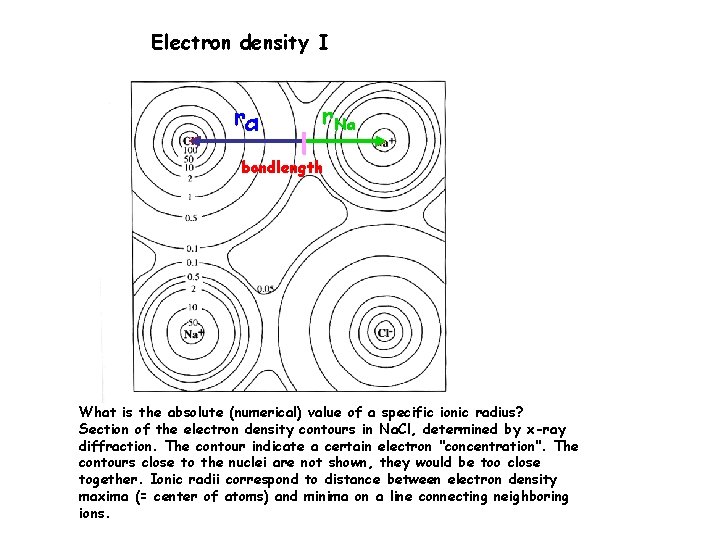
Electron density I r. Cl r. Na bondlength What is the absolute (numerical) value of a specific ionic radius? Section of the electron density contours in Na. Cl, determined by x-ray diffraction. The contour indicate a certain electron "concentration". The contours close to the nuclei are not shown, they would be too close together. Ionic radii correspond to distance between electron density maxima (= center of atoms) and minima on a line connecting neighboring ions.
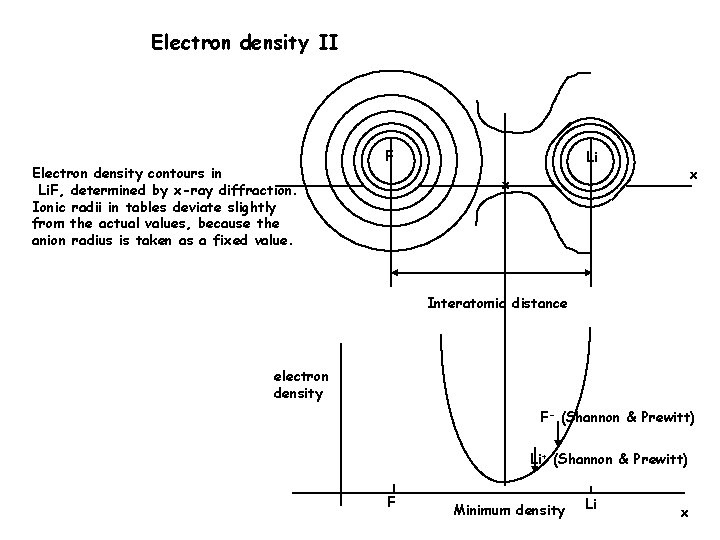
Electron density II Electron density contours in Li. F, determined by x-ray diffraction. Ionic radii in tables deviate slightly from the actual values, because the anion radius is taken as a fixed value. F Li x x Interatomic distance electron density F- (Shannon & Prewitt) Li+ (Shannon & Prewitt) F Minimum density Li x
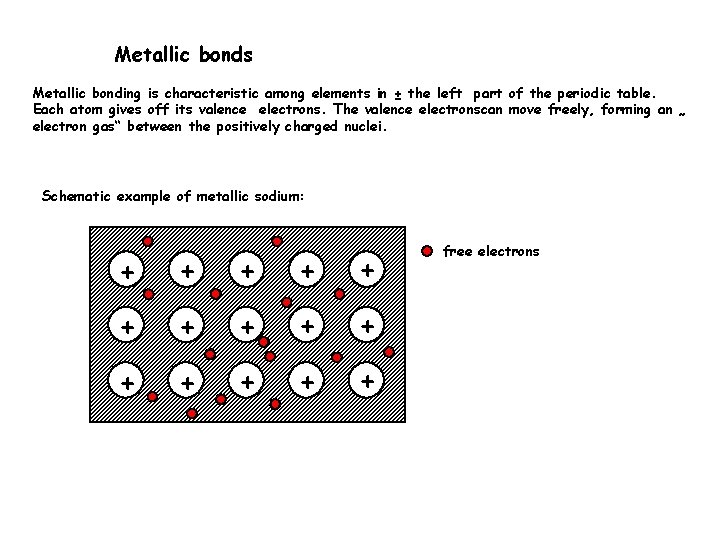
Metallic bonds Metallic bonding is characteristic among elements in ± the left part of the periodic table. Each atom gives off its valence electrons. The valence electronscan move freely, forming an „ electron gas“ between the positively charged nuclei. Schematic example of metallic sodium: + + + + free electrons
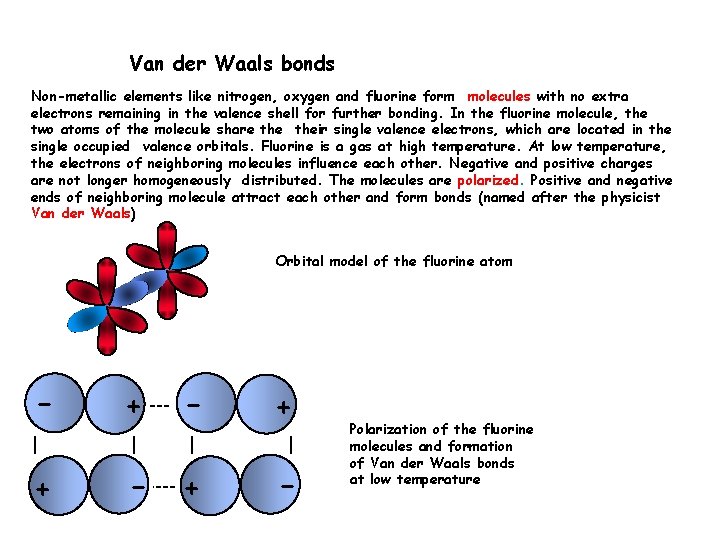
Van der Waals bonds Non-metallic elements like nitrogen, oxygen and fluorine form molecules with no extra electrons remaining in the valence shell for further bonding. In the fluorine molecule, the two atoms of the molecule share their single valence electrons, which are located in the single occupied valence orbitals. Fluorine is a gas at high temperature. At low temperature, the electrons of neighboring molecules influence each other. Negative and positive charges are not longer homogeneously distributed. The molecules are polarized. Positive and negative ends of neighboring molecule attract each other and form bonds (named after the physicist Van der Waals) Orbital model of the fluorine atom - + + - Polarization of the fluorine molecules and formation of Van der Waals bonds at low temperature
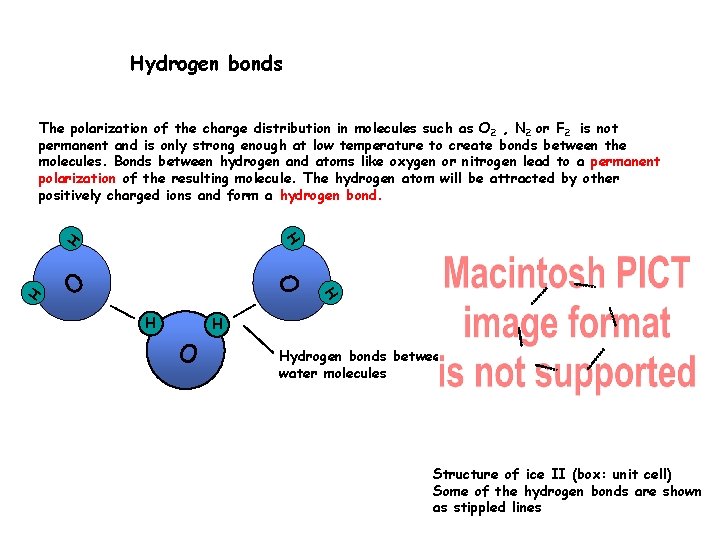
Hydrogen bonds The polarization of the charge distribution in molecules such as O 2 , N 2 or F 2 is not permanent and is only strong enough at low temperature to create bonds between the molecules. Bonds between hydrogen and atoms like oxygen or nitrogen lead to a permanent polarization of the resulting molecule. The hydrogen atom will be attracted by other positively charged ions and form a hydrogen bond. O H H H O Hydrogen bonds between water molecules Structure of ice II (box: unit cell) Some of the hydrogen bonds are shown as stippled lines
- Slides: 23