Bohr Rutherford Atomic Model Neils Bohr improved Rutherfords

Bohr – Rutherford Atomic Model Neils Bohr improved Rutherford’s model by placing electrons in distinct energy levels or “shells” rather than an electron “cloud”

Bohr – Rutherford Atomic Model: ¡ Bohr & Rutherford combined their theories to create the current simplified atomic model ¡ The atom is made up of three subatomic particles: l Protons l Electrons l Neutrons
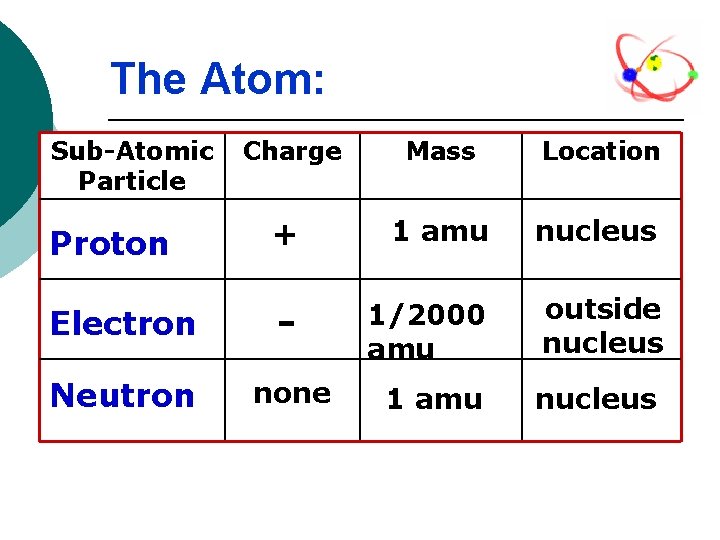
The Atom: Sub-Atomic Particle Charge Mass Location Proton + 1 amu nucleus Electron - 1/2000 amu outside nucleus Neutron none 1 amu nucleus

The Atom: electron neutron nucleus proton education. jlab. org/ qa/atom_model. html

Nucleus: ¡ ¡ ¡ Contains protons & neutrons Overall positive charge (+) Contains nearly all mass (dense) Electrons: ¡ ¡ ¡ Located outside nucleus Easily moved, lost or gained Negative charge (-)

The Atom – Important Info: ¡ ¡ are neutral, Every Since atoms has a symbol & two the number of protons numbers associated with it. (+) equals the number of electrons (-) Ex. C 6 12 Atomic Number • The number of protons • The smaller number Mass Number • The mass of an atom • The number of protons & neutrons • The bigger number

For the following atom, state the… i) mass number 16 ii) atomic number 8 iii) # of protons 8 iv) # of electrons 8 v) # of neutrons 16 – 8 = 8 O 8 16

For the following atom, state the… i) mass number 23 ii) atomic number 11 iii) # of protons 11 iv) # of electrons 11 v) # of neutrons 23 – 11 = 12 Na 11 23

For the following atom, state the… i) mass number 40 ii) atomic number 18 iii) # of protons 18 iv) # of electrons 18 v) # of neutrons 40 – 18 = 22 Ar 18 40

For the following atom, state the… i) mass number 64 ii) atomic number 29 iii) # of protons 29 iv) # of electrons 29 v) # of neutrons 64 – 29 = 35 Cu 29 64

For the following atom, which statement is TRUE? 15 A) The atomic number is 31. B) The atom has 15 neutrons. C) The atom has 16 electrons. D) The atom has 15 protons & 15 electrons. P 31

Given a neutral atom, which statement is TRUE? A) An atom always has the same number of electrons as neutrons. B) Electrons & neutrons are found in the nucleus. C) The atomic number states the number of neutrons. D) An atom has the same number of electrons as protons.

¡ Atoms are made up of: l l l ¡ ¡ Protons Electrons Neutrons Protons & Neutrons are located in the nucleus Electrons are located outside the nucleus

¡ ¡ ¡ An atom’s atomic number is equal to the number of protons (smaller number) An atom is neutral, therefore the number of protons & electrons are equal An atom’s mass number is equal to the protons & neutrons together (bigger number)

Activities: ¡ Physical Science Worksheet #7 Once you’ve completed the worksheet, continue with the rest of the lesson

Electrons: ¡ ¡ Recall, in the Bohr-Rutherford atomic model, electrons are located outside the nucleus Electrons are organized into: Electron shells or l Energy levels or l Orbitals l Be familiar with all the terms, especially the first two

*Simplified – Electrons: 3 shell holds 18 electrons rd ¡ 4 th shell holds 32 electrons Each electron shell differs in size & the number of electrons it can hold (capacity) Electron Shell 1 st Capacity 2 electrons 2 nd 8 electrons 3 rd 8 electrons* 4 th 2 electrons* Shows maximum # of electrons in each shell Shells can be partially filled or empty

Bohr – Rutherford Diagram: ¡ ¡ Is a picture of an atom, showing its protons, electrons & neutrons. Ex. Li 3 7 Protons = 3 Electrons= 3 Neutrons= 4 3 P+ 4 N 0 2 e- 1 e-

Draw a Bohr-Rutherford diagram for: F 9 19 Protons = 9 Electrons= 9 Neutrons= 10 9 P+ 10 N 0 2 e- 7 e-

Draw a Bohr-Rutherford diagram for: B 5 Protons = 11 5 Electrons= 5 Neutrons= 6 5 P+ 6 N 0 2 e- 3 e-

Draw a Bohr-Rutherford diagram for: Mg 12 Protons = 24 Electrons= 12 12 Neutrons= 12 12 P+ 12 N 0 2 e- 8 e- 2 e-

Draw a Bohr-Rutherford diagram for: K 19 Protons = 39 Electrons= 19 19 Neutrons= 20 19 P+ 20 N 0 2 e- 8 e- 1 e-

Which of the following diagrams best represents the Bohr-Rutherford model of… 4 Be 9 A) C) P+ 4 3 e 5 N 0 2 e. B) 4 P+ 2 e- 2 e 5 N 0 5 P+ 4 N 0 2 e-

Draw a Bohr-Rutherford diagram for each of the following atoms: P 15 31 15 P+ 8 e 16 N 0 2 e- 5 e- N 7 14 Al 13 27 7 P+ 8 e 5 e- 13 P+ 7 N 0 2 e 14 N 02 e- 3 e-

The Neutron ¡ The Rutherford-Bohr model of the atom did not answer the question: “What holds the nucleus together given that it is made up of entirely of positive protons? ”

The Neutron In 1932, James Chadwick discovered that a third sub-atomic particle called the neutron holds the protons in the nucleus together. ¡ Neutrons are located in the nucleus and have no charge…they are neutral. ¡
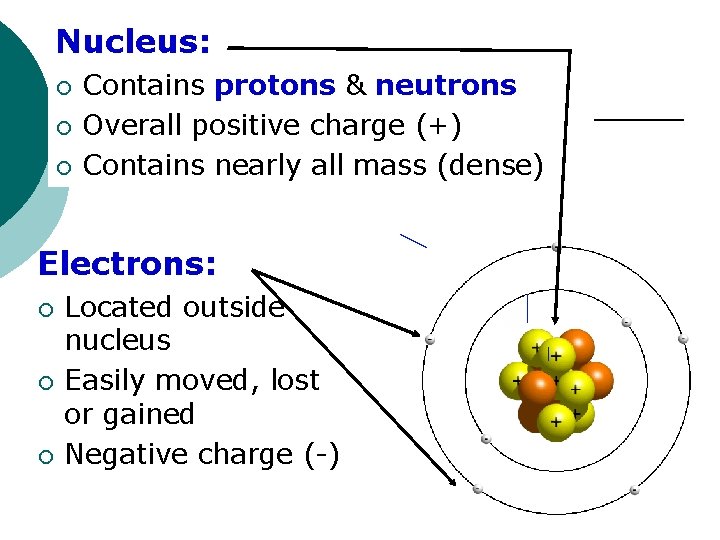
Nucleus: ¡ ¡ ¡ Contains protons & neutrons Overall positive charge (+) Contains nearly all mass (dense) Electrons: ¡ ¡ ¡ Located outside nucleus Easily moved, lost or gained Negative charge (-)

Electrons are located outside the nucleus in electron shells ¡ In the… ¡ l l ¡ 1 st shell – 2 electrons 2 nd shell – 8 electrons 3 rd shell – 8 electrons* 4 th shell – 2 electrons* In Bohr-Rutherford diagrams: l l Find the number of Protons, Electrons & Neutrons Place Protons & Neutrons in nucleus Place Electrons in appropriate shells

Activities: ¡ Physical Science Worksheet #8
- Slides: 29