BOHR MODELS Science 9 EUREKA VIDEO Eureka Episode

BOHR MODELS Science 9

EUREKA VIDEO Eureka_ Episode 23 – Electrons https: //www. youtube. com/watch? v=k. Hup. Oggh 3 U 0

NIELS BOHR, FAMOUS SCIENTIST Niels Bohr (1885 - 1962) He studied gaseous samples of atoms, which were made to glow by passing an electric current through them. Based on his observations, Bohr proposed that electrons surround the nucleus in specific “energy levels” or “shells. ”
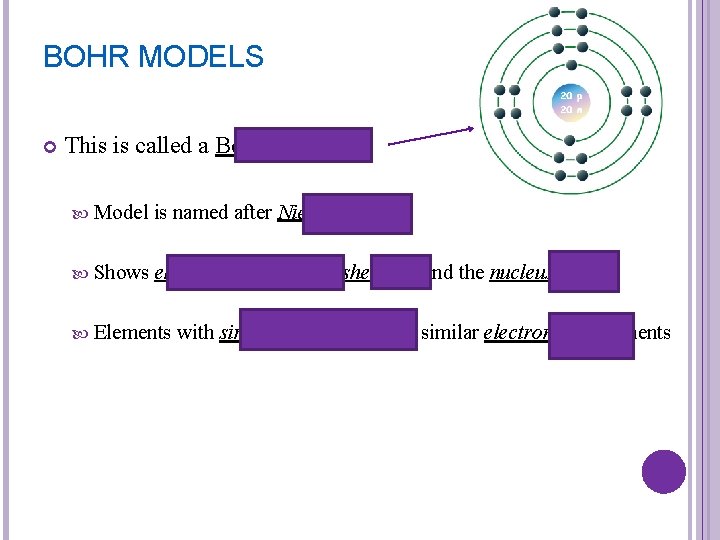
BOHR MODELS This is called a Bohr model Model is named after Niels Bohr Shows electrons arranged in shells around the nucleus Elements with similar properties have similar electron arrangements

BOHR MODELS Like rows of seats in a theatre, each shell around the nucleus can only hold a certain number of electrons before it is “full”
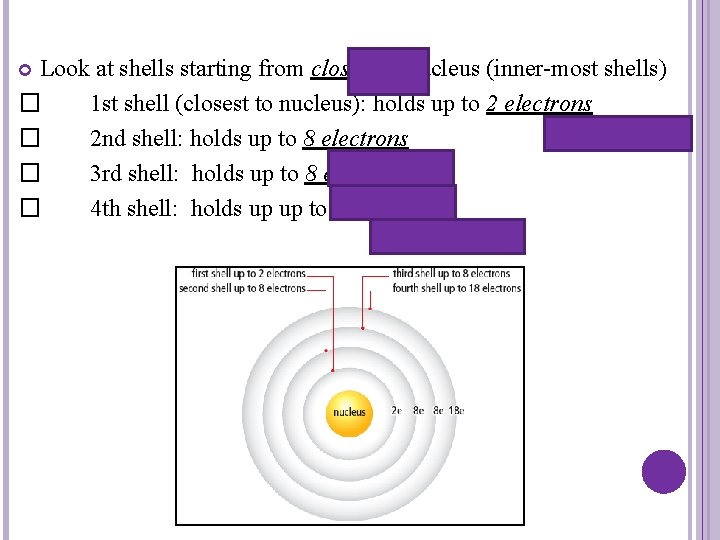
Look at shells starting from closest to nucleus (inner-most shells) � 1 st shell (closest to nucleus): holds up to 2 electrons � 2 nd shell: holds up to 8 electrons � 3 rd shell: holds up to 8 electrons � 4 th shell: holds up up to 18 electrons
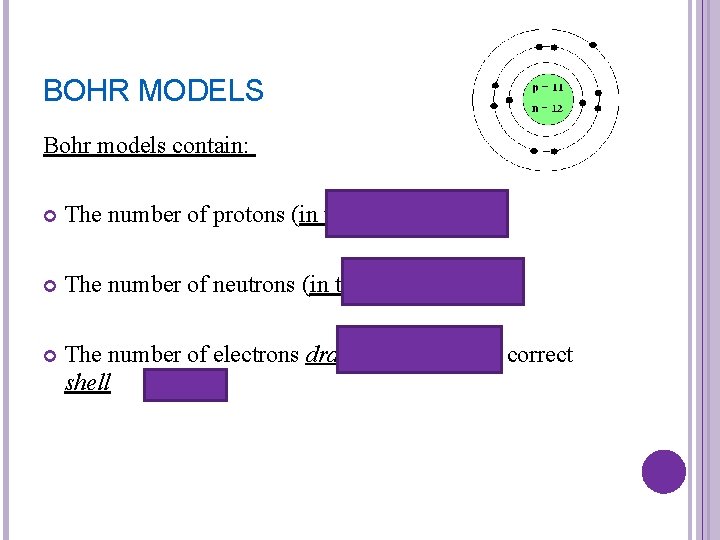
BOHR MODELS Bohr models contain: The number of protons (in the nucleus) The number of neutrons (in the nucleus) The number of electrons drawn as dots in the correct shell
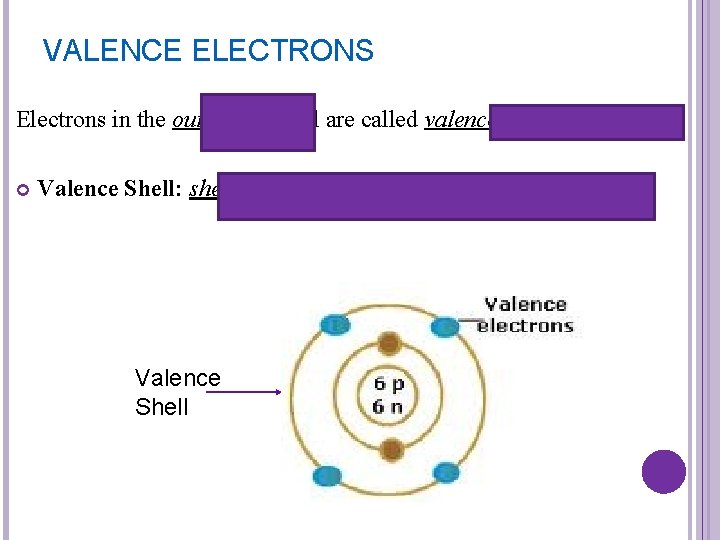
VALENCE ELECTRONS Electrons in the outermost shell are called valence electrons Valence Shell: shell that contains outermost electrons Valence Shell
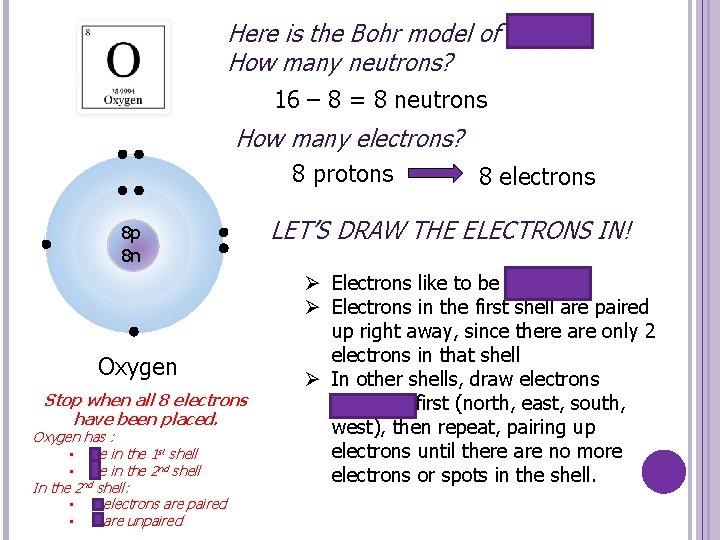
Here is the Bohr model of oxygen. How many neutrons? 16 – 8 = 8 neutrons How many electrons? 8 protons 8 p 8 n Oxygen Stop when all 8 electrons have been placed. Oxygen has : • 2 e in the 1 st shell • 6 e in the 2 nd shell In the 2 nd shell: • 4 electrons are paired • 2 are unpaired 8 electrons LET’S DRAW THE ELECTRONS IN! Ø Electrons like to be paired up Ø Electrons in the first shell are paired up right away, since there are only 2 electrons in that shell Ø In other shells, draw electrons unpaired first (north, east, south, west), then repeat, pairing up electrons until there are no more electrons or spots in the shell.
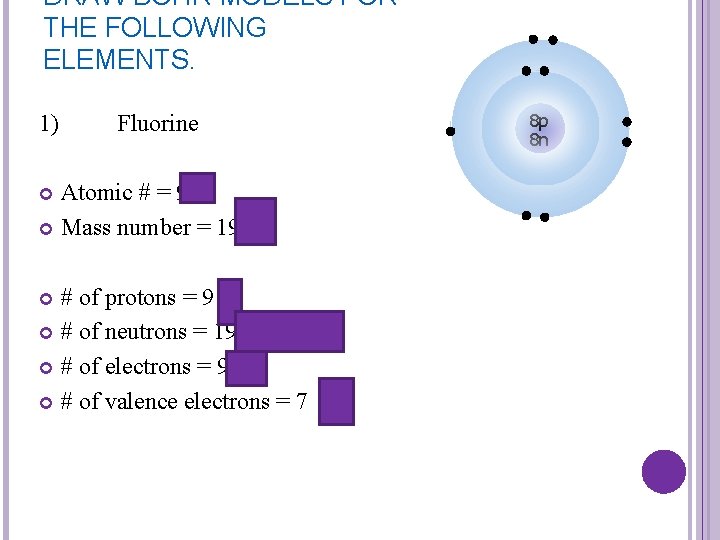
DRAW BOHR MODELS FOR THE FOLLOWING ELEMENTS. 1) Fluorine Atomic # = 9 Mass number = 19 # of protons = 9 # of neutrons = 19 -9 = 10 # of electrons = 9 # of valence electrons = 7 8 p 8 n
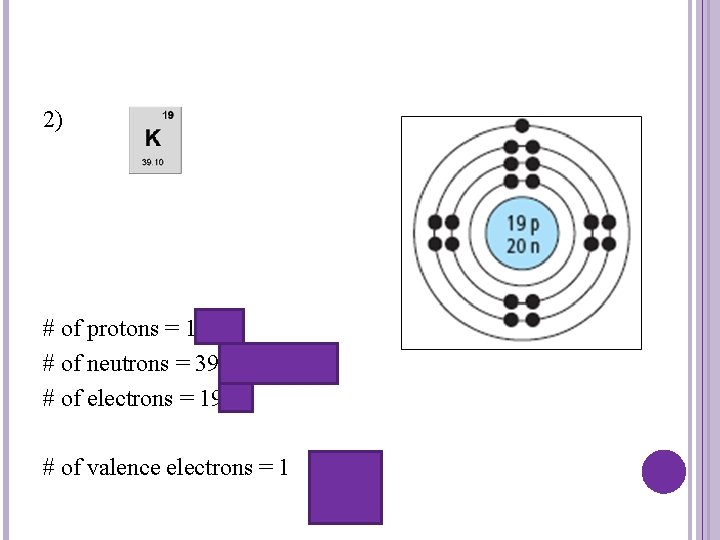
2) # of protons = 19 # of neutrons = 39 -19 = 20 # of electrons = 19 # of valence electrons = 1
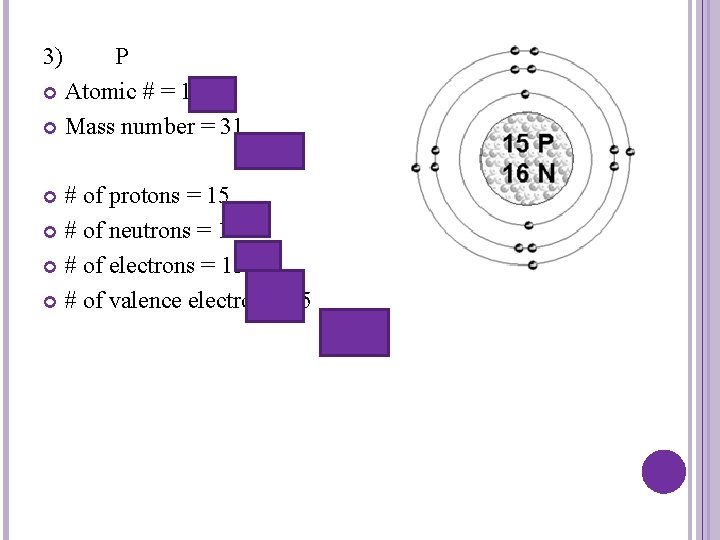
3) P Atomic # = 15 Mass number = 31 # of protons = 15 # of neutrons = 16 # of electrons = 15 # of valence electrons = 5

- Slides: 13