Bohr Models Making a Bohr Model Bohr Model

Bohr Models
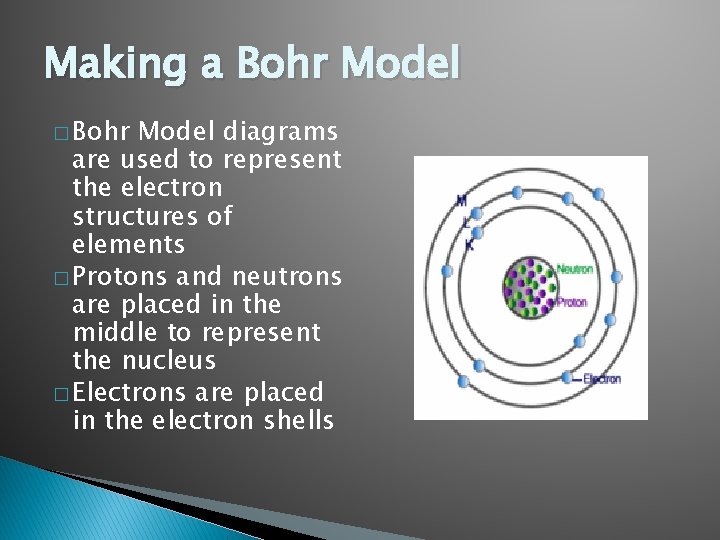
Making a Bohr Model � Bohr Model diagrams are used to represent the electron structures of elements � Protons and neutrons are placed in the middle to represent the nucleus � Electrons are placed in the electron shells

How to Construct Bohr Model Diagrams � Using the example Fluorine, we will work through the steps in creating a Bohr model � Right now, look at your periodic table and find the element Fluorine. � Draw in what Fluorine looks like on the Periodic Table in the box on your sheet

Steps to Make a Bohr Model 1. Determine the number of subatomic particles found in the element ◦ Number of electrons is equal to the number of protons in the neutral atom. This value is equal to the atomic number ◦ Number of neutrons is calculated using the equation Atomic Mass MINUS Atomic Number

Steps to Make a Bohr Model � Fluorine Example ◦ Number of protons = 9 ◦ Number of electrons = 9 ◦ Number of neutrons = 19 – 9 = 10

Steps to Make a Bohr Model 2. Draw a circle for the nucleus with the correct number of and symbol for both protons and neutrons. ◦ Letter symbol for protons: p ◦ Letter symbol for neutrons: n Fluorine example p 9 n 10

Steps to Make a Bohr Model 3. Draw the correct number of electron shells ◦ First ring has a maximum of 2 electrons ◦ Second ring has a maximum of 8 electrons ◦ Third ring has a maximum of 8 electrons *Electron shells must be filled to their maximum before another ring is added

Steps to Make a Bohr Model FLUORINE EXAMPLE � Fluorine has 9 electrons � The first electron shell has 2 electrons � 9 minus 2 = 7 � The second electron shell will have 7 electrons ** The electrons in the outer shell are referred to as valence electrons.

Steps to Make a Bohr Model 4. Represent electrons as dots and position electrons in shells. � The electron shells are also referred to as electron orbitals � The electrons will fill the orbit of the lowest energy level first (i. e. first electron shell closest to the nucleus). The first shell can hold a maximum of 2 electrons

Steps to Make a Bohr Model � An electron shell must be filled with its maximum number of electrons before any additional electron shells can be filled � Electrons should be distributed evenly and neatly throughout the shell FLUORINE EXAMPLE In the Fluorine atom, the first shell contains 2 electrons and the second contains 7 electrons.

Bohr Model Example
- Slides: 11