Bohr Model Review are horizontal rows Groups are
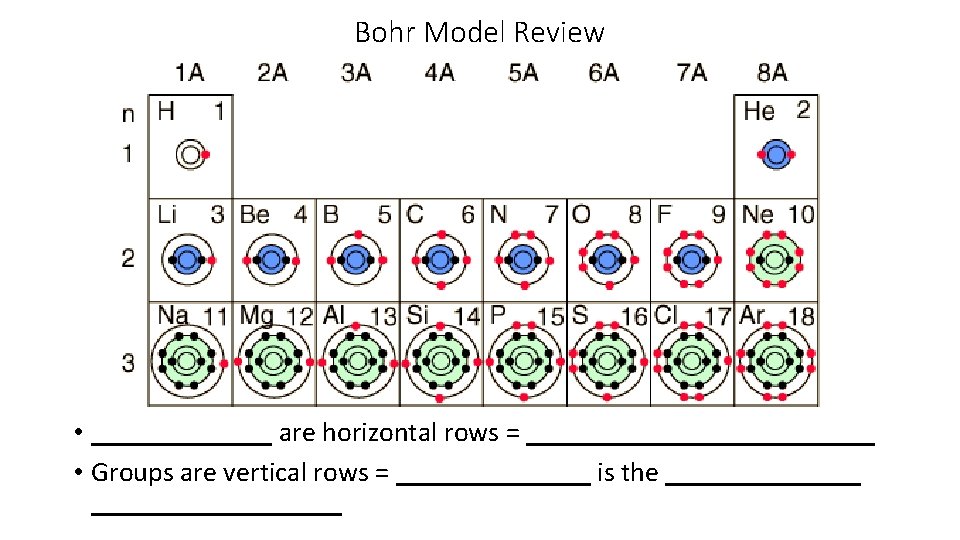
Bohr Model Review • _______ are horizontal rows = _____________ • Groups are vertical rows = _______ is the __________________

Gizmo – Element Builder (Lewis Dot Intro) GROUP NUMBER 1 or 1 A 2 or 2 A 13 or 3 A 14 or 4 A 15 or 5 A 16 or 6 A 17 or 7 A 18 or 8 A # VALENCE ELECTRONS

Lewis Dot Structures • Lewis Dot Diagrams are shown with _______________. The dots represent the ________.

CATION & ANION REVIEW • ___________________________________to become _______ly charged.

Metals vs. Nonmetals • ______ want to _______to become ________. • ________ want to _______ to become ______.
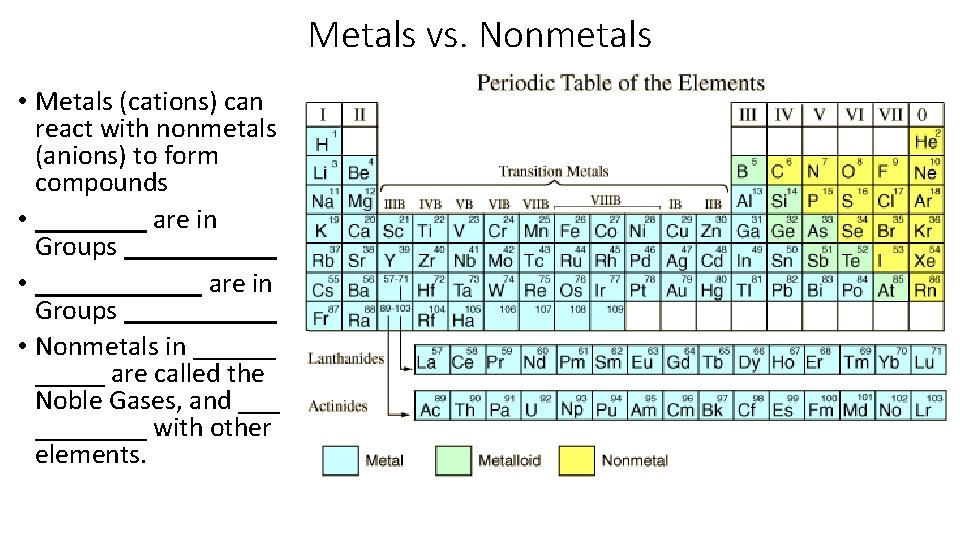
Metals vs. Nonmetals • Metals (cations) can react with nonmetals (anions) to form compounds • ____ are in Groups ______ • ______ are in Groups ______ • Nonmetals in ______ are called the Noble Gases, and ________ with other elements.

The Octet Rule • https: //www. youtube. com/watch? v=4 OKy 782 e. PKM • Elements in Group 1 A, 2 A and 3 A want to lose electrons to achieve an outermost shell of 2 (Lithium and Beryllium only) or 8 valence electrons. Elements in Groups 1 A – 2 A become cations. • Elements in Group 1 A want to lose 1 electron • Elements in Group 2 A want to lose 2 electrons • Elements in Group 3 A want to lose 3 electrons • Elements in Group 5 A, 6 A, 7 A want to gain electrons to achieve an outermost shell of 8 valence electrons. Elements in Groups 5 A – 7 A become anions. • Elements in Group 5 A want to gain 3 electrons • Elements in Group 6 A want to gain 2 electrons • Elements in Group 7 A want to gain 1 electron

VALENCE ELECTRON REVIEW

• When the _____ in ___________lose electrons, they form cations with __________________________. • The __________ from a ________________ is determined by ________________________.

Lewis Model of Bonding • For ionic bonds, this model shows the transfer of electrons from cations to anions. • _______________________________ (TOTAL OF 8 ELECTRONS).

Example Problems Mg Mg Mg Al Al Al Si Si Si P P P S S S Cl Cl Cl
- Slides: 11