Bohr Model In addition to the atomic line

Bohr Model ØIn addition to the atomic line spectra of single electron atoms, there were other successes of the Bohr Model n n X-ray spectra Frank-Hertz experiment 1
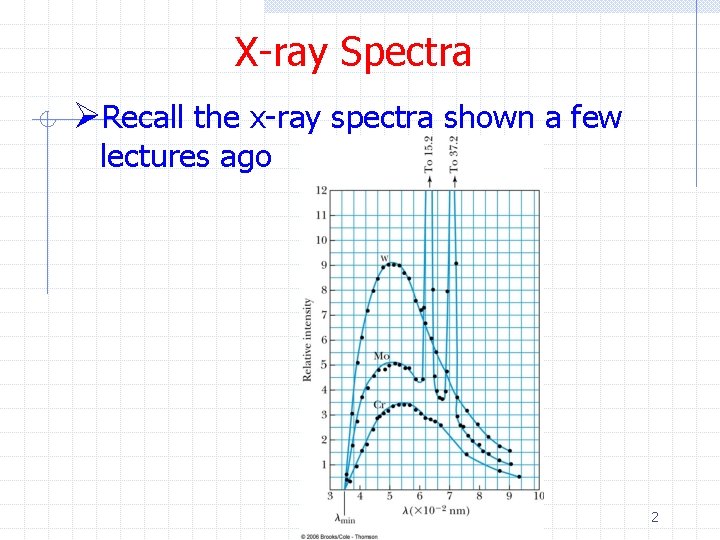
X-ray Spectra ØRecall the x-ray spectra shown a few lectures ago 2

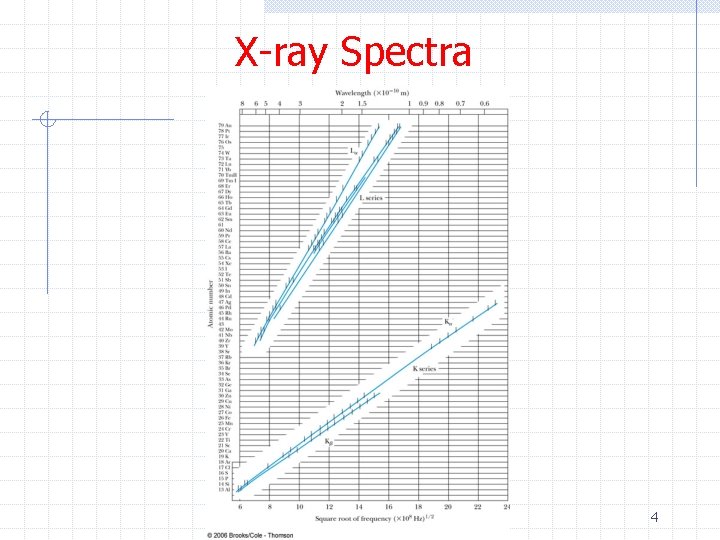
X-ray Spectra Ø Moseley found experimentally that the wavelengths of characteristic x-ray lines of elements followed a regular pattern Ø A similar formula described the L-series x-rays 3
X-ray Spectra 4
X-ray Spectra 5

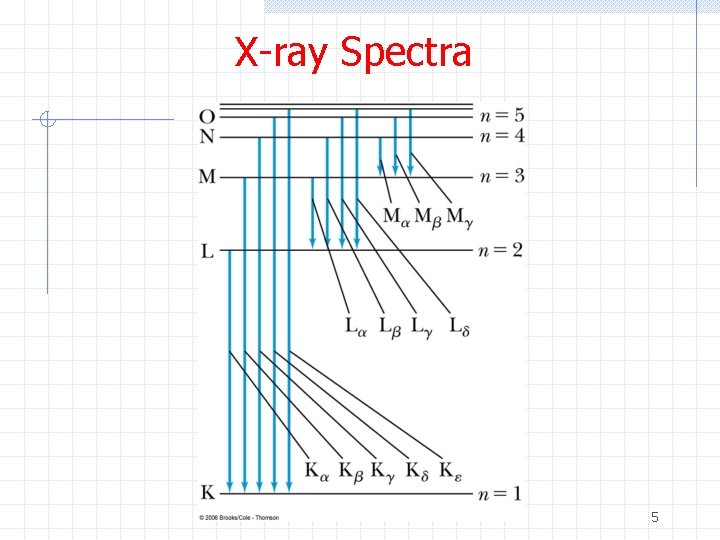
X-ray Spectra Ø Moseley’s law can be easily understood in terms of the Bohr model n If a K (n=1) shell electron is ejected, an electron in the L (n=2) shell will feel an effective charge of Z-1 (Z from the nucleus – 1 electron remaining in the K shell) n We have then for the n=2 to n=1 transition n Exactly agreeing with Moseley’s law 6

X-ray Spectra Ø Applying the Bohr model to the L-series Ø Now there are two electrons in the K shell and several in the L shell thus we might expect a (Z -2 -several)2 dependence n The data show Zeff = (Z - 7. 4) Ø Aside, based on the regular patterns in his data he showed n n The periodic table should be ordered by Z not A Elements with Z=43, 61, and 75 were missing 7
Franck-Hertz Experiment 8
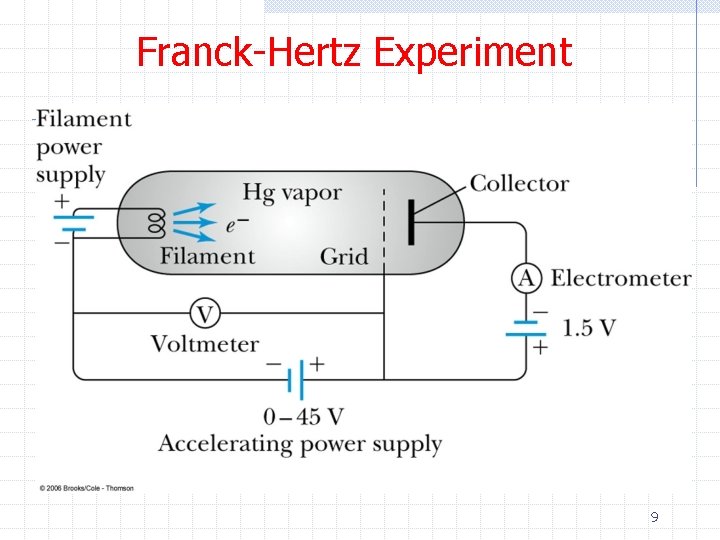
Franck-Hertz Experiment 9
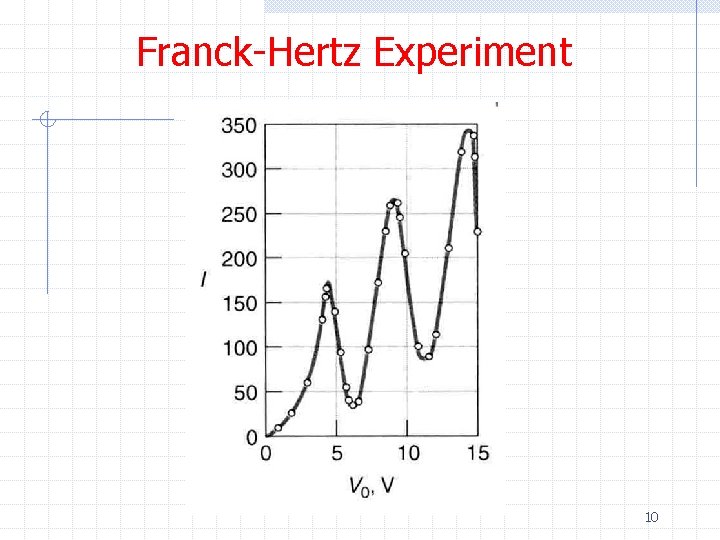
Franck-Hertz Experiment 10

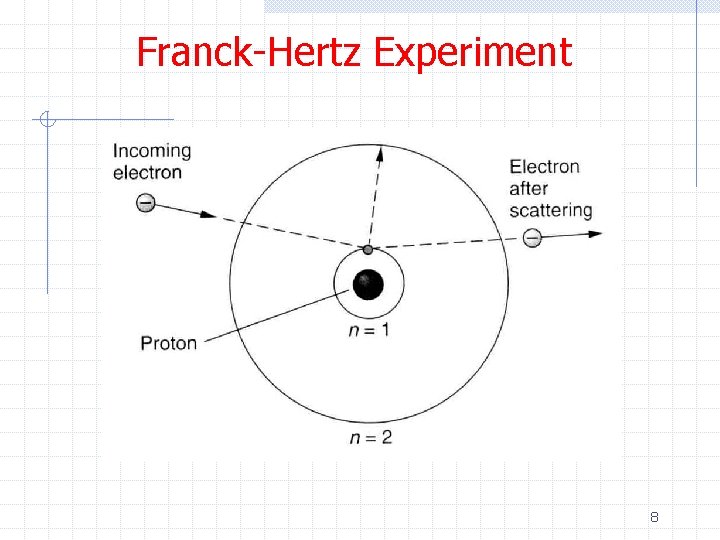
Franck-Hertz Experiment ØThe data show n The current increases with increasing voltage up to V=4. 9 V followed by a sudden drop in current w This is interpreted as a significant fraction of electrons with this energy exciting the Hg atoms and hence losing their kinetic energy w We would expect to see a spectral line associated with de-excitation of 11

Franck-Hertz Experiment n As the voltage is further increased there is again an increase in current up to V=9. 8 V followed by a sharp decrease w This is interpreted as the electron possessing enough kinetic energy to generate two successive excitations from the Hg ground state to the first excited state w Excitations from the ground state to the second excited state are possible but less probable ØThe observation of discrete energy levels was an important confirmation of the Bohr model 12

Frank-Hertz Experiment 13

Correspondence Principle Ø There were difficulties in reconciling the new physics in the Bohr model and classical physics n When does an accelerated charge radiate? Ø Bohr developed a principle to try to bridge the gap n n The predictions of quantum theory must agree with the predictions of classical physics in the limit where the quantum numbers n become large A selection rule holds true over the entire range of quantum numbers n (both small and large n) 14

Correspondence Principle Ø Consider the frequency of emitted radiation by atomic electrons n Classical 15

Correspondence Principle Ø Quantum 16

Wilson-Sommerfeld Quantization ØQuantization appears to play an important role in this “new physics” n n Planck, Einstein invoked energy quantization Bohr invoked angular momentum quantization ØWilson and Sommerfeld developed a general rule for the quantization of periodic systems 17

Wilson-Sommerfeld Quantization 18

Wilson-Sommerfeld Quantization Ø For a particle moving in a central field (like the Coulomb field) n n P=L q=φ Ø Then Ø Which is just the Bohr condition 19

Wilson-Sommerfeld Quantization Ø For a particle undergoing simple harmonic motion n n P = p. X q=x 20

Wilson-Sommerfeld Quantization Ø Continuing on Ø Which is just the Planck condition 21
- Slides: 21