BODY FLUID ANALYSIS Chapter One Cerebrospinal fluid analysis

BODY FLUID ANALYSIS

Chapter One Cerebrospinal fluid analysis

Acknowledgements � � � � Addisa Ababa University Jimma University Hawassa University Haramaya University of Gondar American Society for Clinical Pathology Center for Disease Control and Prevention-Ethiopia

Chapter outline � � Introduction to Cerebrospinal fluid Routine laboratory assays Collection of sample Gross appearance Cell counts Chemical analysis Morphological Examination Microbiological Examination Serological Examination

CEREBRAL SPINAL FLUIDS

Learning Objectives Upon completion of this chapter the student will be able to: 1. Describe the formation of CSF from blood. 2. Describe the appearance of normal CSF. 3. Define xanthochromia and state its significance. 4. Differentiate between CSF specimens caused by intracranial hemorrhage and a traumatic tap. 5 Differentiate between CSF specimens caused by intracranial hemorrhage and a traumatic tap.

Learning Objectives (continued) Given the laboratory observations of a bloody CSF, evaluate the supernatant and propose the type of pathological condition associated with a clear supernatant versus a xanthochromic supernatant. 9. Compare the difference of pathological conditions associated with the types of cells observed in a CSF. 10. List the normal range of glucose, protein, and cell count for a CSF. 11. Evaluate abnormal laboratory results with a pathological condition related to CSF. 12. Discuss appropriate collection requirements for CSF following a lumbar puncture. 8.

CSF Formation and Physiology First recognized by Cotugno in 1764, cerebrospinal fluid (CSF) is a major fluid of the body. CSF provides a physiologic system to supply nutrients to the nervous tissue, remove metabolic wastes, and produce a mechanical barrier to cushion the brain and spinal cord against trauma. CSF is produced in the choroid plexuses of the two lumbar ventricles and the third and fourth venticles. In adults, approximately 20 m. L of fluid is produced every hour.

Formation and Physiology The fluid flows through the subarachnoid space located between the arachnoid and pia mater. To maintain a volume of 90 to 150 m. L in adults and 10 to 60 m. L in neonates. the circulating fluid is reabsorbed back into the blood capillaries in the arachnoid granulations/villae at a rate equal to its production
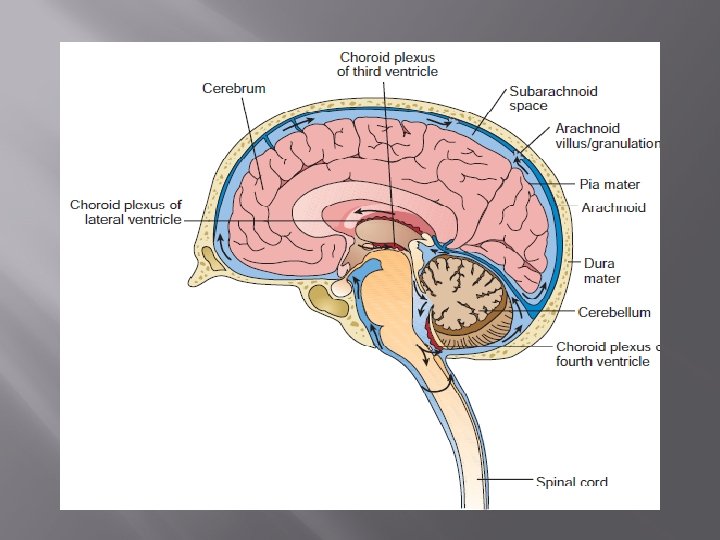

Cerebrospinal fluid (CSF) � Fluid in the space called sub-arachnoid space between the arachnoid mater and pia mater � Protects the underlying tissues of the central nervous system (CNS) � Serve as mechanical buffer to prevent trauma, regulate the volume of intracranial pressure circulate nutrients remove metabolic waste products from the CNS Act as lubricant � Has composition similar to plasma except that it has less protein, less glucose and more chloride ion

Cerebrospinal fluid analysis � Collection of CSF sample � Routine Laboratory assays of CSF Gross appearance RBC &WBC counts Morphological Examination Microbiological Examination Serological Examination

CSF cont’d � � Maximum volume of CSF � Adults 150 m. L � Neonates 60 m. L Rate of formation in adult is 450 -750 m. L per day or 20 ml per hour � reabsorbed at the same rate to maintain constant volume Collection by lumbar puncture done by experienced medical personnel About 1 -2 ml of CSF is collected for examination � lumbar puncture is made from the space between the 4 th and 5 th lumbar vertebrae under sterile conditions.
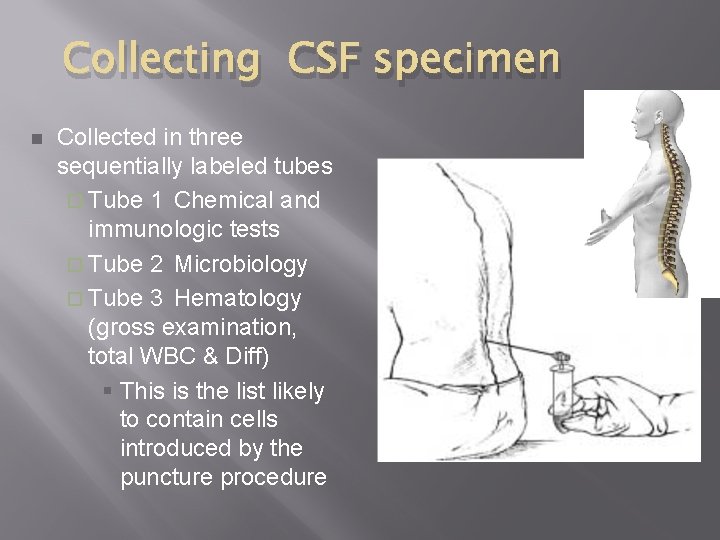
Collecting CSF specimen n Collected in three sequentially labeled tubes ¨ Tube 1 Chemical and immunologic tests ¨ Tube 2 Microbiology ¨ Tube 3 Hematology (gross examination, total WBC & Diff) § This is the list likely to contain cells introduced by the puncture procedure Location of CSF

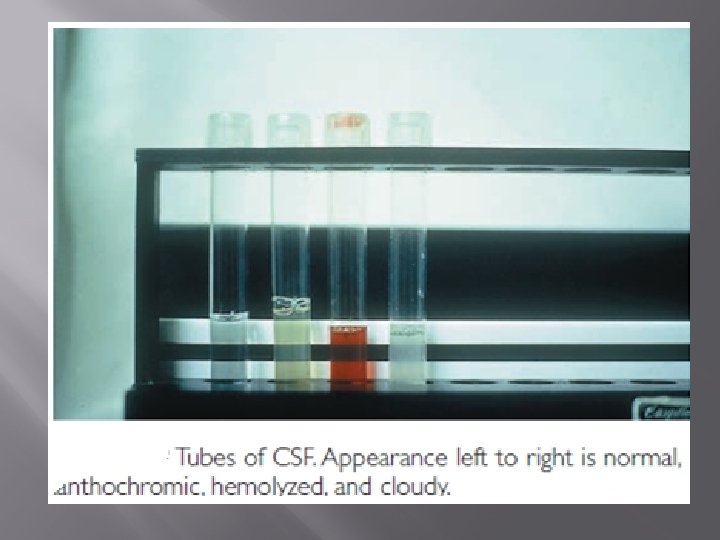
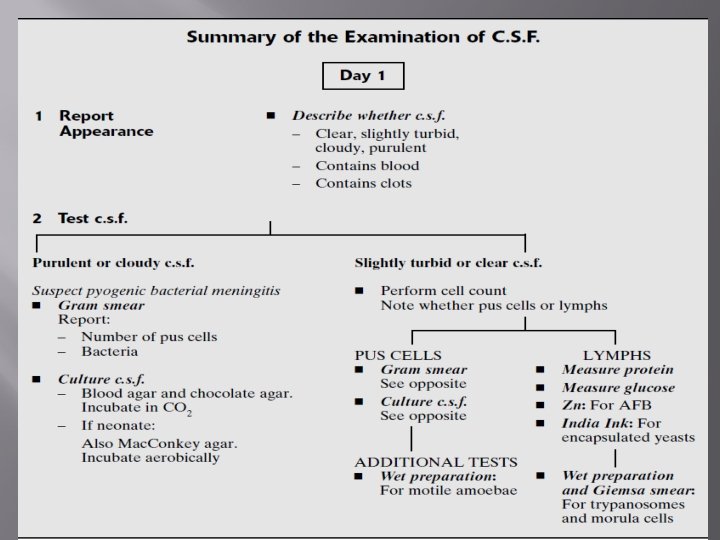
Report the appearance of the c. s. f. As soon as the c. s. f. reaches the laboratory, note its appearance. Report whether the fluid: � – is clear, slightly turbid, cloudy or definitely purulent (looking like pus), � – contains blood, � – contains clots. Normal c. s. f. Appears clear and colourless. �
Report the appearance of the c. s. f. � � Purulent or cloudy c. s. f. Indicates presence of pus cells, suggestive of acute pyogenic bacterial meningitis. Blood in c. s. f. This may be due to a traumatic (bloody) lumbar puncture or less commonly to haemorrhage in the central nervous system. When due to a traumatic lumbar puncture, sample No. 1 will usually contain more blood than sample No. 2.

Report the appearance of the c. s. f. � � Following a subarachnoid haemorrhage, the fluid may appear xanthrochromic, i. e. yellow-red (seen after centrifuging). Clots in c. s. f. Indicates a high protein concentration with increased fibrinogen, as can occur with pyogenic meningitis or when there is spinal constriction.

Clinical Significance � � Diagnosis of meningitis of bacterial, fungal, mycobacterial and amoebic origin or differential diagnosis of other infectious diseases subarachnoid hemorrhage or intracerebral hemorrhage

Principle of the test � CSF specimen examined visually and microscopically and total number of cells can be counted and identified Specimen: � the third tube in the sequentially collected tubes � must be counted within 1 hour of collection (cells disintegrate rapidly). If delay is unavoidable store 2 -8 o. C. � All specimens should be handled as biologically hazardous

Uptake and Utilization of Glucose � � Glucose is major energy substrate for brain as well as a major carbon source for many molecules. Brain uses 20 -25% of total oxygen and 15% of cardiac output is directed to CNS.

Glucose Utilization � � � When body glucose supply is decreased, other organs decrease glucose utilization to maintain adequate supply of glucose to brain. Other organs can readily switch to oxidation of another substrate for energy production. Under certain conditions, such as chronic starvation, the brain can oxidize other substances but maintains a minimal obligatory requirement for glucose.

Brain Utilization of Glucose � � Glycolysis--conversion to lactic acid Hexokinase has high activity in brain � Serves to trap glucose and maintain concentration gradient for diffusion

Brain Utilization of Glucose � 2 -deoxyglucose is also taken up by brain and phosphorylated by hexokinase, but then becomes trapped � Marker to correlate changes in neural activity with changes in glucose utilization. � � Enolase, an enzyme in glycolytic pathway, exists in nerve cells in unique isoform (neuron specific enolase, NSE) Used as a specific marker for neurons.

Brain Utilization of Glucose � � Pentose shunt Provides source of D-ribose for synthesis of DNA and RNA Produces NADPH required for lipid syntheses Most active during development

Utilization of Amino Acids � � Concept: Amino acids serve many functions in CNS Peptide and Protein synthesis Precursors for transmitters Neurotransmitters

Proteins in the CNS � Concept: Neurons must produce those proteins essential for their special functions: � conduction of action potentials � synaptic transmission � axoplasmic transport � establishment of specific connections

Specific Neural/Glial. Peptides/Proteins � � � Structual-cytoskeletal Cell Surface proteins play a role during development in directing neural connections Contractile proteins � function � � in axoplasmic movement Neurotubular protein Glial proteins (glial fibrillary protein)

Specific Neural/Glial Peptides/Proteins � � � Enzymes for transmitter synthesis and degradation Transmitter receptors Membrane transporters Ion Channels Growth Factors Synaptic Vesicle

MICROSCOPIC OBSERVATIONS OF CEREBRAL SPINAL FLUID

Neutrophils and Bands � � Morphologically identical to neutrophils and bands in blood Occasionally granulation disappears and pseudo-hypersegmentation is observed.

Lymphocytes � � � Almost identical morphology to lymphocytes in the blood Due to "flattening-out" of the lymphs during cytocentrifugation, nucleoli may be visible. Found in all fluid

Macrophages � � � Leukophages: Macrophagescontaining phagocytized WBCs are often pyknotic and easily confused with NRBC's. Found in all fluids. Erythrophages: Macrophages containing phagocytized RBC or RBC fragments. May contain several RBC. Found in all fluids. Siderophages: Macrophages containing phagocytized particles of hemosiderin, which stain a blue-black color.

Hematoidin Crystals � � These are bright-yellow diamond-shaped crystals of hemosiderin intracellular or extracellular on the slide. They are iron-negative on the Prussian blue stain and therefore Can be noted on the patient report without performing an iron stain.

Immature Granulocytes � � � Metamyelocytes, and promyelocytes may be found in fluids, though they are rarely seen. They are morphologically identical to those in the blood May be due to bone marrow contamination in CSF

Blasts � � � Morphologically similar to blasts found in the blood There may be some clover-leaf shaped nuclei due to cytocentrifugal distortion. May be found in all fluids Seen in association with leukemias, lymphomas Bone marrow contamination of CSF

Nucleated Red Blood Cells � � � NRBC are rarely seen body fluids. If observed, they should be reported as the number of NRBC per number of WBC counted They must be differentiated from pyknotic WBCs NRBC’s are commonly due to peripheral blood or bone marrow contamination of CSF

Abnormal Lymphocytes � Plasmacytoid lymphs: Identical in morphology to plasmacytoid lymphs in blood � Found � in all fluids. Mott cells: Plasma cells with numerous clear cytoplasmic vacuoles containing immunoglobulins

Reactive Macrophages � � These are most common in CSF from small children with subarachnoid hemorrhage but may be found in all body fluids May be very difficult to distinguish morphologically from large atypical lymphocytes

Malignant Cells � � � Malignant cells may be shed from solid tissue (non-hematopoietic) neoplasms into CSF or body cavity fluid submitted for cell counts Fluid will be turbid or bloody Malignant cells are usually seen in clusters of 3 -5 or more, but may occur singly

Microorganisms � � Intracellular bacteria or yeast can be observed in acute bacterial or fungal infections It is important to coordinate your findings with those of the Microbiology Section of the laboratory

Bloody CSF � When the CSF is pinkish red, this usually indicates the presence of blood, which may have resulted from: � Sub arachnoid hemorrhage � Intra cerebral hemorrhage � Infarct � traumatic tap

Order of Draw of Lumbar Puncture � � � 1 st - Chemistry 2 nd - Microbiology 3 rd - Hematology

Physical Examination � Color – Xanthochromia � Hyperbilirubinemia � Increased � Protein Turbidity � Increased White Blood Cells (Pleocytosis)

CSF Supernatant � � A traumatic tap shows progressively decreasing RBC in serial samples Generally, in subarachnoid hemorrhage, the RBC would be consistent from one tube to the next

CSF Supernatant � � After the CSF is centrifuged, the supernatant fluid is clear in a traumatic tap, but it is xanthochromic in a subarachnoid hemorrhage Xanthochromia of the CSF refers to a pink, orange, or yellow color of the supernatant after the CSF has been centrifuged

Cell Count � � The white cell count is increased when there is inflammation of the central nervous system, particularly the meninges Bacterial infections are usually associated with the presence of neutrophils in the CSF
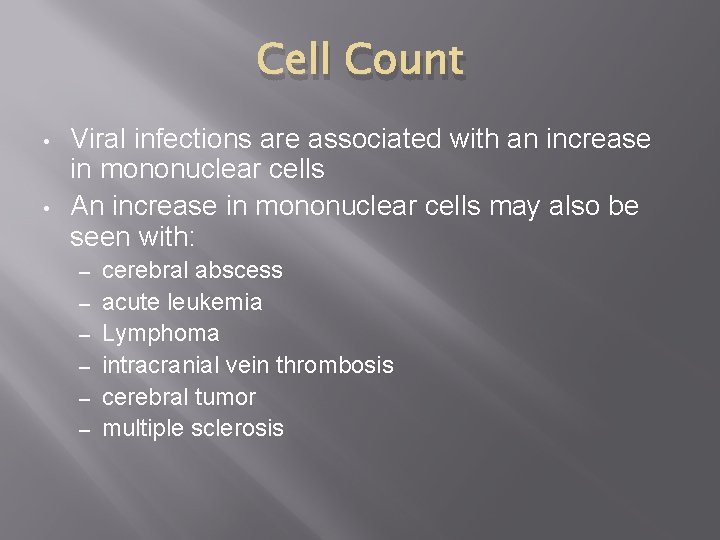
Cell Count • • Viral infections are associated with an increase in mononuclear cells An increase in mononuclear cells may also be seen with: – – – cerebral abscess acute leukemia Lymphoma intracranial vein thrombosis cerebral tumor multiple sclerosis

Cell Count cont’d � � A white cell count with an indication whether the cells are pus cells or lymphocytes, is required when the c. s. f. appears slightly cloudy or clear or when the Gram smear does not indicate pyogenic bacterial meningitis

Cell Count cont’d Method � � To identify whether white cells in the c. s. f. are polymorphonuclear neutrophils (pus cells) or lymphocytes, dilute the c. s. f. in a fluid which stains the cells. Istonic 0. 1% toluidine blue is recommended because it stains lymphocytes and the nuclei of pus cells blue. C. neoformans yeast cells stain pink. Red cells remain unstained. The motility of trypanosomes is not affected by the dye. When toluidine blue is unavailable, isotonic methylene blue can be used which will also stain the nuclei of leucocytes.

CSF Cell Count procedures 1 2 � � Mix 1 drop of the c. s. f. (sample No. 2 uncentrifuged c. s. f. ) with 1 drop of toluidine blue diluting fluid, Assemble a modified Fuchs-Rosenthal ruled counting chamber, making sure the chamber and cover glass are completely clean. When unavailable, an improved Neubauer (preferably Bright-Line) chamber can be used. A Fuchs-Rosenthal chamber is recommended because it has twice the depth (0. 2 mm) and is more suitable for counting WBCs in c. s. f.

CSF Cell Count procedures 3 Using a fine bore Pasteur pipette or capillary tube, carefully fill the counting chamber with the well-mixed diluted c. s. f. The fluid must not overflow into the channels on each side of the chamber. 4 Wait 2 minutes for the cells to settle. 5 Count the cells microscopically. Focus the cells and rulings using the 10 objective Count the cells in 5 of the large squares

Modified Fuchs-Rosenthal ruled chamber Improved Neubauer ruled chamber

Calculation of CSF Cell Counts

Chemistry Tests � � . Because CSF is formed by filtration of the plasma, chemicals in the CSF are that are found in the plasma. chemical composition is controlled by the blood-brain barrier, normal values for CSF chemicals are not the same as the plasma values. Abnormal values result from alterations in the permeability of the blood-brain barrier or increased production or metabolism by the neural cells in response to a pathologic condition.

Measurement of c. s. f total protein � � � Use the supernatant fluid from centrifuged c. s. f. or uncentrifuged c. s. f. when the sample appears clear. Total protein can be measured in c. s. f. using a colorimetric technique or a visual comparative technique, Normal Total c. s. f. protein is normally 0. 15– 0. 40 g/l (15– 40 mg%).

Measurement of c. s. f. total protein � When the total protein exceeds 2. 0 g/l (200 mg%), the fibrinogen level is usually increased sufficiently to cause the c. s. f. to clot. This may occur in severe pyogenic meningitis, spinal block, or following haemorrhage.

Clinical Significance of Elevated Protein Values � � � The causes of elevated CSF protein include damage to the blood-brain barrier, production of immunoglobulins within the CNS, decreased clearance of normal protein from the fluid, and degeneration of neural tissue. Meningitis and hemorrhage conditions that damage the blood-brain barrier are the most common causes of elevated CSF protein. Many other neurologic disorders can elevate the CSF protein, and finding an abnormal result on clear fluid with a low cell count is not unusual
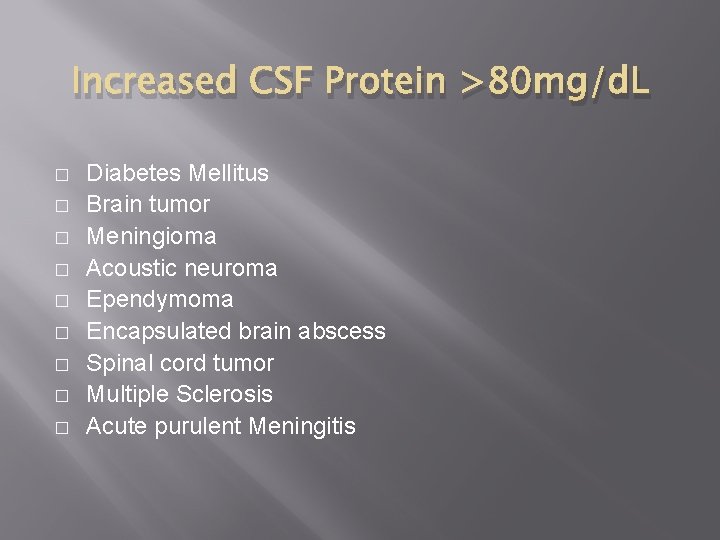
Increased CSF Protein >80 mg/d. L � � � � � Diabetes Mellitus Brain tumor Meningioma Acoustic neuroma Ependymoma Encapsulated brain abscess Spinal cord tumor Multiple Sclerosis Acute purulent Meningitis

Increased CSF Protein >80 mg/d. L Granulomatous Meningitis Carcinomatous Meningitis Syphilis (protein may be normal if longstanding) Guillain-Barre Syndrome (Infectious polyneuritis) Cushing's Disease Connective tissue disease Uremia Myxedema Cerebral hemorrhage

Cerebrospinal Fluid Glucose � � Glucose enters the CSF by selective transport across the bloodbrain barrier, a normal value 60% to 70% that of the plasma glucose. If the plasma glucose is 100 mg/d. L, then a normal CSF glucose would be approximately 65 mg/d. L. For an accurate evaluation of CSF glucose, a blood glucose test must be run for comparison. CSF glucose is analyzed using the same procedures employed for blood glucose. Specimens should be tested immediately because glycolysis occurs rapidly in the CSF.

Clinical Significance of Elevated glucose Values The diagnostic significance of CSF glucose is confined to the finding of values that are decreased in relation to plasma values. Elevated CSF glucose values are always a result of plasma elevations. Low CSF glucose values can be of considerable diagnostic value in determining the causative agents in meningitis. The finding of a markedly decreased CSF glucose accompanied by an increased WBC count and a large percentage of neutrophils is indicative of bacterial meningitis.

Measurement of c. s. f. glucose Glucose must be measured within 20 minutes of the c. s. f. being withdrawn otherwise a falsely low result will be obtained due to glycolysis. Use the supernatant fluid from centrifuged c. s. f. or uncentrifuged c. s. f. if the sample appears clear. Glucose can be measured in c. s. f. using a colorimetric technique or a simpler semiquantitative technique using Benedict’s reagent.

Measurement of c. s. f. glucose Normal c. s. f. glucose: This is about half to two thirds that found in blood i. e. 2. 5– 4. 0 mmol/1 (45– 72 mg%). Raised c. s. f. glucose: Occurs when the blood glucose level is raised (hyperglycaemia) and sometimes with encephalitis. Low c. s. f. glucose: The c. s. f. glucose concentration is reduced in most forms of meningitis, except viral meningitis. In pyogenic bacterial meningitis it is markedly reduced and may even be undetectable.

Glucose � Low glucose levels, as compared to plasma levels, are seen in: � bacterial meningitis � cryptococcal meningitis � malignant involvement of the meninges and sarcoidosis � Glucose levels are usually normal in viral infections of the CNS

CSF Normal Adult Lab Ranges � � Normal CSF Levels: Protein (10 - 45 mg/d. L) Glucose (40 - 70 mg/d. L) Physical Appearance Clear/colorless RBC <5/m. L WBC <5/m. L

Lactate � � � In bacterial and cryptococcal infection, an increased CSF lactate is found earlier than a reduced glucose In viral meningitis, lactate levels remain normal, even when neutrophils are present in the CSF Raised levels may also occur with severe cerebral hypoxia or genetic lactic acidosis

CSF India ink preparation � When cryptococcal meningitis is clinically suspected, e. g. patient with HIV disease, or when yeast cells are detected when performing a cell count or examining a Gram smear, examine an India ink preparation or a wet preparation by dark-field microscopy for encapsulated yeasts.

India ink preparation 1 Centrifuge the c. s. f. for 5– 10 minutes. Remove the supernatant fluid and mix the sediment. 2 Transfer a drop of the sediment to a slide, cover with a cover glass and examine by dark-field Microscopy or add a drop of India ink , use nigrosin 200 g/l (20% w/v solution. 3 Examine the preparation using the 40 objective Look for oval or round cells, some showing budding, irregular in size, measuring 2– 10 m in diameter and surrounded by a large unstained capsule

Wet preparation to detect amoebae Examine a wet preparation for motile amoebae when primary amoebic meningoencephalitis is clinically suspected (rare condition caused by N. fowleri) or the c. s. f. contains pus cells with raised protein and low glucose, but no bacteria are seen in the Gram smear. Red cells may also be present.

Wet preparation to detect amoebae 1 Transfer a drop of uncentrifuged purulent c. s. f. or a drop of sediment from a centrifuged specimen to a slide and cover with a cover glass. 2 Examine the preparation using the 10 and 40 objectives, with the condenser closed sufficiently to give good contrast. Look for small, clear, motile, elongated forms among the pus cells. Use the 40 objective

cont’d

Exercise 1 A CSF WBC count is diluted with: 2 A total CSF cell count on a clear fluid should be: 3 The purpose of adding albumin to CSF before cytocentrifugation is to: 4 The primary concern when pleocytosis of neutrophils and lymphocytes is found in the CSF is: 5 Neutrophils with pyknotic nuclei may be mistaken for: 6 The presence of which of the following cells is increased when a CNS shunt malfunctions?

Summary � You should now be able to discuss the formation and collection procedure for cerebral spinal fluid, normal and abnormal findings in CSF and methods of analysis and evaluation of CSF

References: � � � � � Urinalysis and body fluids / Susan King Strasinger, 5 th ed. 2008 District laboratory practice in tropical countries. 2 nd ed. Part I. Monica Cheesbrough, 2005 Text book of urinalysis and body fluids. Doris LR, Ann EN, 1983 Urinalysis and body fluids: A color text and atlas. Karen MR, Jean JL. 1995 Clinical chemistry: Principles, procedures, correlation. 3 rd ed. Michael L. Bishop et al. 1996 Tietz Text book of clinical chemistry. 3 rd ed. Carl AB, Edward RA, 1999 Clinical chemistry: Theory, analysis, correlation 4 th ed. Lawrence AK. 2003 ASCP Document Urinalysis lecture note. Mistire W. , Dawite Y. 76
- Slides: 76