Boardworks Ltd 2003 Teachers Notes A slide contains
© Boardworks Ltd 2003
Teacher’s Notes A slide contains teacher’s notes wherever this icon is displayed To access these notes go to ‘Notes Page View’ (Power. Point 97) or ‘Normal View’ (Power. Point 2000). Notes Page View Normal View Flash Files A flash file has been embedded into the Power. Point slide wherever this icon is displayed – These files are not editable. © Boardworks Ltd 2003

Energy: Heat Transfer Heat is the name for the type of kinetic energy possessed by particles. Heat energy is measured in joules [J]. How many joules are there in a kilojoule [k. J]? If something gains a lot of heat energy, it becomes hot so what is temperature? © Boardworks Ltd 2003

Temperature measurement Temperature is a measure of the hotness or coldness of something, not the total amount of energy contained. Temperature is measured in degrees Celsius (or centigrade) - ºC. The freezing point of water is DEFINED as 0 o. C (at 1 atm. ). The boiling point of water is DEFINED as 100 o. C (at 1 atm. ). The temperature can be measured in a variety of different thermometers. These include liquid in glass, digital, thermocouple and bimetal strip thermometers. © Boardworks Ltd 2003

Energy : heat transfer The correct phrase is ‘thermal transfer’. Heat energy can be transferred (moved) in 4 ways: 1. Conduction 2. Convection 3. Evaporation 4. Radiation Which ever way heat moves, it always moves from HOT to COLD. Heat energy only flows when there is a temperature difference. © Boardworks Ltd 2003

The Particle Model The existence of solids, liquids and gases is explained by the following ideas: 1. All substances are made up of particles (atoms, ions and molecules). 2. That these particles are attracted to each other, some strongly and others weakly. 3. That these particles move around (i. e. Have kinetic energy). 4. That their kinetic energy increases with temperature. © Boardworks Ltd 2003
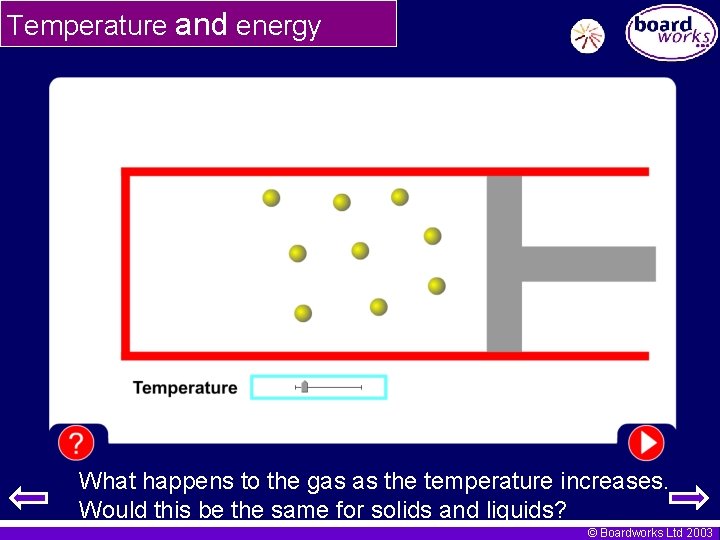
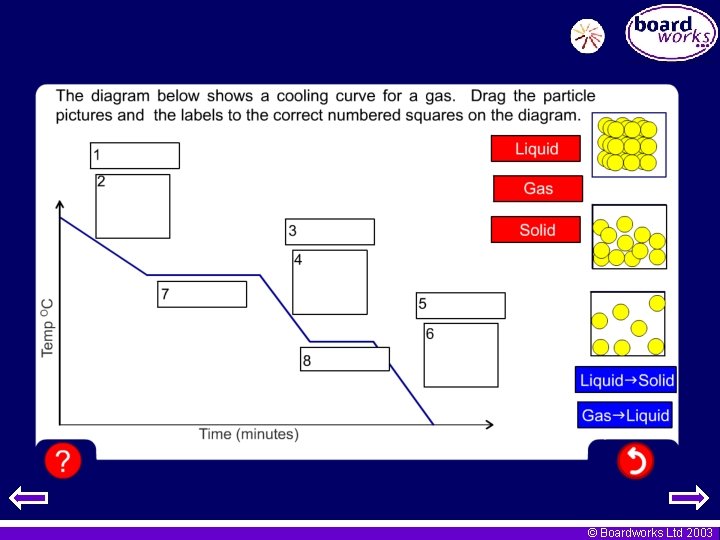
Temperature and energy What happens to the gas as the temperature increases. Would this be the same for solids and liquids? © Boardworks Ltd 2003

Energy : heat transfer 1. Conduction Take care whilst doing this experiment: ALWAYS LET GO OF THE METAL QUICKLY WASH ALL BURNS WITH COLD WATER FOR 10 MINS You need an: 8 cm strip of Cu 8 cm piece of Wood Bunsen Burner Stopwatch © Boardworks Ltd 2003

Energy : heat transfer 1. WEAR SAFETY GLASSES Conduction Hold the piece of Cu in the YELLOW Bunsen Burner flame. How long does it take you to feel the heat? Now do the same for wood. Why are the times very different? © Boardworks Ltd 2003

Energy : heat transfer 1. Conduction in metals Metals have atoms inside them and lots of free electrons. The free electrons can move around and vibrate. The heat energy is passed on by neighbouring particles vibrating along the metal. There are too few free electrons in a non-metal for this to happen. © Boardworks Ltd 2003

Energy : heat transfer Which would feel warmer if we picked up a piece of wood and a piece copper metal both at room temperature (i. e. both at 250 C)? The wood feels warmer because it is a POOR conductor. So it would not conduct heat away from your hand as quickly as the copper. © Boardworks Ltd 2003
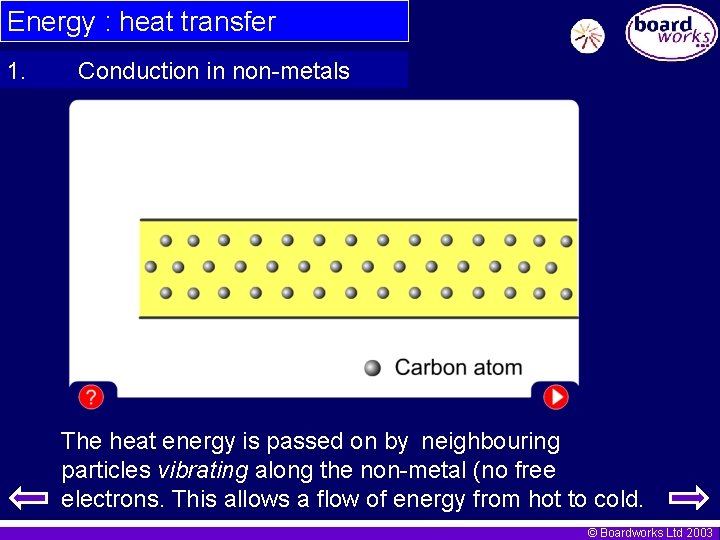
Energy : heat transfer 1. Conduction in non-metals The heat energy is passed on by neighbouring particles vibrating along the non-metal (no free electrons. This allows a flow of energy from hot to cold. © Boardworks Ltd 2003
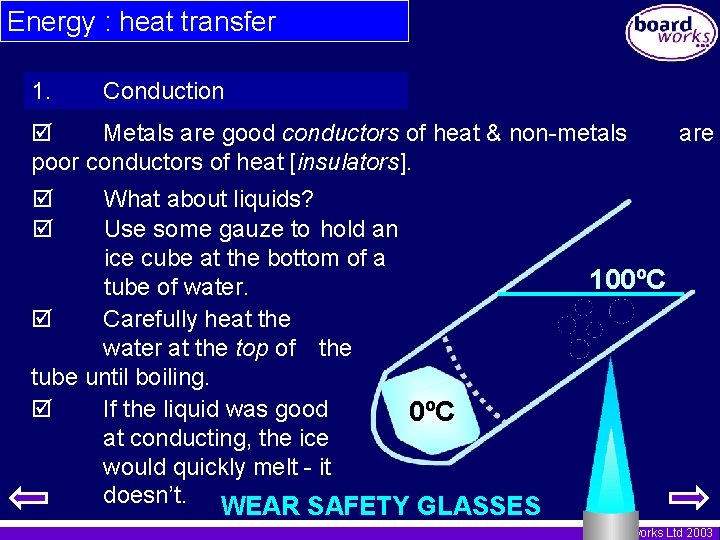
Energy : heat transfer 1. Conduction Metals are good conductors of heat & non-metals poor conductors of heat [insulators]. What about liquids? Use some gauze to hold an ice cube at the bottom of a tube of water. Carefully heat the water at the top of the tube until boiling. If the liquid was good 0ºC at conducting, the ice would quickly melt - it doesn’t. WEAR SAFETY GLASSES are 100ºC © Boardworks Ltd 2003
![Energy : heat transfer 1. Conduction Liquids are poor conductors of heat [insulators]. What Energy : heat transfer 1. Conduction Liquids are poor conductors of heat [insulators]. What](http://slidetodoc.com/presentation_image_h2/73df843cb38139de3d0409871e1563e7/image-14.jpg)
Energy : heat transfer 1. Conduction Liquids are poor conductors of heat [insulators]. What about gases? Carefully put your finger 1 cm away from a bunsen burner flame. If gases were good conductors, you’d burn your finger - you don’t. Gases are good insulators. WEAR SAFETY GLASSES © Boardworks Ltd 2003

Energy : heat transfer 1. Conduction Copy this summary table into your book : © Boardworks Ltd 2003
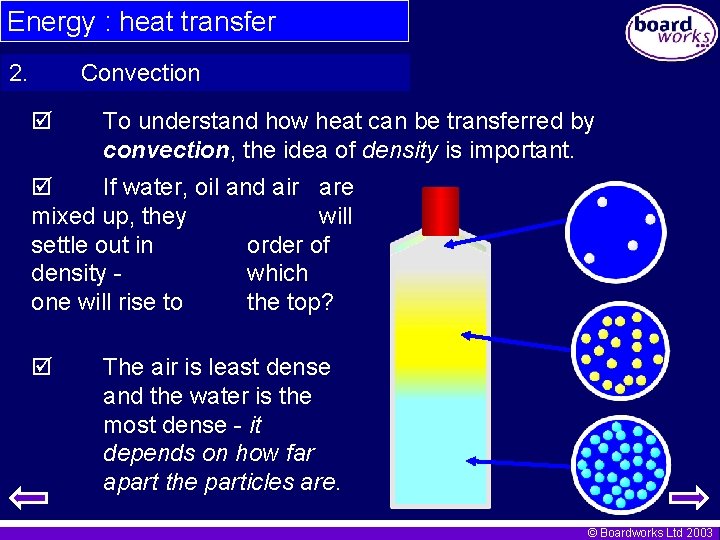
Energy : heat transfer 2. Convection To understand how heat can be transferred by convection, the idea of density is important. If water, oil and air are mixed up, they will settle out in order of density which one will rise to the top? The air is least dense and the water is the most dense - it depends on how far apart the particles are. © Boardworks Ltd 2003

Energy : heat transfer 2. Convection The movement of hotter areas in a liquid can be seen using potassium permanganate as a dye: This cycle is called a convection current. Can you explain how the convection current moves using the idea of density? HE AT The diagrams on the left will help you. © Boardworks Ltd 2003

Energy : heat transfer 2. Convection currents can’t occur in solids because the particles are held in fixed positions - but can they occur in gases? Place a candle at one side of a litre beaker. Place a piece of card down the centre, leaving a gap of 2 cm at the bottom. Make some smoke with smouldering spills and watch the path of the smoke. Can you explain why this happens? © Boardworks Ltd 2003

Energy : heat transfer 2. Convection When shaft mining was first used to mine coal, convection currents caused by an underground fire were used to ventilate the shafts: Why do you think miners don’t use this method anymore? © Boardworks Ltd 2003
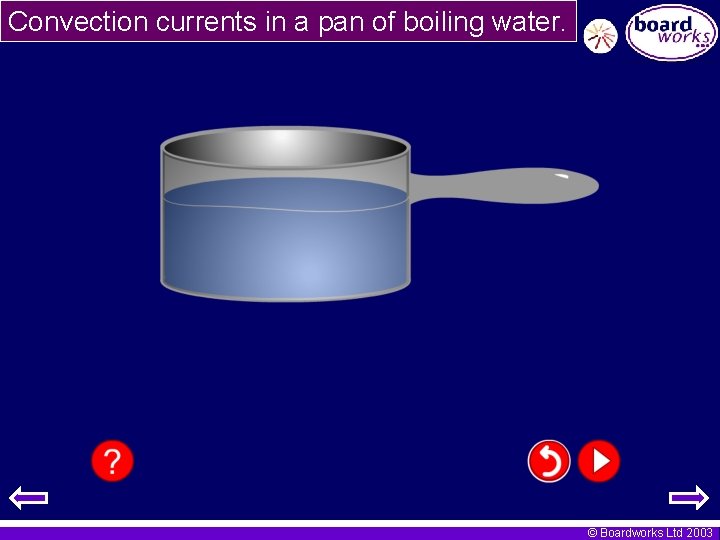
Convection currents in a pan of boiling water. © Boardworks Ltd 2003

Energy : heat transfer 3. Evaporation is when the particles in a liquid escape to form a vapour. Evaporation can occur at any temperature but it occurs most rapidly at a liquid’s boiling point. The particles that escape take some energy from the remaining particles and so the temperature of the liquid falls. Take 4 equal masses of cotton wool and soak them in the 4 different liquids provided. Wrap the cotton wool around the bottom of a thermometer and secure it with an rubber band. Take the temperature every 0. 5 minutes and record your results in the table on the next slide © Boardworks Ltd 2003
Energy : heat transfer 3. Evaporation © Boardworks Ltd 2003

Energy : heat transfer 3. Evaporation Either : 1. Draw a bar chart of your results. Put temperature change on the y axis Put ‘liquid’ on the x axis 2. OR Plot a line graph of your results. Put temperature on the y axis Put time on the x axis Plot 4 lines, one for each liquid Are any of the results anomalous? © Boardworks Ltd 2003

Energy : heat transfer 4. Radiation Heat can move by travelling as Infra Red waves These are electromagnetic waves, like light waves, but with a longer wavelength. This means that infra red waves act very much like light waves: They can travel through a vacuum They travel at 300, 000 m/s They can be reflected They can’t travel through opaque materials. © Boardworks Ltd 2003

Energy : heat transfer 4. Radiation Paint 4 thermometer bulbs with the following colours Black White Silver Red Place thermometers into very hot water for 1 minute. Take it out of the water, start the stopwatch and read the temperature. Take the temperature every 30 seconds and put the answers in the results table on the next page. © Boardworks Ltd 2003
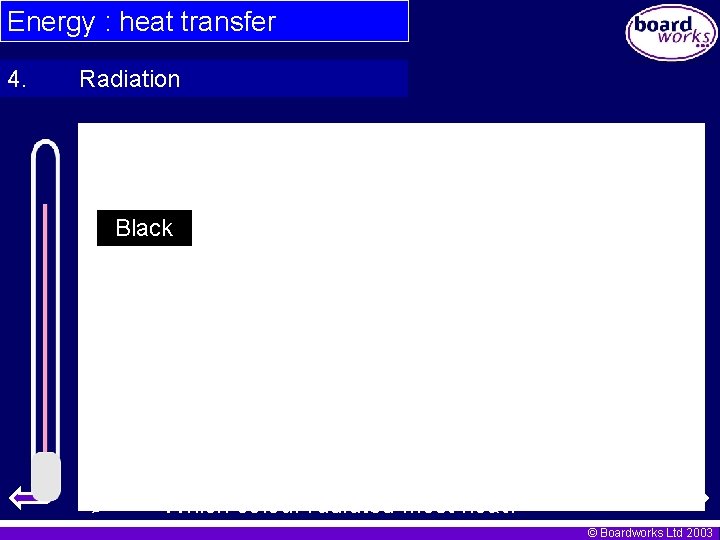
Energy : heat transfer 4. Radiation Black Which colour radiated most heat? © Boardworks Ltd 2003

Energy : heat transfer 4. Radiation Either : a) Draw a bar chart of your results. Put temperature change on the y axis and colour on the x axis. OR b) Draw a line graph of your results. Put temperature on the y axis and time on the x axis. Draw 4 lines on the graph, one for each colour. © Boardworks Ltd 2003

Energy : heat transfer 4. Radiation 1. How does a cup of tea lose heat by conduction, convection, evaporation and radiation? 2. Why does take-away food often come in aluminium containers? 3. Why do elephants have big ears? © Boardworks Ltd 2003

Energy : heat transfer 4. Radiation 5. A cup of tea takes up to 30 minutes to go cold. This depends on the colour and shape of the cup. Design an investigation to find out the best shape and colour of a cup to keep tea warm for longer. © Boardworks Ltd 2003

Energy losses Name three processes which cause energy to be lost from the home. Which spots on the house diagram lose energy? What can we do to prevent this energy loss? © Boardworks Ltd 2003

Changes of state • At cold enough temperatures even things that are normally gases become solid. • At higher temperatures solids change to become liquids or gases – as long as they don’t catch fire or decompose first. Water can be solid, liquid or gas © Boardworks Ltd 2003
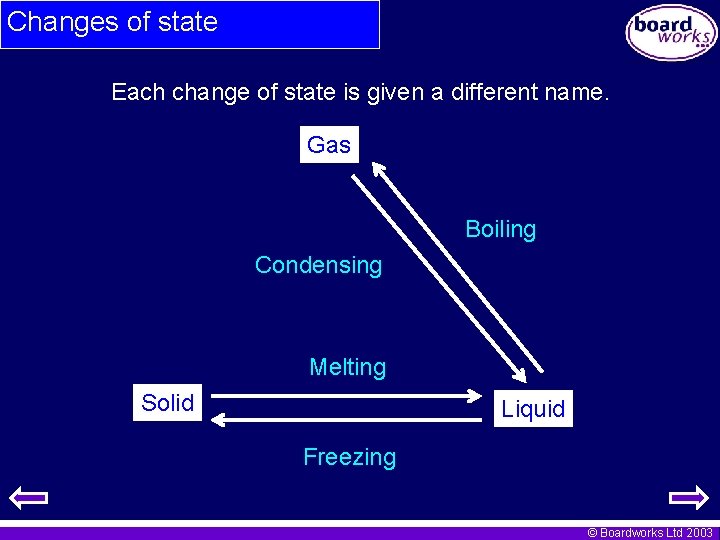
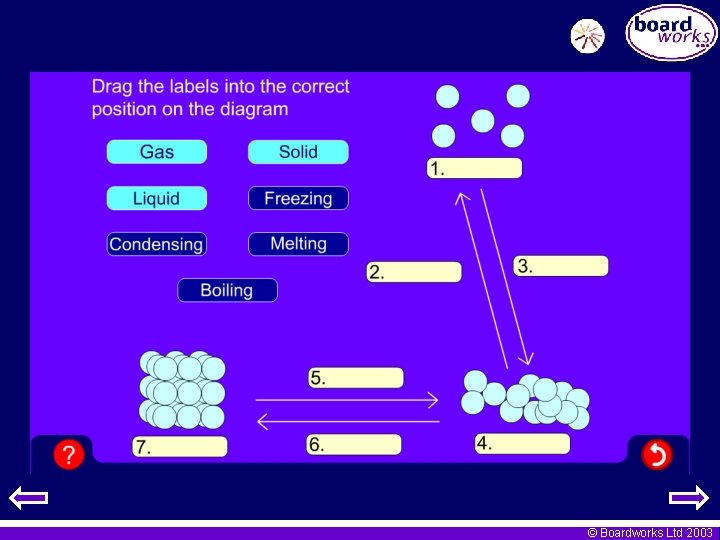
Changes of state Each change of state is given a different name. Gas Boiling Condensing Melting Solid Liquid Freezing © Boardworks Ltd 2003
© Boardworks Ltd 2003

Changes of state – heating curves • If a solid is heated its temperature rises until it reaches its melting point. • At the melting point the temperature stops rising whilst the solid melts. This is because heat energy is going into separating the particles rather than raising the temperature. • Once all the solid has melted the temperature starts to rise again until it reaches the boiling point. • At the boiling point the temperature again stays the same as energy goes into further separating the particles. © Boardworks Ltd 2003
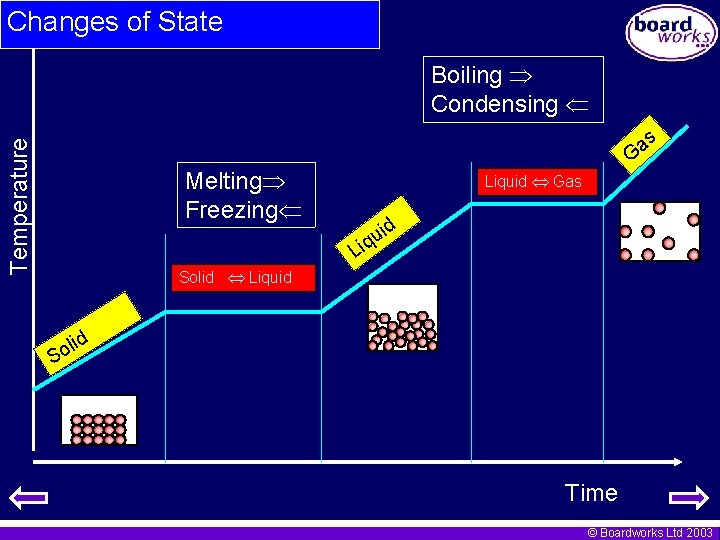
Changes of State Boiling Condensing Temperature as G Melting Freezing Liquid Gas id u iq L Solid Liquid lid o S Time © Boardworks Ltd 2003
© Boardworks Ltd 2003
- Slides: 36