BMS 631 LECTURE 3 Light and Matter J

BMS 631 - LECTURE 3 Light and Matter J. Paul Robinson SVM Professor of Cytomics Professor of Biomedical Engineering Purdue University Reading materials: (Shapiro 3 rd ed. Pp 75 -93; 4 th Ed. Shapiro pp 101 -136) Lab: Bindley 122 Purdue University Office Phone: (765)-494 0757 Fax (765)-494 0517 Email: robinson. ATflowcyt. cyto. purdue. edu WEB: http: //www. cyto. purdue. edu Notice: The materials in this presentation are copyrighted materials. If you want to use any of these slides, you may do so if you credit each slide with the author’s name. It is illegal to place these notes on Course. Hero or any other site. All materials used in this course are available for download on the web at http: //tinyurl. com/2 wkpp Slide last modified January, 2013 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 1

Learning Objectives • Understand the basic properties of light • Understand basic principles of light propagation • Understand the constraints that are placed in measurement systems • Understand how image formation, numerical aperture and absorption impact instrument design 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 2

Light and Matter • Energy – joules, radiant flux (energy/unit time) – watts (1 watt=1 joule/second) • Angles – steradians - sphere radius r - circumference is 2 r 2; the angle that intercepts an arc r along the circumference is defined as 1 radian. (57. 3 degrees) a sphere of radius r has a surface area of 4 r 2. One steradian is defined as the solid angle which intercepts as area equal to r 2 on the sphere surface 3 rd Ed - Shapiro p 75 4 th Ed – Shapiro p 101 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 3

Terms • • Side scatter, forward angle scatter, cell volume, coulter volume: Understand light scattering concepts; intrinsic and extrinsic parameters • • Photometry: Light - what is it - wavelengths we can see 400 -750 nm, most sensitive around 550 nm. Below 400 nm essentially measuring radiant energy. Joules (energy) radiant flux (energy per unit time) is measured in watts (1 watt=1 joule/second). Steradian (sphere radius r has surface area of 4 r 2; one steradian is defined as that solid angle which intercepts an area equal to r 2 on the surface. Mole - contains Avogadro's number of molecules (6. 02 x 1023) and contains a mass in grams = molecular weight. Photons - light particles - waves - Photons are particles which have no rest mass - pure electromagnetic energy - these are absorbed and emitted by atoms and molecules as they gain or release energy. This process is quantized, is a discrete process involving photons of the same energy for a given molecule or atom. The sum total of this energy gain or loss is electromagnetic radiation propagating at the speed of light (3 x 108 m/s). The energy (joules) of a photon is E=h and E=h /l [ -frequency, l-wavelength, h-Planck's constant 6. 63 x 10 -34 jouleseconds] Energy - higher at short wavelengths - lower at longer wavelengths. • • 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 4

Photons and Quantum Theory • Photons – particles have no rest mass - composed of pure electromagnetic energy - the absorption and emission of photons by atoms and molecules is the only mechanism for atoms and molecules can gain or lose energy • Quantum mechanics – absorption and emission are quantized - i. e. discrete process of gaining or losing energy in strict units of energy - i. e. photons of the same energy (multiple units are referred to as electromagnetic radiation) • Energy of a photon – can be computed from its frequency ( ) in hertz (Hz) or its wavelength (l) in meters from E=h and E=hc/ 3 rd Ed Shapiro p 76 2: 27 PM = wavelength h = Planck’s constant (6. 63 x 10 -34 joule-seconds c = speed of light (3 x 108 m/s) © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 5

Electromagnetic Spectrum We can see from about 370 nm to about 700 nm This is known as the visible spectrum Images from: http: //www. lbl. gov/Micro. Worlds/ALSTool/EMSpec 2. html 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 6
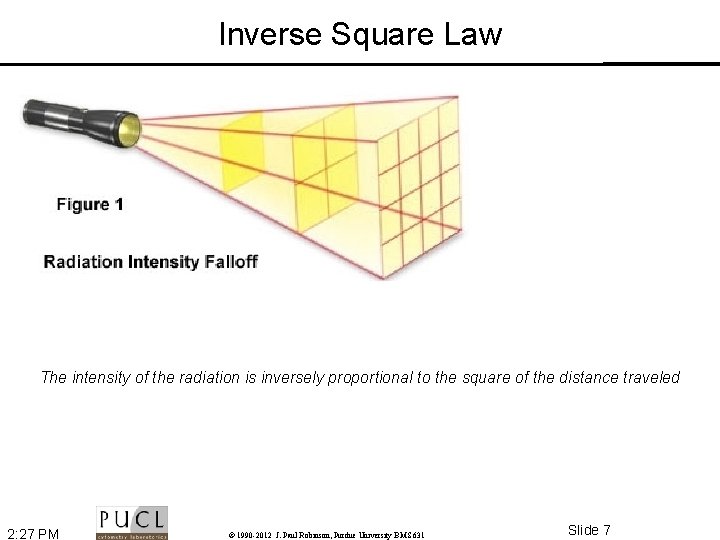
Inverse Square Law The intensity of the radiation is inversely proportional to the square of the distance traveled 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 7

Laser power E=h and E=hc/ • One photon from a 488 nm argon laser has an energy of 6. 63 x 10 -34 joule-seconds x 3 x 108 E= = 4. 08 x 10 -19 J 488 x 10 -3 • To get 1 joule out of a 488 nm laser you need 2. 45 x 1018 photons • 1 watt (W) = 1 joule/second a 10 m. W laser at 488 nm is putting out 2. 45 x 1016 photons/sec 3 rd Ed. Shapiro p 77 4 th Ed Shapiro p 109 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 8

Laser power What about a UV laser? E= E=h and E=hc/ 6. 63 x 10 -34 joule-seconds x 3 x 108 325 x 10 -3 = 6. 12 x 10 -19 J so 1 Joule at 325 nm = 1. 63 x 1018 photons What about a He-Ne laser? E= 6. 63 x 10 -34 joule-seconds x 3 x 108 633 x 10 -3 = 3. 14 x 10 -19 J so 1 Joule at 633 nm = 3. 18 x 1018 photons 3 rd Ed. Shapiro p 77 4 th Ed Shapiro p 109 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 9
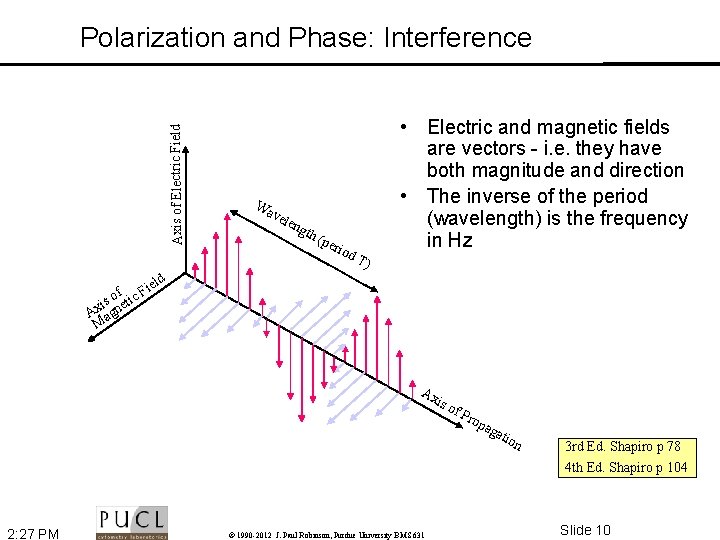
Axis of Electric Field Polarization and Phase: Interference i of tic F s i e Ax agn M Wa vel eng th ( per iod T) • Electric and magnetic fields are vectors - i. e. they have both magnitude and direction • The inverse of the period (wavelength) is the frequency in Hz eld Ax is o 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 f. P rop aga tio n 3 rd Ed. Shapiro p 78 4 th Ed. Shapiro p 104 Slide 10
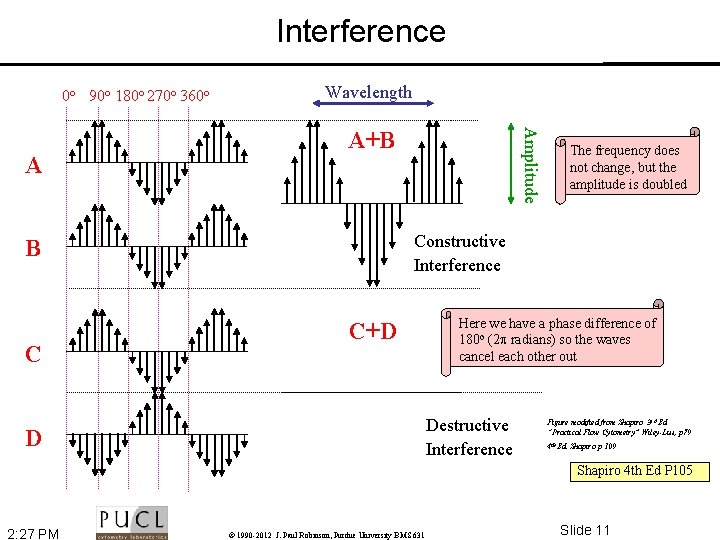
Interference 0 o 90 o 180 o 270 o 360 o A+B The frequency does not change, but the amplitude is doubled Constructive Interference B C Amplitude A Wavelength C+D Here we have a phase difference of 180 o (2 radians) so the waves cancel each other out Destructive Interference D Figure modified from Shapiro 3 rd Ed “Practical Flow Cytometry” Wiley-Liss, p 79 4 th Ed. Shapiro p 109 Shapiro 4 th Ed P 105 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 11
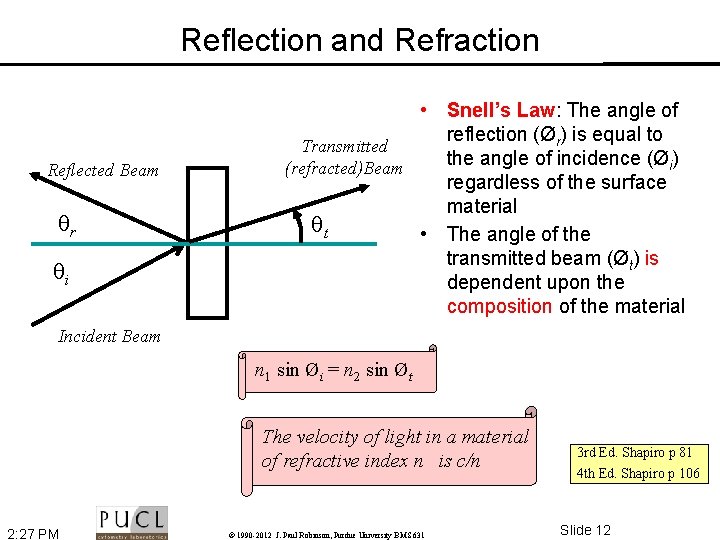
Reflection and Refraction Reflected Beam r i • Snell’s Law: The angle of reflection (Ør) is equal to Transmitted the angle of incidence (Øi) (refracted)Beam regardless of the surface material t • The angle of the transmitted beam (Øt) is dependent upon the composition of the material Incident Beam n 1 sin Øi = n 2 sin Øt The velocity of light in a material of refractive index n is c/n 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 3 rd Ed. Shapiro p 81 4 th Ed. Shapiro p 106 Slide 12
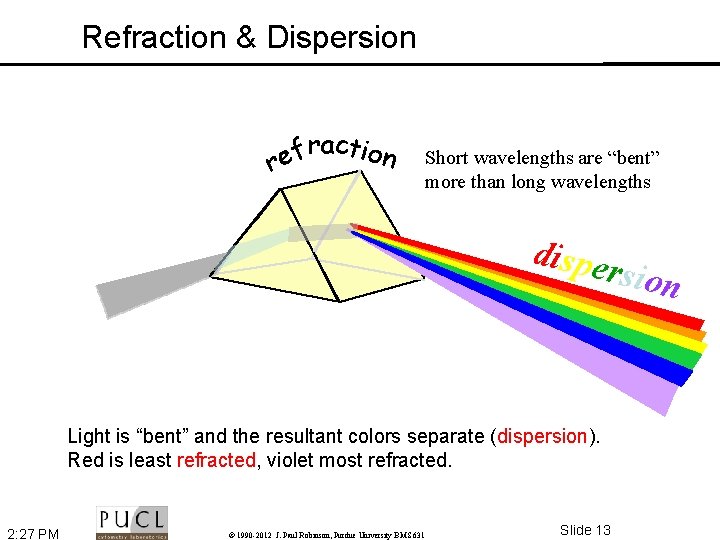
Refraction & Dispersion raction f e r Short wavelengths are “bent” more than long wavelengths dispe rsion Light is “bent” and the resultant colors separate (dispersion). Red is least refracted, violet most refracted. 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 13

Light Propagation & Vergence • Considering a point source emission of light, rays emanate over 4 pi steradians • If the ray of light travels through a length L of a medium of RI n, the optical path length S=Ln (thus S represents the distance light would have traveled in a vacuum in the same time it took to travel the distance L in the medium (RI n). • Rays diverge (because they come from a point source) • Vergence is measured in diopters 3 rd Shapiro p 93 4 th Shapiro p 119 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 14

Image Formation • Object plane - (originating image) • Image plane - inverted real image • A real image is formed whenever rays emanating from a single point in the object plane again converge to a single point Shapiro p 94 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 15

Numerical Aperture • The wider the angle the lens is capable of receiving light at, the greater its resolving power • The higher the NA, the shorter the working distance Shapiro p 96 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 16

Numerical Aperture • Resolving power is directly related to numerical aperture. • The higher the NA the greater the resolution • Resolving power: The ability of an objective to resolve two distinct lines very close together NA = n sin – (n=the lowest refractive index between the object and first objective element) (hopefully 1) – is 1/2 the angular aperture of the objective 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 17
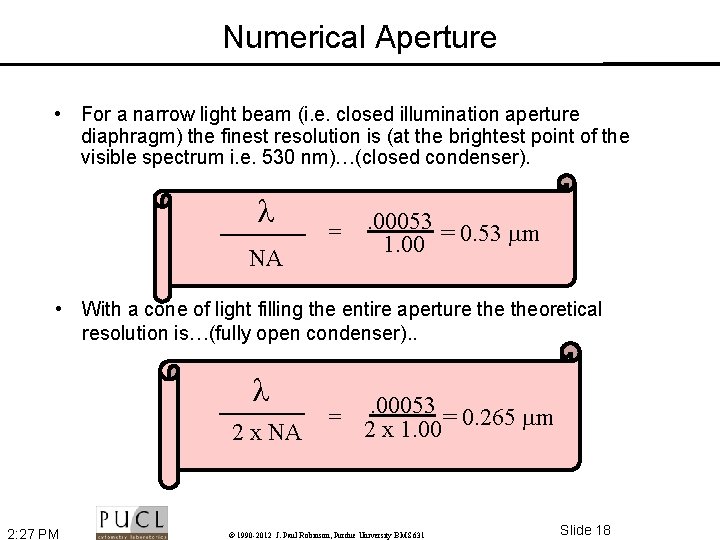
Numerical Aperture • For a narrow light beam (i. e. closed illumination aperture diaphragm) the finest resolution is (at the brightest point of the visible spectrum i. e. 530 nm)…(closed condenser). NA = . 00053 1. 00 = 0. 53 m • With a cone of light filling the entire aperture theoretical resolution is…(fully open condenser). . 2 x NA 2: 27 PM = . 00053 = 0. 265 m 2 x 1. 00 © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 18
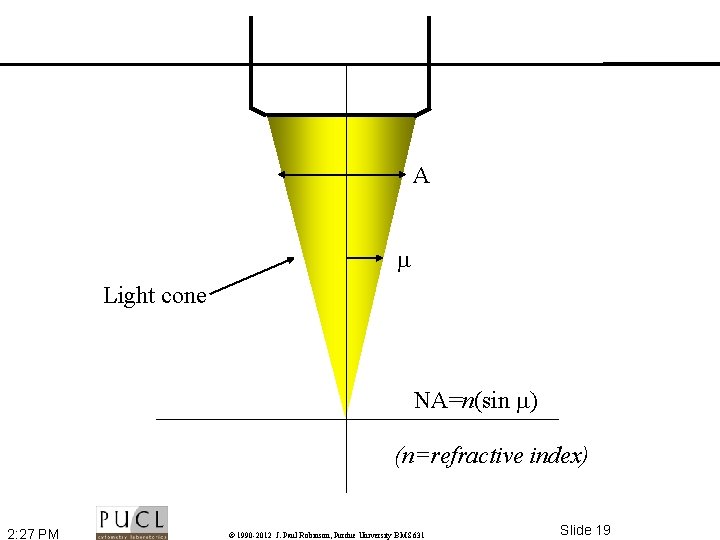
A Light cone NA=n(sin ) (n=refractive index) 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 19
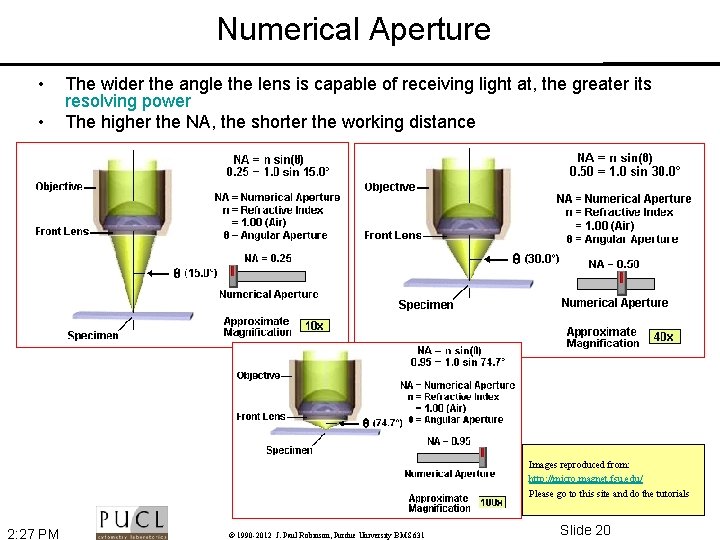
Numerical Aperture • • The wider the angle the lens is capable of receiving light at, the greater its resolving power The higher the NA, the shorter the working distance Images reproduced from: http: //micro. magnet. fsu. edu/ Please go to this site and do the tutorials 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 20
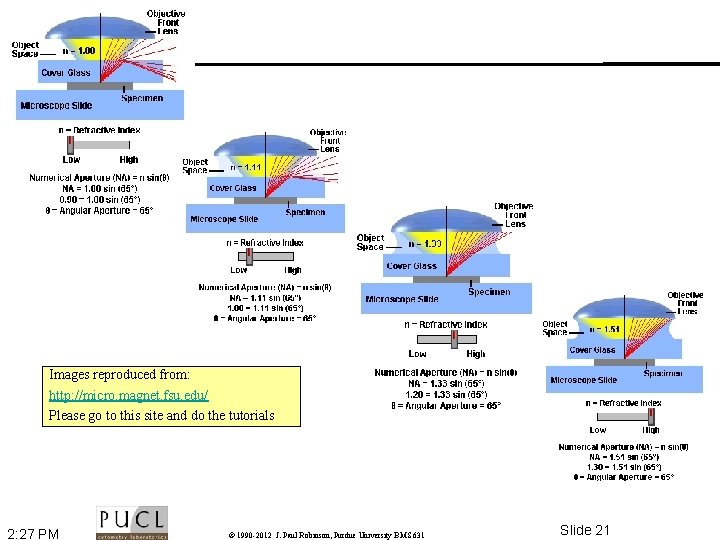
Images reproduced from: http: //micro. magnet. fsu. edu/ Please go to this site and do the tutorials 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 21

Some Definitions • Absorption – When light passes through an object the intensity is reduced depending upon the color absorbed. Thus the selective absorption of white light produces colored light. • Refraction – Direction change of a ray of light passing from one transparent medium to another with different optical density. A ray from less to more dense medium is bent perpendicular to the surface, with greater deviation for shorter wavelengths • Diffraction – Light rays bend around edges - new wavefronts are generated at sharp edges - the smaller the aperture the lower the definition • Dispersion – Separation of light into its constituent wavelengths when entering a transparent medium - the change of refractive index with wavelength, such as the spectrum produced by a prism or a rainbow 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 22
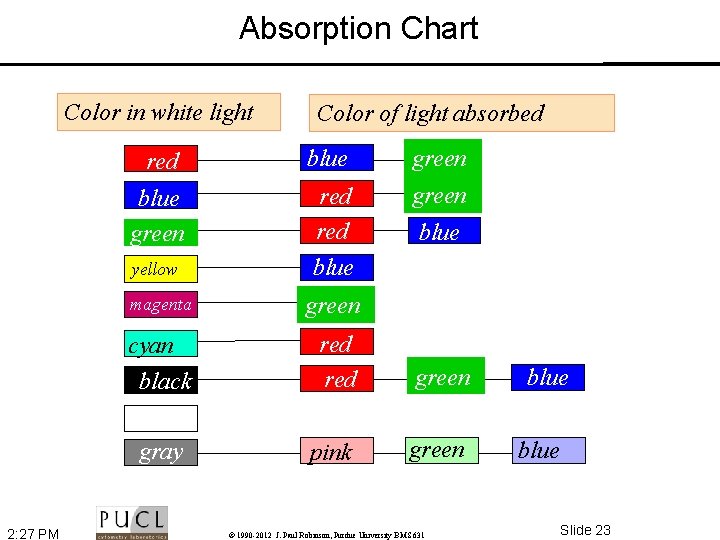
Absorption Chart Color in white light 2: 27 PM Color of light absorbed red blue green red green yellow blue magenta green cyan black red green gray pink green © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 blue Slide 23
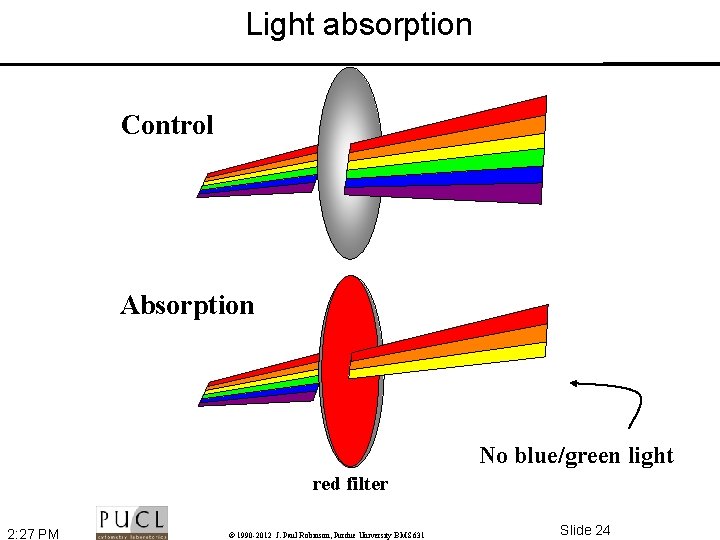
Light absorption Control Absorption No blue/green light red filter 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 24
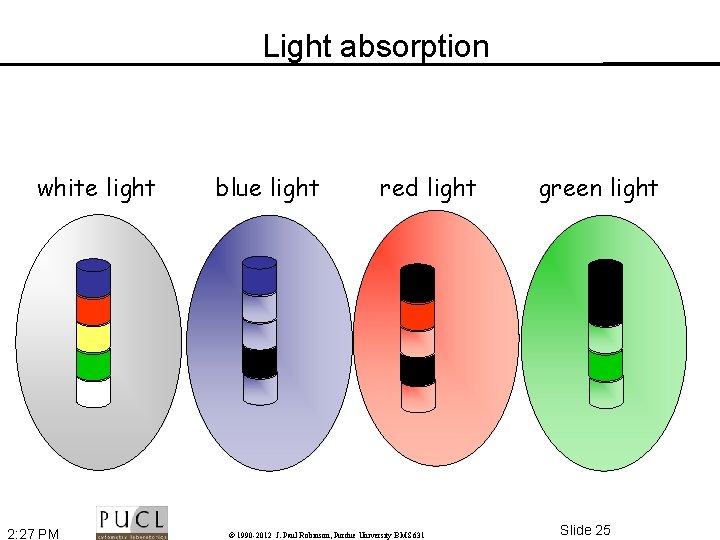
Light absorption white light 2: 27 PM blue light red light © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 green light Slide 25
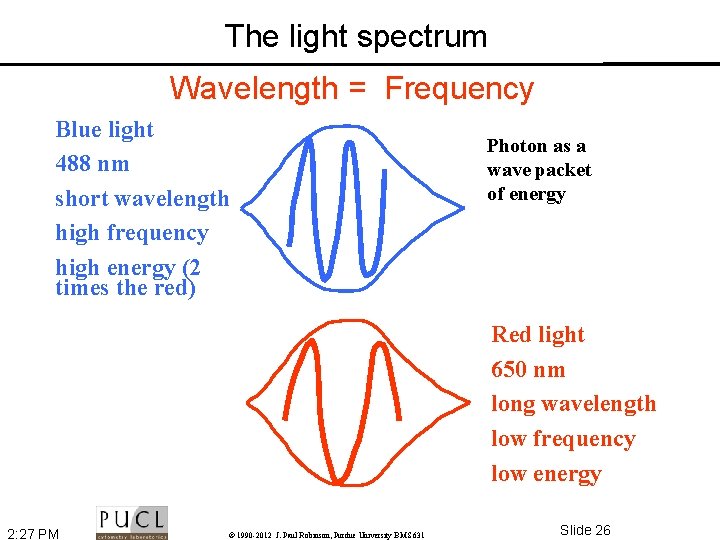
The light spectrum Wavelength = Frequency Blue light 488 nm short wavelength high frequency high energy (2 times the red) Photon as a wave packet of energy Red light 650 nm long wavelength low frequency low energy 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 26

Light Scatter • Materials scatter light at wavelengths at which they do not absorb • If we consider the visible spectrum to be 400 -750 nm then small particles (< 1/10 ) scatter rather than absorb light • For small particles (molecular up to sub micron) the Rayleigh scatter intensity at 0 o and 180 o are about the same • For larger particles (i. e. size from 1/4 to tens of wavelengths) larger amounts of scatter occur in the forward not the side scatter direction - this is called Mie Scatter (after Gustav Mie) - this is how we come up with forward scatter be related to size 3 rd Ed. Shapiro p 79 4 th Ed. Shapiro p 105 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 27

Mie scattering § Interaction between light and particles of size larger than the wavelength § Complete mathematical-physical theory of the scattering of electromagnetic radiation by spherical particles § Embraces all possible ratios of diameter to wavelength. § Assumes a homogeneous, isotropic and optically linear material irradiated by an infinitely extending plane wave http: //en. wikipedia. org/wiki/Mie_scattering 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 28

Rayleigh scattering § Particles much smaller than the wavelength of the light § short wavelengths are scattered more than long wavelengths § It occurs when light travels in transparent solids and liquids, but is most prominently seen in gases. § Dependent upon the size of the particles and the wavelength of the light. http: //en. wikipedia. org/wiki/Rayleigh_scattering 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 29

Rayleigh Scatter • Molecules and very small particles do not absorb, but scatter light in the visible region (same freq as excitation) • Rayleigh scattering is directly proportional to the electric dipole and inversely proportional to the 4 th power of the wavelength of the incident light the sky looks blue because the gas molecules scatter more light at shorter (blue) rather than longer wavelengths (red) 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 30

Some Key issues with regard to light • When you use a very bright light source to excite molecules in a cell, you also produce a large amount of reflected light • This light is what we measure when we measure forward and side scatter • Typically it can be 105 or 106 times the fluorescence signal produced • So what does this mean? 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 31

Introduction to light scatter in flow § Light scattering is a complex phenomenon. § If the refractive index is not different from the surrounding medium (which may be water) there will be a negligible difference between RI of cell and medium § Light scattering can be measured based on § light scattered by a particle suspension § light scattered by an individual particle § The scattered fields; scattering angle and/or wavelength are important § Particle parameters can be: § 2: 27 PM Size, morphology, viability, structure, effective refractive index, etc. © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 32
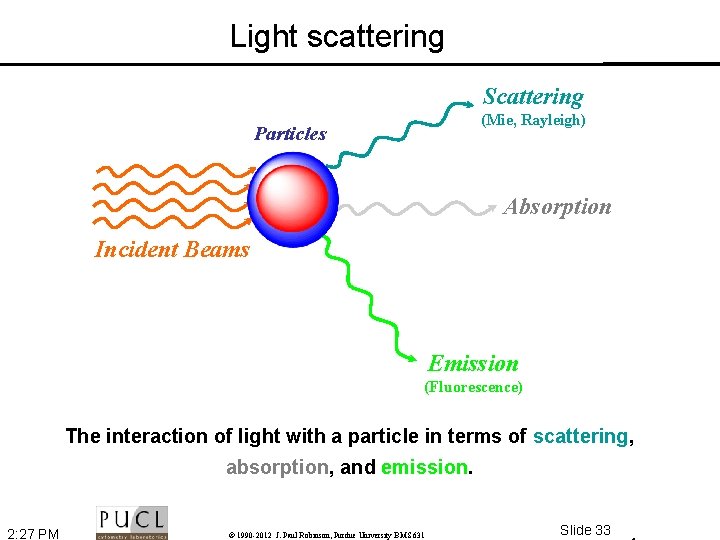
Light scattering Scattering (Mie, Rayleigh) Particles Absorption Incident Beams Emission (Fluorescence) The interaction of light with a particle in terms of scattering, absorption, and emission. 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 33

Light scattering in flow cytometry § Qualitative analyses of individual cells as well as particle suspension § The angular dependence of light-scattering intensity § Size and shape dependent: Forward scatter § dependence of scattered intensity on particle RI and geometrical cross-section § Structure dependent: Side scatter § anisotropic cell structures and multiple scattering processes 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 34

Forward & Side Scatter § Forward Scatter (FSC): Diffracted light §FSC is related to cell surface area (at narrow angles) §FSC is detected along axis of incident light in the forward direction §BUT – the accuracy for cell sizing is not good – it is very dependent upon the exact angle of scatter which can vary even between similar instruments § Side Scatter (SSC): Reflected and Refracted light §SSC is related to cell granularity and complexity §SSC is detected (mostly) at 90° to the laser beam § Factors that affect light scatter §Shape, surface, size, granularity, internal complexity, refractive index. 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 35
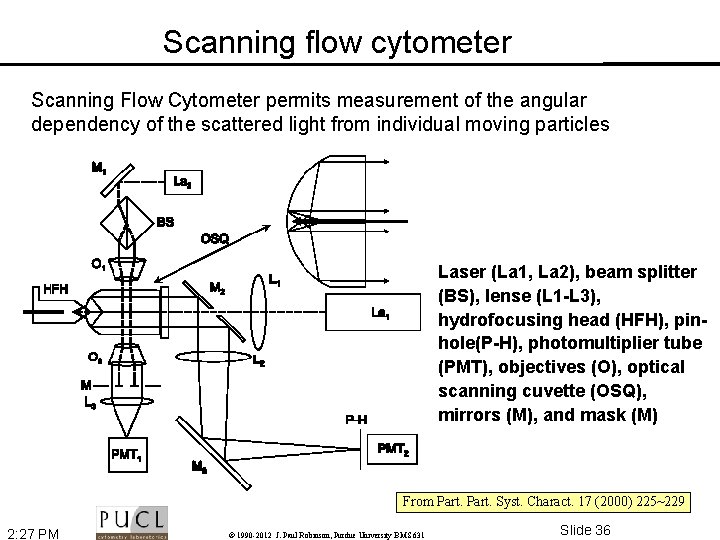
Scanning flow cytometer Scanning Flow Cytometer permits measurement of the angular dependency of the scattered light from individual moving particles Laser (La 1, La 2), beam splitter (BS), lense (L 1 -L 3), hydrofocusing head (HFH), pinhole(P-H), photomultiplier tube (PMT), objectives (O), optical scanning cuvette (OSQ), mirrors (M), and mask (M) From Part. Syst. Charact. 17 (2000) 225~229 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 36
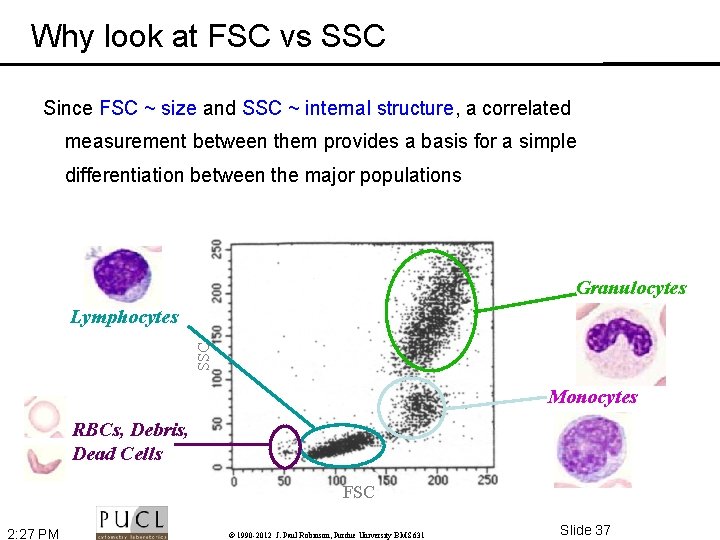
Why look at FSC vs SSC Since FSC ~ size and SSC ~ internal structure, a correlated measurement between them provides a basis for a simple differentiation between the major populations Granulocytes SSC Lymphocytes Monocytes RBCs, Debris, Dead Cells FSC 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 37
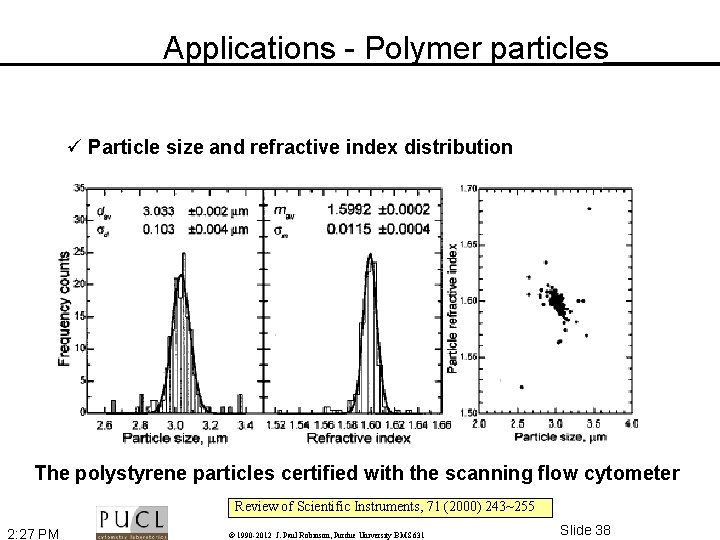
Applications - Polymer particles ü Particle size and refractive index distribution The polystyrene particles certified with the scanning flow cytometer Review of Scientific Instruments, 71 (2000) 243~255 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 38
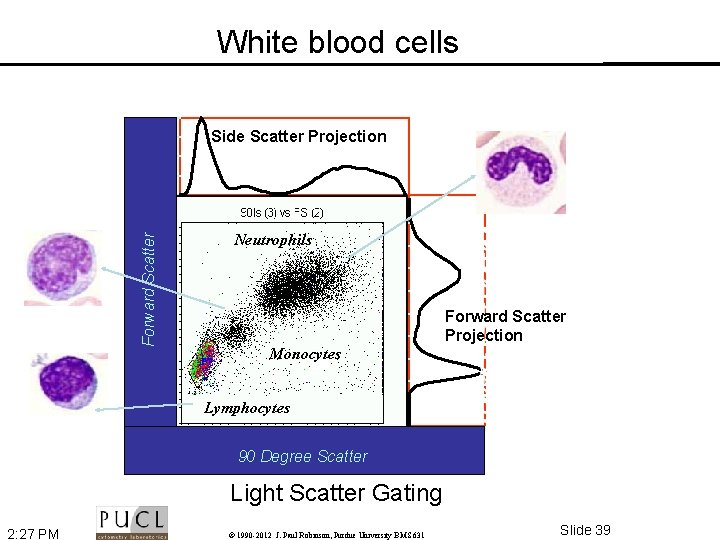
White blood cells Side Scatter Projection Forward Scatter Neutrophils Forward Scatter Projection Monocytes Lymphocytes 0 200 400 600 800 1000 90 Degree Scatter Light Scatter Gating 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 39
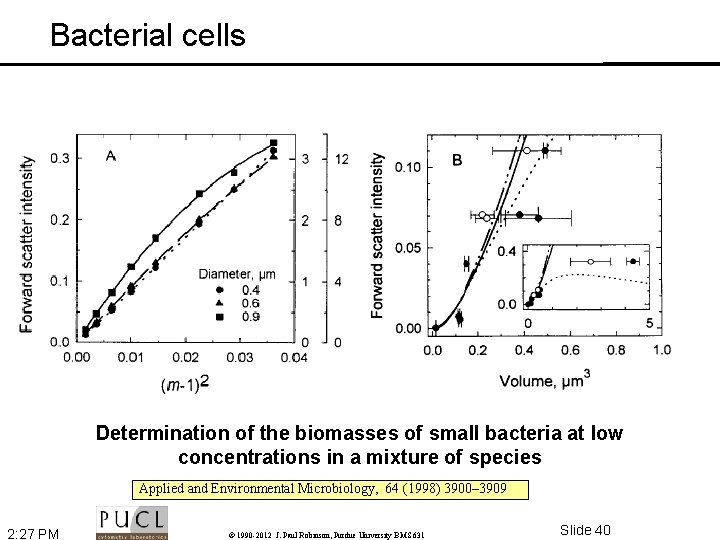
Bacterial cells Determination of the biomasses of small bacteria at low concentrations in a mixture of species Applied and Environmental Microbiology, 64 (1998) 3900– 3909 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 40

Lecture Summary • • Principles of light and matter Basic optics to take into consideration Essentials of light measurement Absorption and optical properties Angular dependency of light scattering intensity Forward scatter: Diffracted light Characterization of spherical and non-spherical particle • Side Scatter: Reflected and refracted light 2: 27 PM © 1990 -2012 J. Paul Robinson, Purdue University BMS 631 Slide 41
- Slides: 41