BME 301 Biomedical Sensors Lecture Note 3 Bioelectric

BME 301: Biomedical Sensors Lecture Note 3: Bioelectric Potentials and Biopotential Electrodes 1 BME 301 Lecture Note 3 - Ali Işın 2013

Bioelectric potentials BME 301 Lecture Note 3 - Ali Işın 2013 2
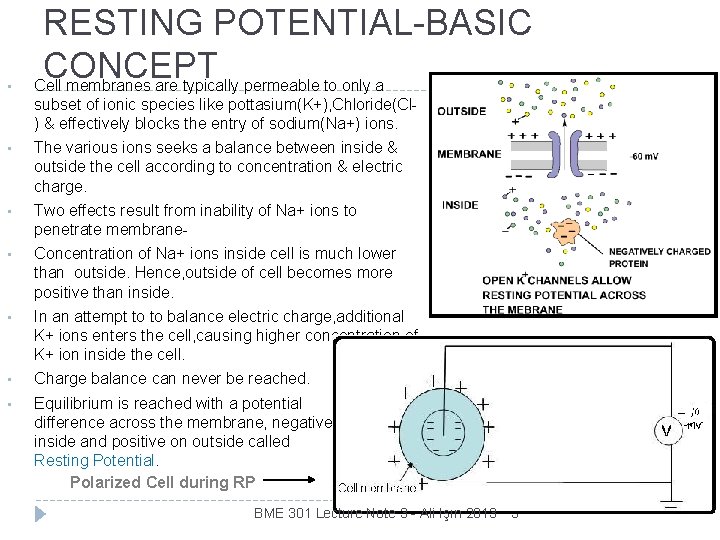
• RESTING POTENTIAL-BASIC CONCEPT Cell membranes are typically permeable to only a subset of ionic species like pottasium(K+), Chloride(Cl) & effectively blocks the entry of sodium(Na+) ions. • The various ions seeks a balance between inside & outside the cell according to concentration & electric charge. • Two effects result from inability of Na+ ions to penetrate membrane- • Concentration of Na+ ions inside cell is much lower than outside. Hence, outside of cell becomes more positive than inside. • In an attempt to to balance electric charge, additional K+ ions enters the cell, causing higher concentration of K+ ion inside the cell. • Charge balance can never be reached. • Equilibrium is reached with a potential difference across the membrane, negative on inside and positive on outside called Resting Potential. Polarized Cell during RP BME 301 Lecture Note 3 - Ali Işın 2013 3
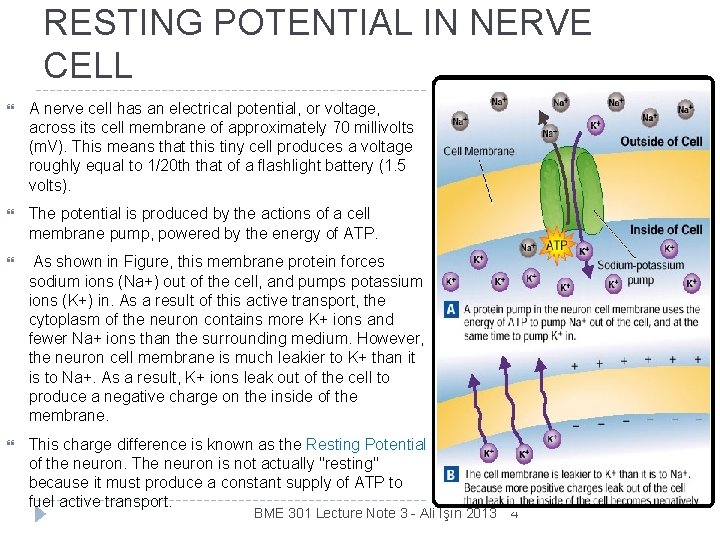
RESTING POTENTIAL IN NERVE CELL A nerve cell has an electrical potential, or voltage, across its cell membrane of approximately 70 millivolts (m. V). This means that this tiny cell produces a voltage roughly equal to 1/20 th that of a flashlight battery (1. 5 volts). The potential is produced by the actions of a cell membrane pump, powered by the energy of ATP. As shown in Figure, this membrane protein forces sodium ions (Na+) out of the cell, and pumps potassium ions (K+) in. As a result of this active transport, the cytoplasm of the neuron contains more K+ ions and fewer Na+ ions than the surrounding medium. However, the neuron cell membrane is much leakier to K+ than it is to Na+. As a result, K+ ions leak out of the cell to produce a negative charge on the inside of the membrane. This charge difference is known as the Resting Potential of the neuron. The neuron is not actually "resting" because it must produce a constant supply of ATP to fuel active transport. BME 301 Lecture Note 3 - Ali Işın 2013 4
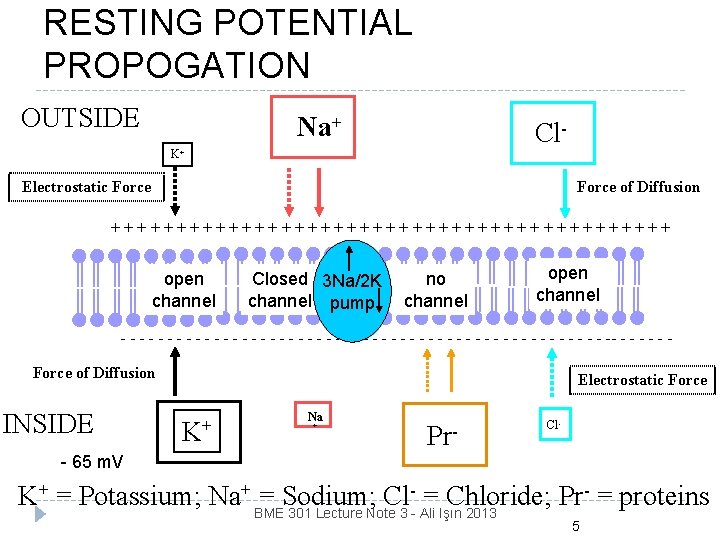
RESTING POTENTIAL PROPOGATION OUTSIDE Na+ Cl- K+ Electrostatic Force of Diffusion ++++++++++++++++++++++ open channel Closed 3 Na/2 K channel pump no channel open channel - - - - - - - - - - - - - -- - - Force of Diffusion INSIDE Electrostatic Force K+ Na + Pr- Cl- - 65 m. V K+ = Potassium; Na+ BME 301 Lecture Note 3 - Ali Işın 2013 = Sodium; Cl- = Chloride; Pr- = proteins 5
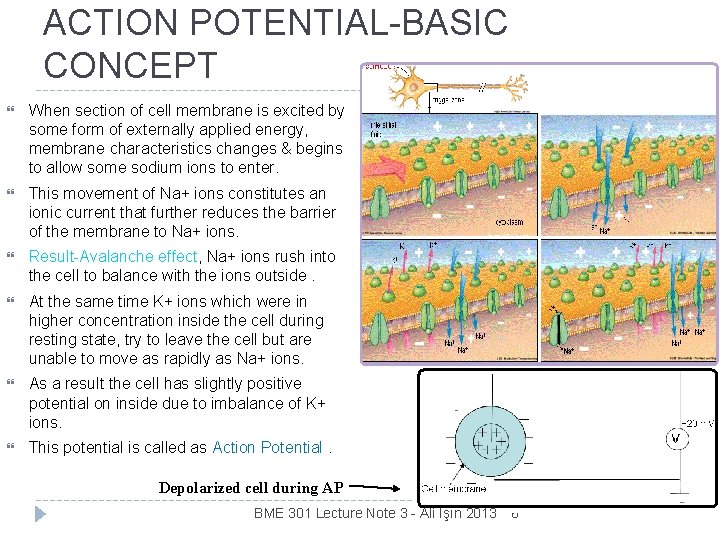
ACTION POTENTIAL-BASIC CONCEPT When section of cell membrane is excited by some form of externally applied energy, membrane characteristics changes & begins to allow some sodium ions to enter. This movement of Na+ ions constitutes an ionic current that further reduces the barrier of the membrane to Na+ ions. Result-Avalanche effect, Na+ ions rush into the cell to balance with the ions outside. At the same time K+ ions which were in higher concentration inside the cell during resting state, try to leave the cell but are unable to move as rapidly as Na+ ions. As a result the cell has slightly positive potential on inside due to imbalance of K+ ions. This potential is called as Action Potential. Depolarized cell during AP BME 301 Lecture Note 3 - Ali Işın 2013 6
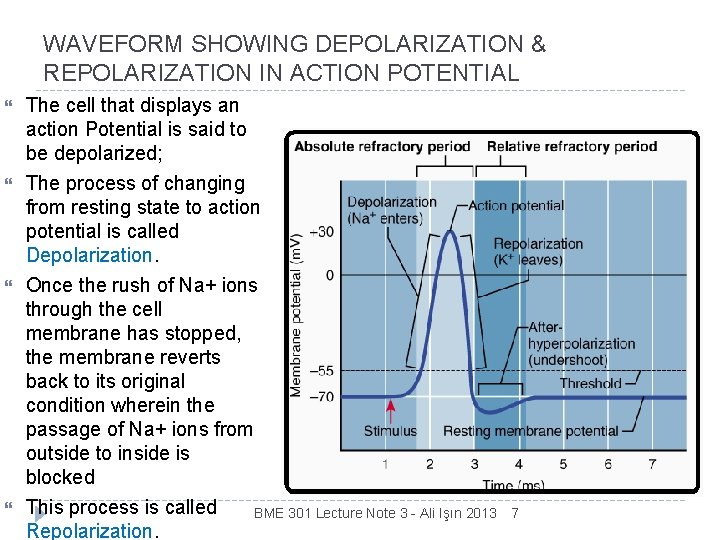
WAVEFORM SHOWING DEPOLARIZATION & REPOLARIZATION IN ACTION POTENTIAL The cell that displays an action Potential is said to be depolarized; The process of changing from resting state to action potential is called Depolarization. Once the rush of Na+ ions through the cell membrane has stopped, the membrane reverts back to its original condition wherein the passage of Na+ ions from outside to inside is blocked This process is called BME 301 Lecture Note 3 - Ali Işın 2013 Repolarization. 7
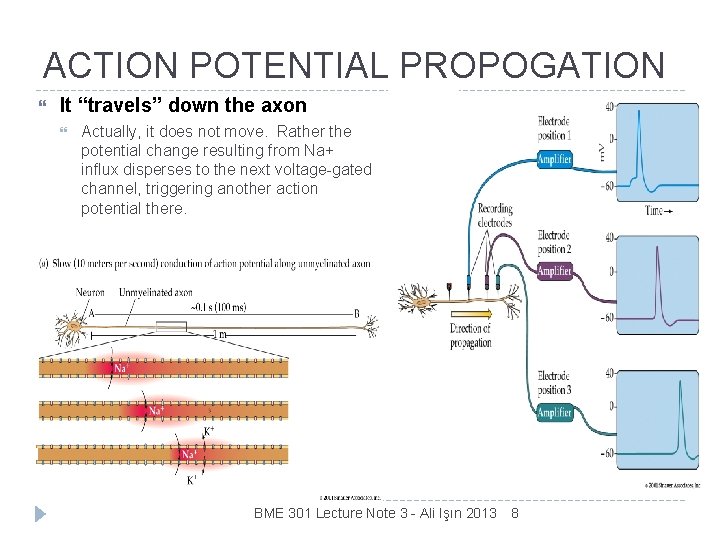
ACTION POTENTIAL PROPOGATION It “travels” down the axon Actually, it does not move. Rather the potential change resulting from Na+ influx disperses to the next voltage-gated channel, triggering another action potential there. BME 301 Lecture Note 3 - Ali Işın 2013 8
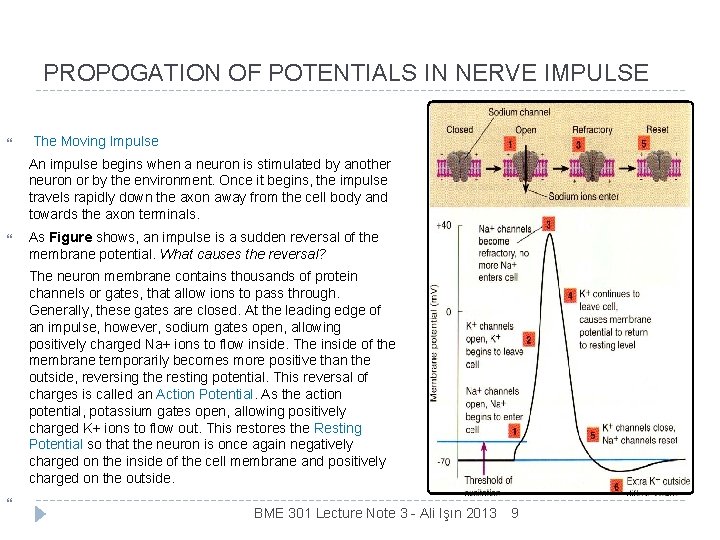
PROPOGATION OF POTENTIALS IN NERVE IMPULSE The Moving Impulse An impulse begins when a neuron is stimulated by another neuron or by the environment. Once it begins, the impulse travels rapidly down the axon away from the cell body and towards the axon terminals. As Figure shows, an impulse is a sudden reversal of the membrane potential. What causes the reversal? The neuron membrane contains thousands of protein channels or gates, that allow ions to pass through. Generally, these gates are closed. At the leading edge of an impulse, however, sodium gates open, allowing positively charged Na+ ions to flow inside. The inside of the membrane temporarily becomes more positive than the outside, reversing the resting potential. This reversal of charges is called an Action Potential. As the action potential, potassium gates open, allowing positively charged K+ ions to flow out. This restores the Resting Potential so that the neuron is once again negatively charged on the inside of the cell membrane and positively charged on the outside. BME 301 Lecture Note 3 - Ali Işın 2013 9

A nerve impulse is self-propagating. That is, an impulse at any point on the membrane causes an impulse at the next point along the membrane. We might compare the flow of an impulse to the fall of a row of dominoes. As each domino falls, it causes its neighbour to fall. Then, as the impulse passes, the dominoes set themselves up again, ready for another Action Potential. 10 BME 301 Lecture Note 3 - Ali Işın 2013

Resting and action potentials The resting potential is the result of an unequal distribution of ions across the membrane. The resting potential is sensitive to ions in proportion to their ability to permeate the membrane. BME 301 Lecture Note 3 - Ali Işın 2013 11

Resting potentials Forget the membrane and consider what factors determine the movement of ions in solution. Aqueous diffusion and Electrophoretic movement BME 301 Lecture Note 3 - Ali Işın 2013 12
Resting potentials 0 m. V BME 301 Lecture Note 3 - Ali Işın 2013 13
Resting potentials 0 m. V BME 301 Lecture Note 3 - Ali Işın 2013 14
Resting potentials -80 m. V BME 301 Lecture Note 3 - Ali Işın 2013 15
Resting potentials + + + - - - + - -80 m. V - 16 - BME 301 Lecture Note 3 - Ali Işın 2013
![Resting potentials [K+] = 2. 5 [Na+] = 125 [Cl-] = 130 A+ + Resting potentials [K+] = 2. 5 [Na+] = 125 [Cl-] = 130 A+ +](http://slidetodoc.com/presentation_image_h/8e7b4c95d0375a2c177d0b61f2e5c672/image-17.jpg)
Resting potentials [K+] = 2. 5 [Na+] = 125 [Cl-] = 130 A+ + + - - - + - -80 m. V [K+] = 135 [Na+] = 7 [Cl-] = 11 BME 301 Lecture Note 3 - Ali Işın 2013 A-17
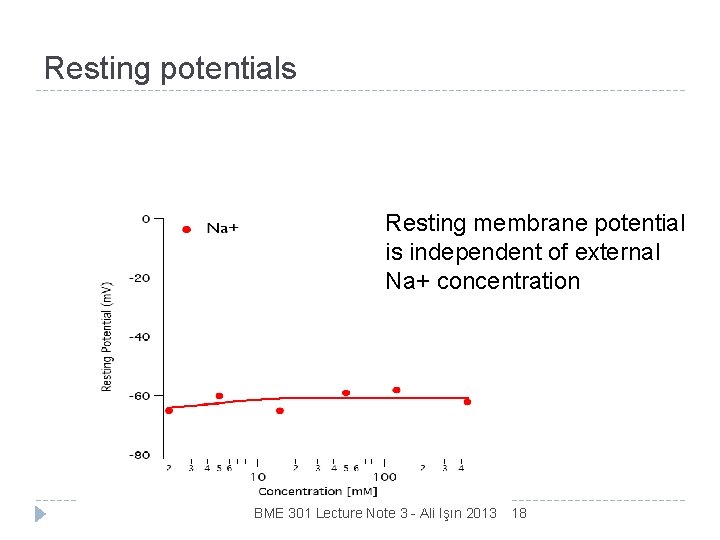
Resting potentials Resting membrane potential is independent of external Na+ concentration BME 301 Lecture Note 3 - Ali Işın 2013 18
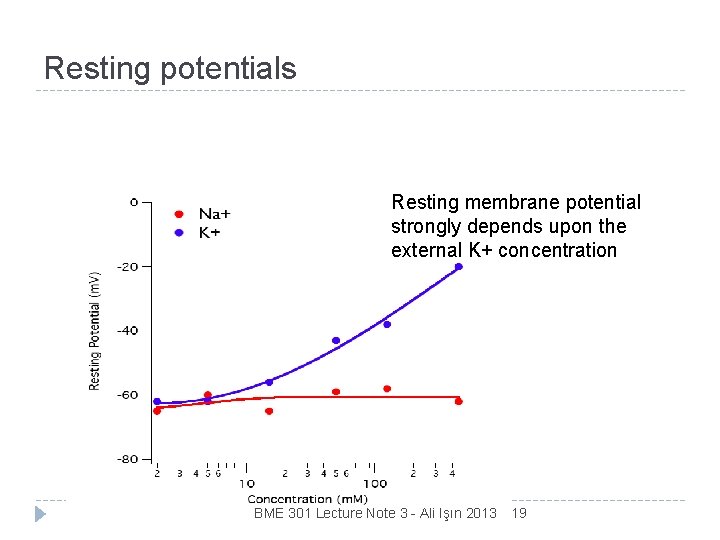
Resting potentials Resting membrane potential strongly depends upon the external K+ concentration BME 301 Lecture Note 3 - Ali Işın 2013 19

Summary The membrane conducts ions very poorly and allows the separation of ionic species. This results in a potential difference between the outside and the inside of the membrane. The magnitude of the resting potential is determined by the selective permeability of the membrane to ionic species. We can quantify the magnitude of the resting potential by considering both the diffusive and electrophoretic properties. In order to understand the time dependence and individual contributions of ionic species to the membrane potential it is convenient to use an electrical equivalent circuit. BME 301 Lecture Note 3 - Ali Işın 2013 20
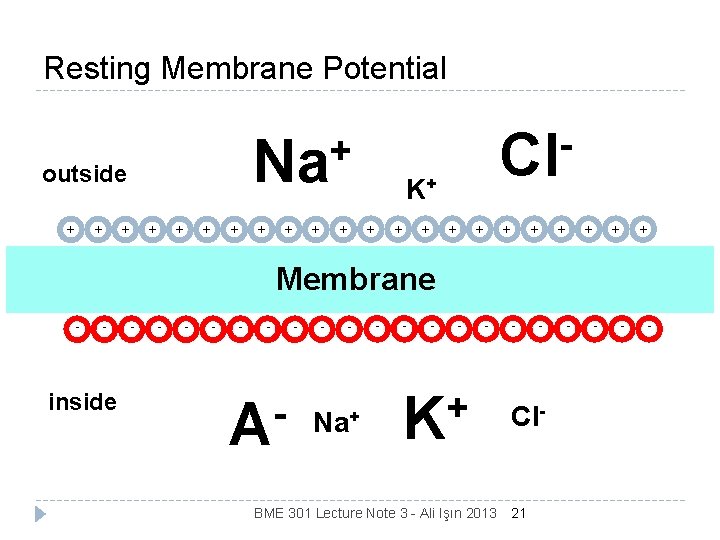
Resting Membrane Potential + + + K+ + + outside Cl + + Na + + + + Membrane - - - BME 301 Lecture Note 3 - Ali Işın 2013 Cl- 21 - + K - Na+ - - A - - - - inside

Membrane is polarized more negative particles in than out Bioelectric Potential like a battery Potential for ion movement current ~ BME 301 Lecture Note 3 - Ali Işın 2013 22
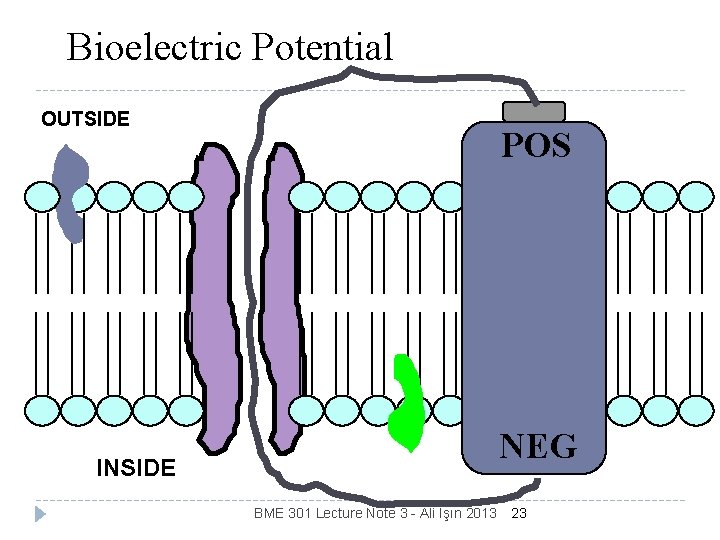
Bioelectric Potential OUTSIDE POS NEG INSIDE BME 301 Lecture Note 3 - Ali Işın 2013 23

Biopotentials ECG EEG electromyography ERG electroencephalography EMG electrocardiogrphy electroretinograpy EOG… electrooculography BME 301 Lecture Note 3 - Ali Işın 2013 24
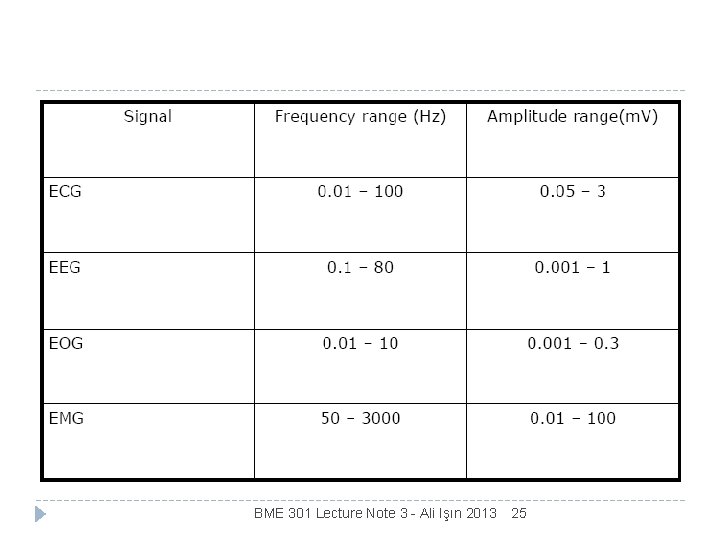
Frequencies of Biopotentials BME 301 Lecture Note 3 - Ali Işın 2013 25
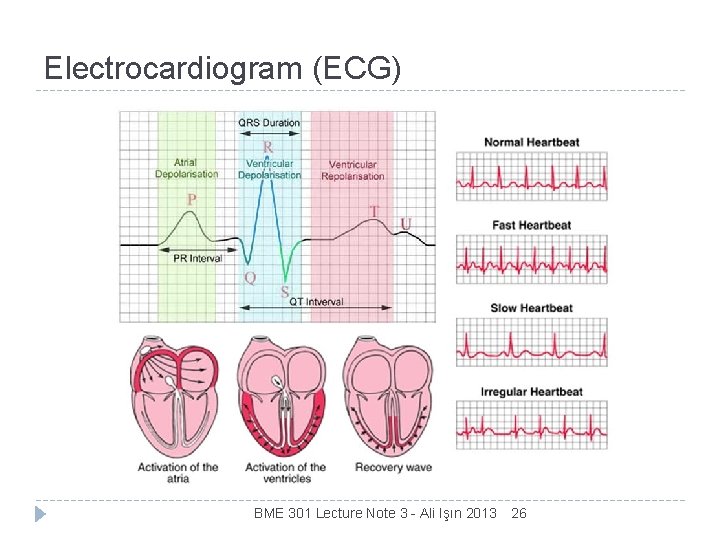
Electrocardiogram (ECG) BME 301 Lecture Note 3 - Ali Işın 2013 26
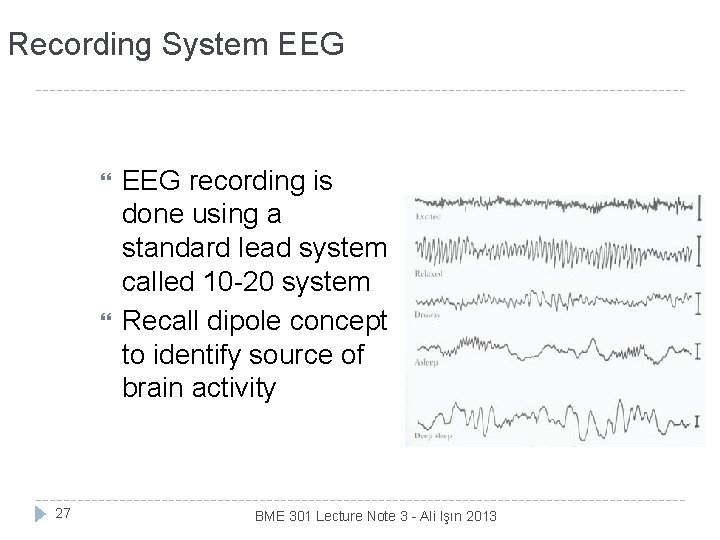
Recording System EEG 27 EEG recording is done using a standard lead system called 10 -20 system Recall dipole concept to identify source of brain activity BME 301 Lecture Note 3 - Ali Işın 2013

Electromyogram (EMG) Measures muscle activity Recordintramuscularly through needle electrodes Record surface EMG using electrodes on biceps, triceps… Use in muscular disorders, muscle based prosthesis –prosthetic arm, leg BME 301 Lecture Note 3 - Ali Işın 2013 28

Electroretinogram (ERG) Biopotential of the eye (retina) Indicator of retinal diseases such as retinal degenration, macular degernation Invasive recording BME 301 Lecture Note 3 - Ali Işın 2013 29
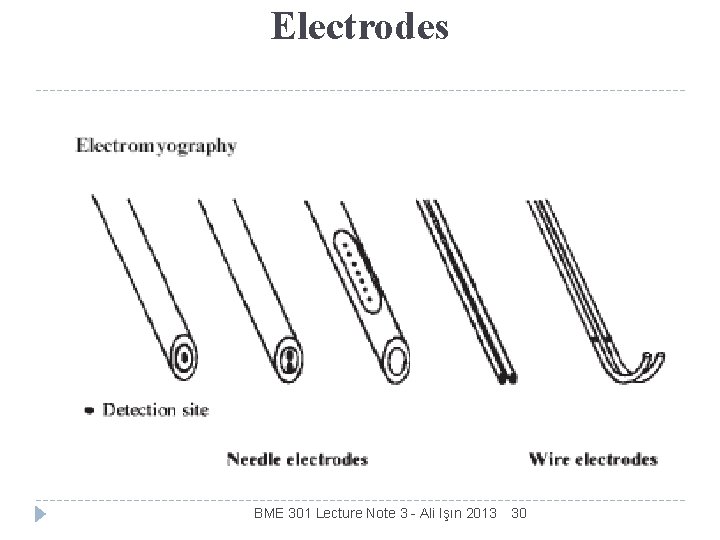
Electrodes KINDS OF ELECTRODES BME 301 Lecture Note 3 - Ali Işın 2013 30
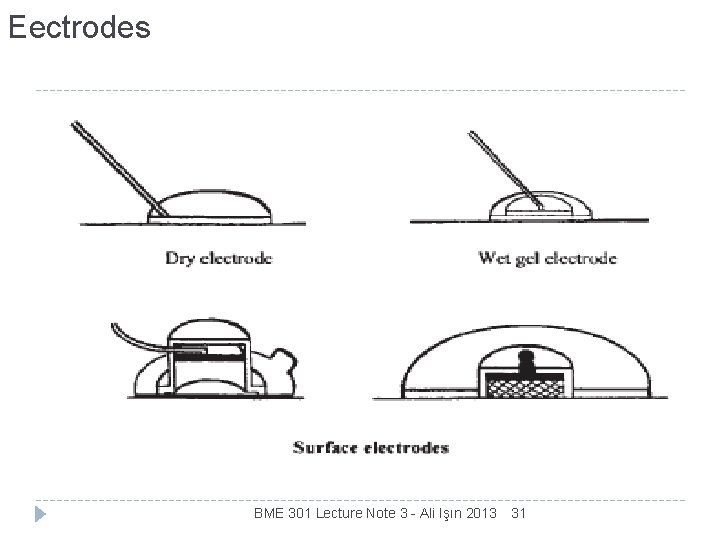
Eectrodes BME 301 Lecture Note 3 - Ali Işın 2013 31
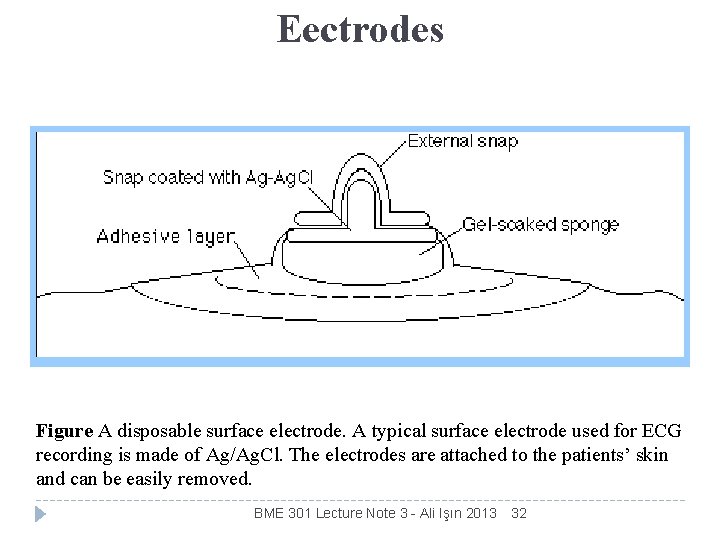
Eectrodes Figure A disposable surface electrode. A typical surface electrode used for ECG recording is made of Ag/Ag. Cl. The electrodes are attached to the patients’ skin and can be easily removed. BME 301 Lecture Note 3 - Ali Işın 2013 32

Biopotential Electrodes BME 301 Lecture Note 3 - Ali Işın 2013 33
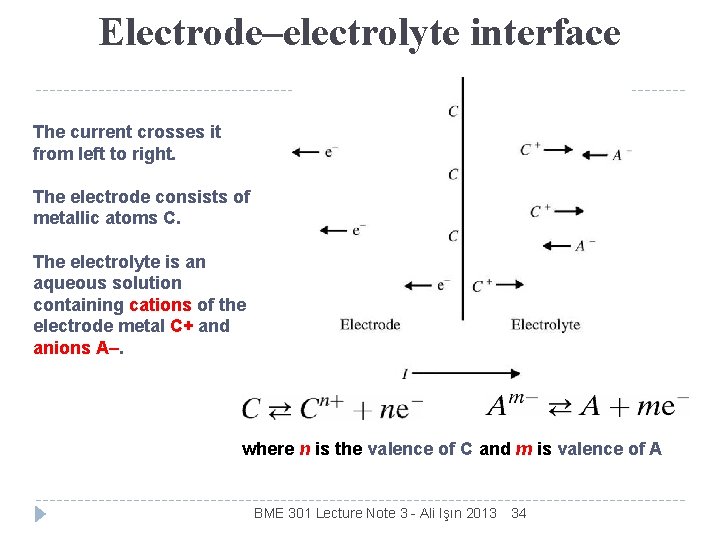
Electrode–electrolyte interface The current crosses it from left to right. The electrode consists of metallic atoms C. The electrolyte is an aqueous solution containing cations of the electrode metal C+ and anions A–. where n is the valence of C and m is valence of A BME 301 Lecture Note 3 - Ali Işın 2013 34

BME 301 Lecture Note 3 - Ali Işın 2013 35

Vp = total patential, or polarization potential, of the electrode E 0 = half-cell potential Vr = ohmic overpotential Vc = concentration overpotential Va = activation overpotential BME 301 Lecture Note 3 - Ali Işın 2013 36
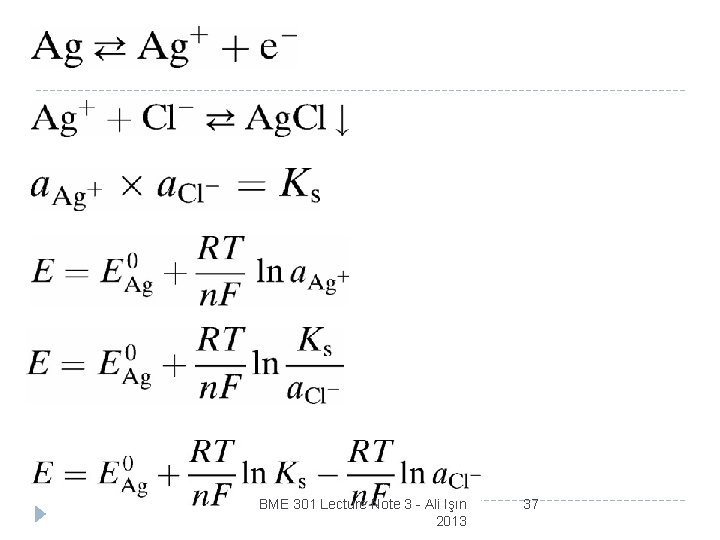
BME 301 Lecture Note 3 - Ali Işın 2013 37
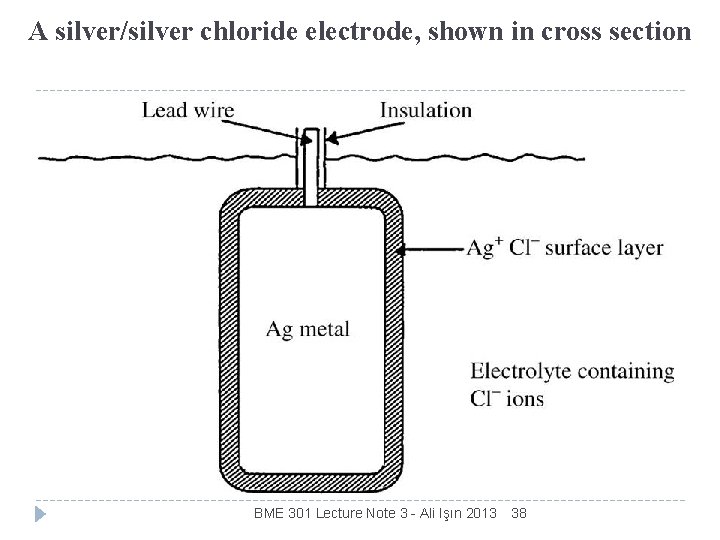
A silver/silver chloride electrode, shown in cross section BME 301 Lecture Note 3 - Ali Işın 2013 38

1. 73 10– 10 142. 3 BME 301 Lecture Note 3 - Ali Işın = 2. 46 10– 8 g 2013 39
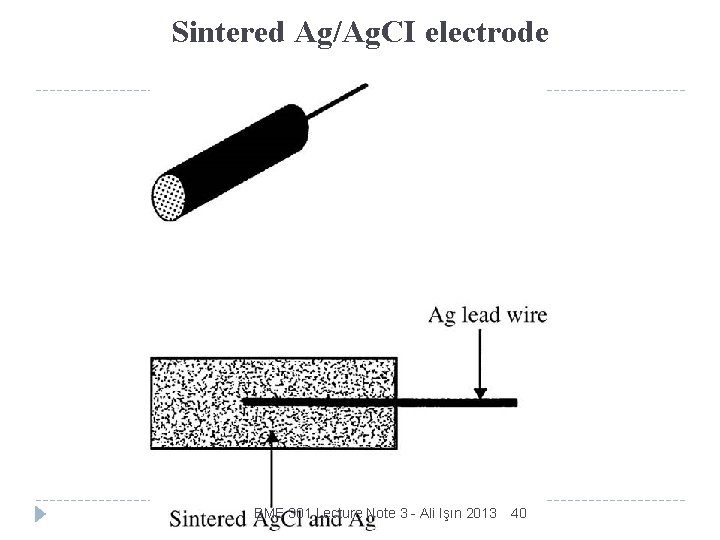
Sintered Ag/Ag. CI electrode BME 301 Lecture Note 3 - Ali Işın 2013 40
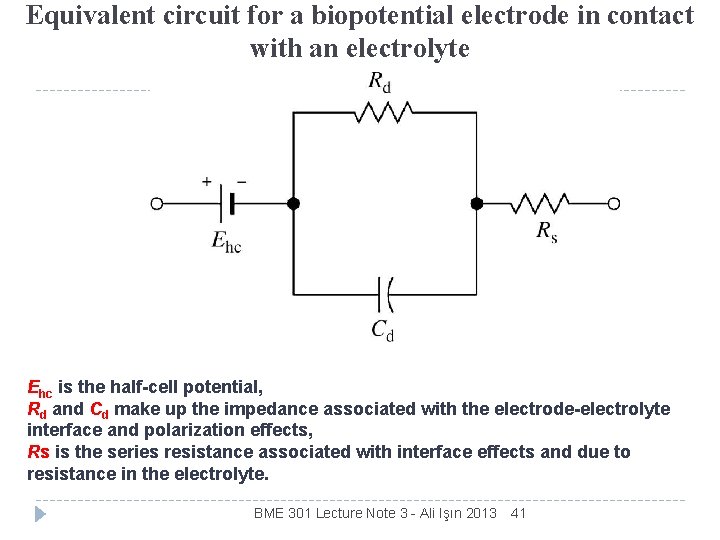
Equivalent circuit for a biopotential electrode in contact with an electrolyte Ehc is the half-cell potential, Rd and Cd make up the impedance associated with the electrode-electrolyte interface and polarization effects, Rs is the series resistance associated with interface effects and due to resistance in the electrolyte. BME 301 Lecture Note 3 - Ali Işın 2013 41
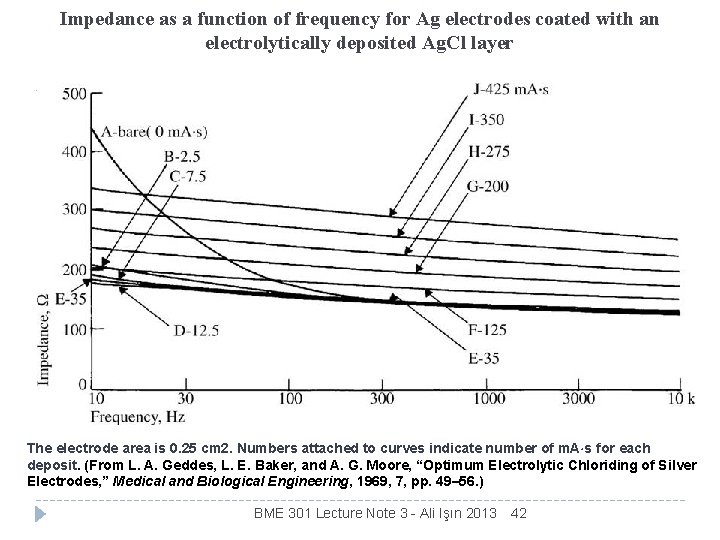
Impedance as a function of frequency for Ag electrodes coated with an electrolytically deposited Ag. Cl layer The electrode area is 0. 25 cm 2. Numbers attached to curves indicate number of m. A s for each deposit. (From L. A. Geddes, L. E. Baker, and A. G. Moore, “Optimum Electrolytic Chloriding of Silver Electrodes, ” Medical and Biological Engineering, 1969, 7, pp. 49– 56. ) BME 301 Lecture Note 3 - Ali Işın 2013 42
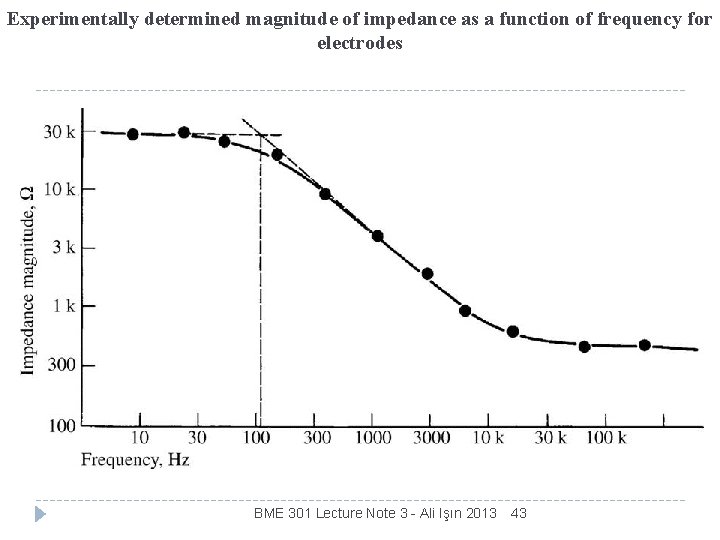
Experimentally determined magnitude of impedance as a function of frequency for electrodes BME 301 Lecture Note 3 - Ali Işın 2013 43

Example We want to develop an electrical model for a specific biopotential electrode studies in the laboratory. The electrode is characterized by placing it in a physiological saline bath in the laboratory, along with an Ag/Ag. Cl electrode having a much greater surface area and a known half-cell potential of 0. 233 V. The dc voltage between the two electrodes is measured with a very-high-impedance voltmeter and found to be 0. 572 V with the test electrode negative The magnitude of the impedance between two electrodes is measured as a function of frequency at very low currents; it is found to be that given in Figure in slide 12. From these data, determine a circuit model for the electrode. BME 301 Lecture Note 3 - Ali Işın 2013 44
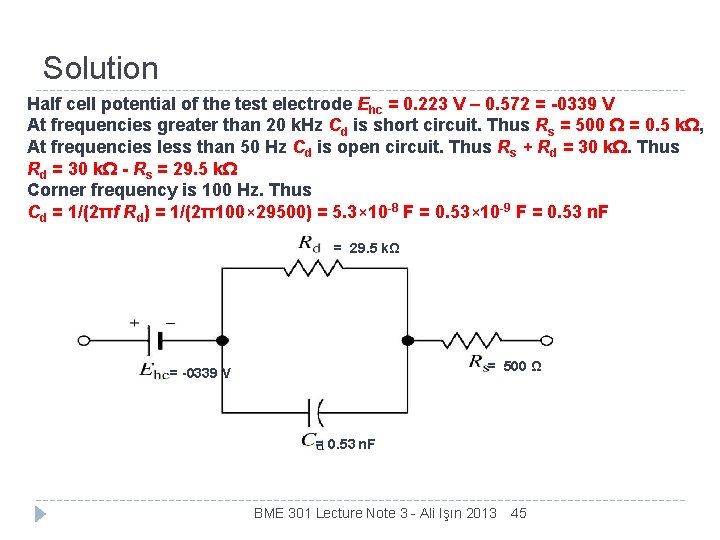
Solution Half cell potential of the test electrode Ehc = 0. 223 V – 0. 572 = -0339 V At frequencies greater than 20 k. Hz Cd is short circuit. Thus Rs = 500 Ω = 0. 5 kΩ, At frequencies less than 50 Hz Cd is open circuit. Thus Rs + Rd = 30 kΩ. Thus Rd = 30 kΩ - Rs = 29. 5 kΩ Corner frequency is 100 Hz. Thus Cd = 1/(2πf Rd) = 1/(2π100× 29500) = 5. 3× 10 -8 F = 0. 53× 10 -9 F = 0. 53 n. F = 29. 5 kΩ = 500 Ω = -0339 V = 0. 53 n. F BME 301 Lecture Note 3 - Ali Işın 2013 45
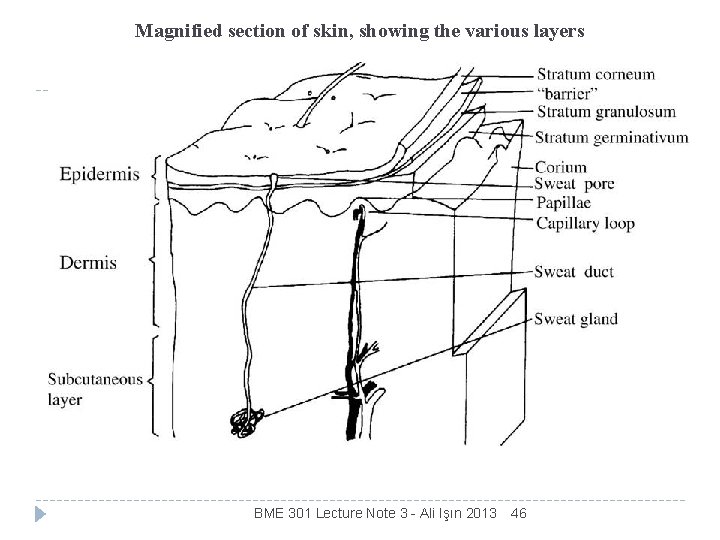
Magnified section of skin, showing the various layers BME 301 Lecture Note 3 - Ali Işın 2013 46
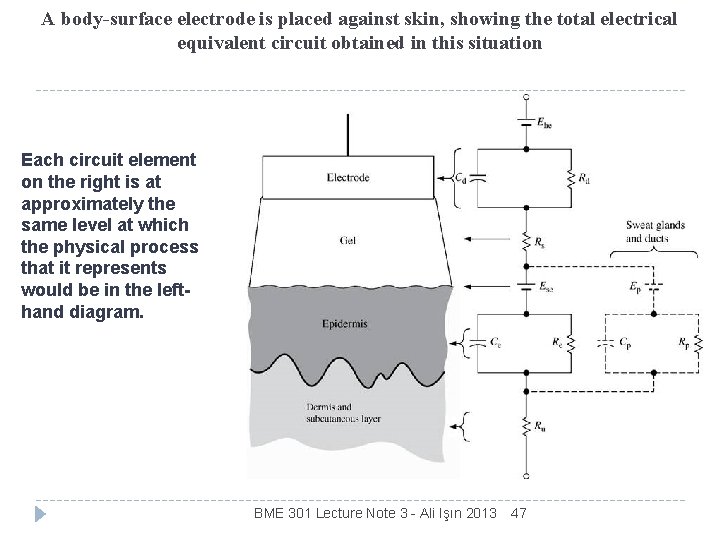
A body-surface electrode is placed against skin, showing the total electrical equivalent circuit obtained in this situation Each circuit element on the right is at approximately the same level at which the physical process that it represents would be in the lefthand diagram. BME 301 Lecture Note 3 - Ali Işın 2013 47
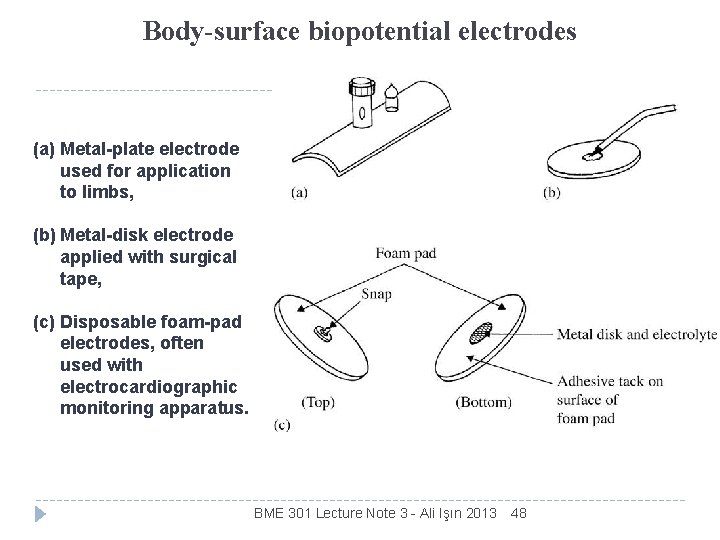
Body-surface biopotential electrodes (a) Metal-plate electrode used for application to limbs, (b) Metal-disk electrode applied with surgical tape, (c) Disposable foam-pad electrodes, often used with electrocardiographic monitoring apparatus. BME 301 Lecture Note 3 - Ali Işın 2013 48
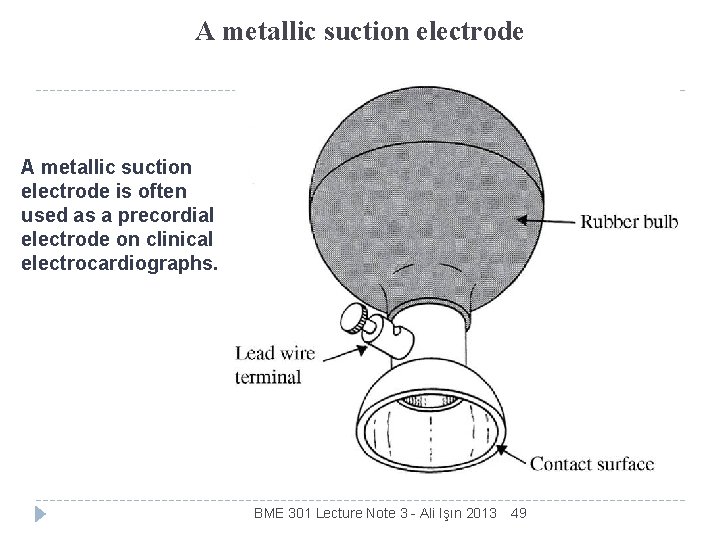
A metallic suction electrode is often used as a precordial electrode on clinical electrocardiographs. BME 301 Lecture Note 3 - Ali Işın 2013 49
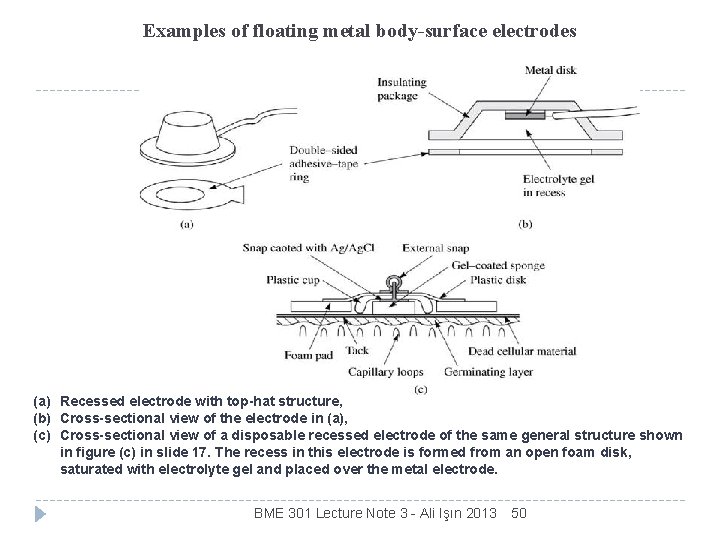
Examples of floating metal body-surface electrodes (a) Recessed electrode with top-hat structure, (b) Cross-sectional view of the electrode in (a), (c) Cross-sectional view of a disposable recessed electrode of the same general structure shown in figure (c) in slide 17. The recess in this electrode is formed from an open foam disk, saturated with electrolyte gel and placed over the metal electrode. BME 301 Lecture Note 3 - Ali Işın 2013 50
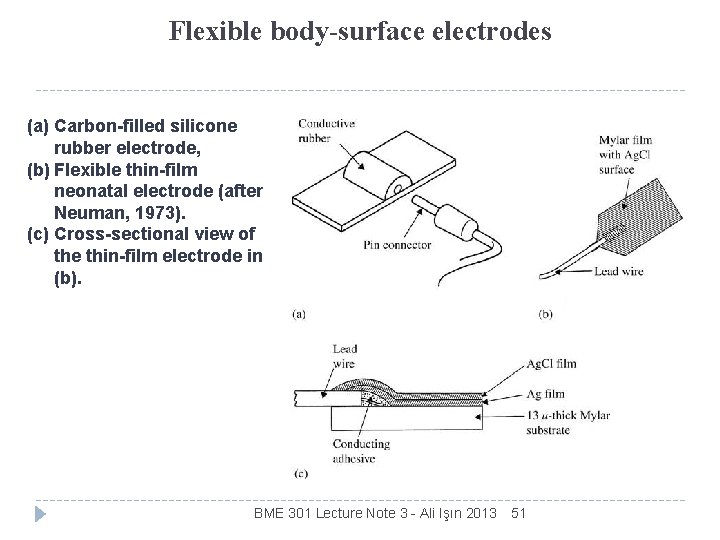
Flexible body-surface electrodes (a) Carbon-filled silicone rubber electrode, (b) Flexible thin-film neonatal electrode (after Neuman, 1973). (c) Cross-sectional view of the thin-film electrode in (b). BME 301 Lecture Note 3 - Ali Işın 2013 51
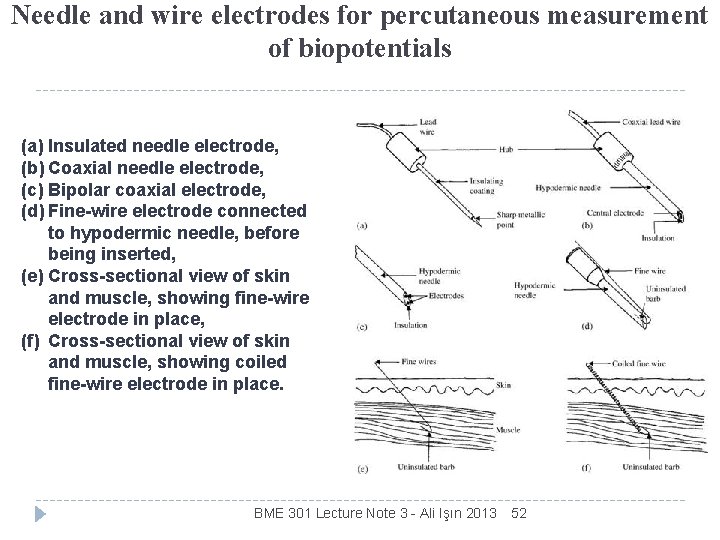
Needle and wire electrodes for percutaneous measurement of biopotentials (a) Insulated needle electrode, (b) Coaxial needle electrode, (c) Bipolar coaxial electrode, (d) Fine-wire electrode connected to hypodermic needle, before being inserted, (e) Cross-sectional view of skin and muscle, showing fine-wire electrode in place, (f) Cross-sectional view of skin and muscle, showing coiled fine-wire electrode in place. BME 301 Lecture Note 3 - Ali Işın 2013 52
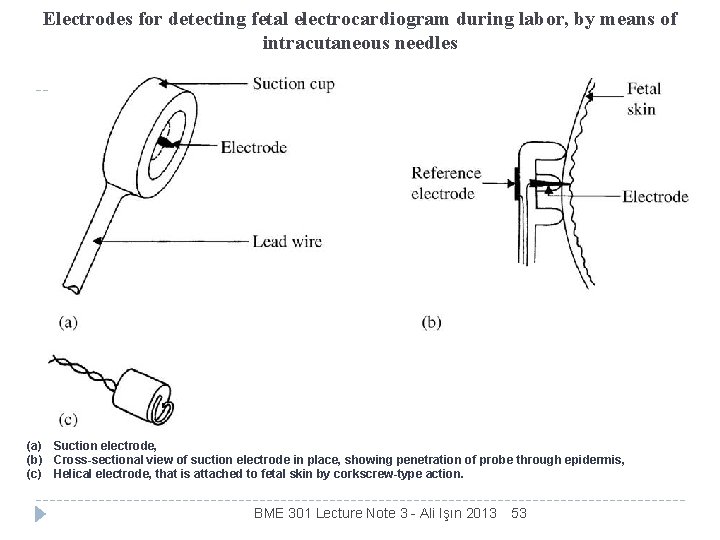
Electrodes for detecting fetal electrocardiogram during labor, by means of intracutaneous needles (a) Suction electrode, (b) Cross-sectional view of suction electrode in place, showing penetration of probe through epidermis, (c) Helical electrode, that is attached to fetal skin by corkscrew-type action. BME 301 Lecture Note 3 - Ali Işın 2013 53
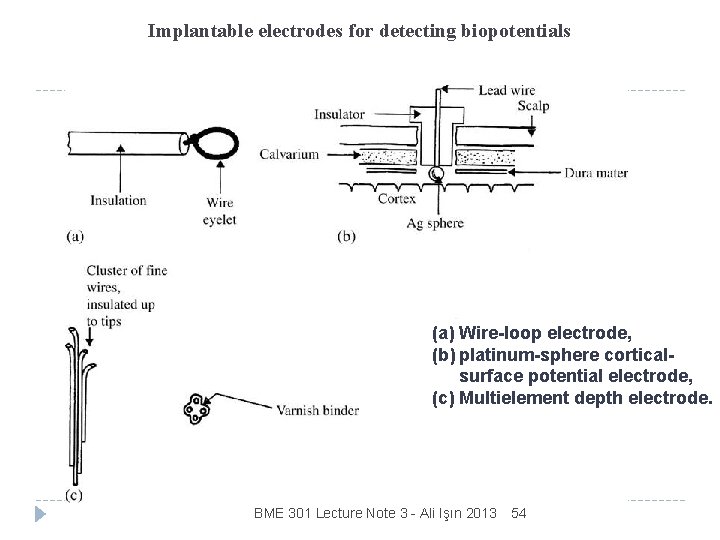
Implantable electrodes for detecting biopotentials (a) Wire-loop electrode, (b) platinum-sphere corticalsurface potential electrode, (c) Multielement depth electrode. BME 301 Lecture Note 3 - Ali Işın 2013 54
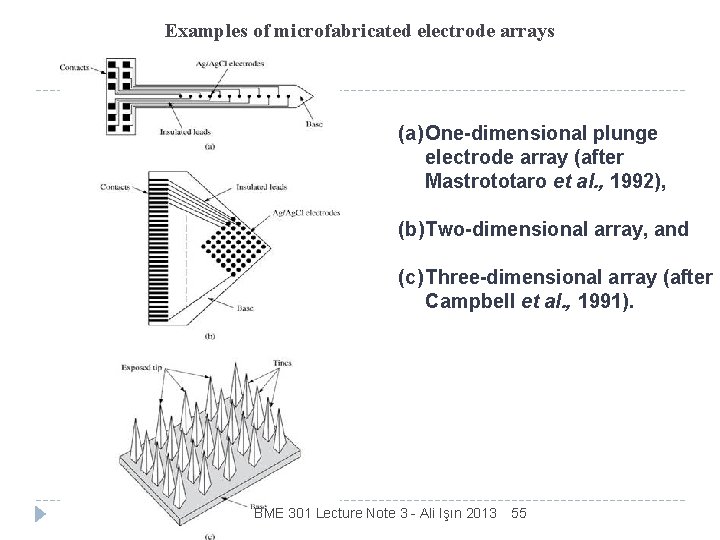
Examples of microfabricated electrode arrays (a) One-dimensional plunge electrode array (after Mastrototaro et al. , 1992), (b) Two-dimensional array, and (c) Three-dimensional array (after Campbell et al. , 1991). BME 301 Lecture Note 3 - Ali Işın 2013 55
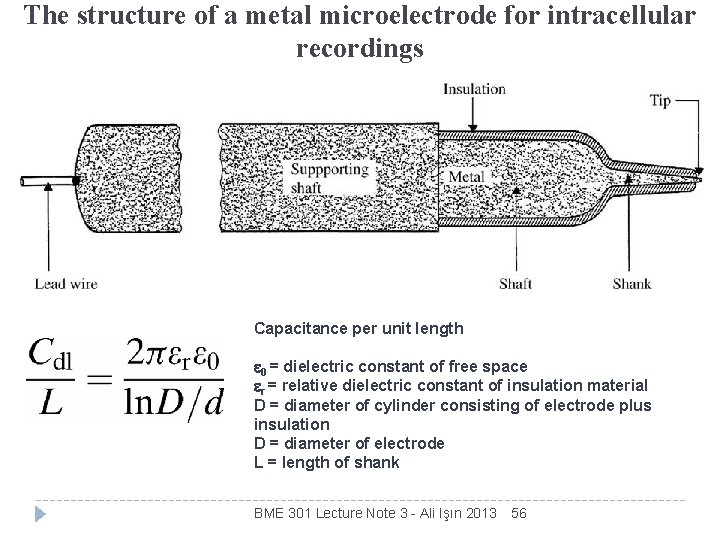
The structure of a metal microelectrode for intracellular recordings Capacitance per unit length 0 = dielectric constant of free space r = relative dielectric constant of insulation material D = diameter of cylinder consisting of electrode plus insulation D = diameter of electrode L = length of shank BME 301 Lecture Note 3 - Ali Işın 2013 56
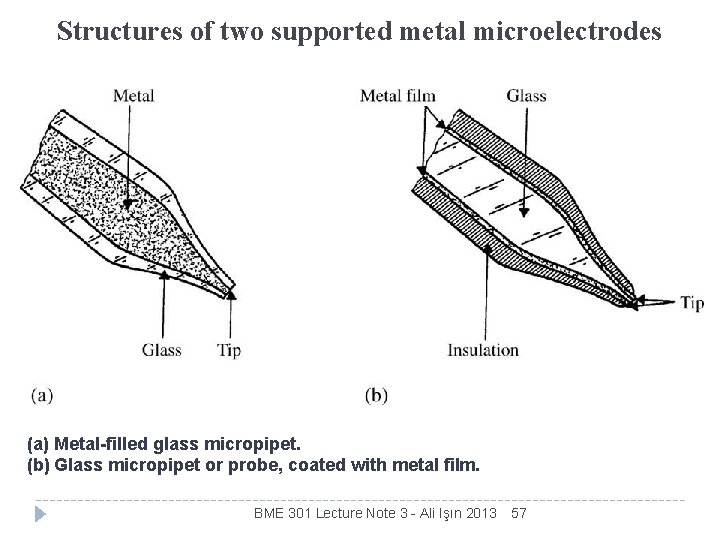
Structures of two supported metal microelectrodes (a) Metal-filled glass micropipet. (b) Glass micropipet or probe, coated with metal film. BME 301 Lecture Note 3 - Ali Işın 2013 57
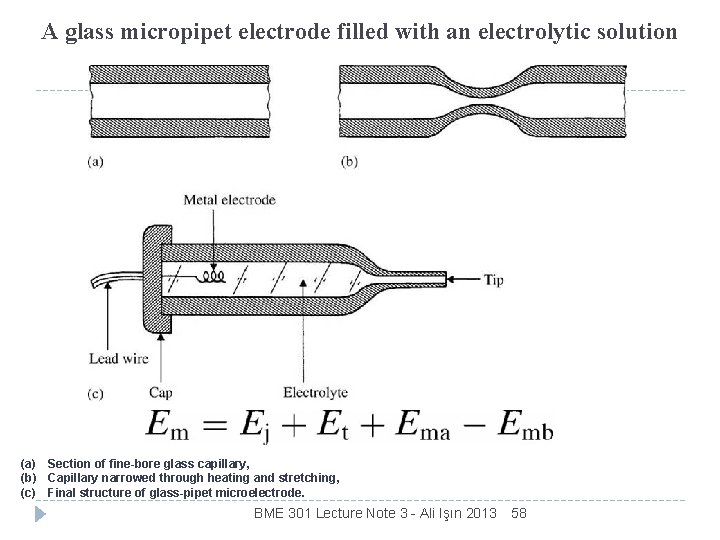
A glass micropipet electrode filled with an electrolytic solution (a) Section of fine-bore glass capillary, (b) Capillary narrowed through heating and stretching, (c) Final structure of glass-pipet microelectrode. BME 301 Lecture Note 3 - Ali Işın 2013 58
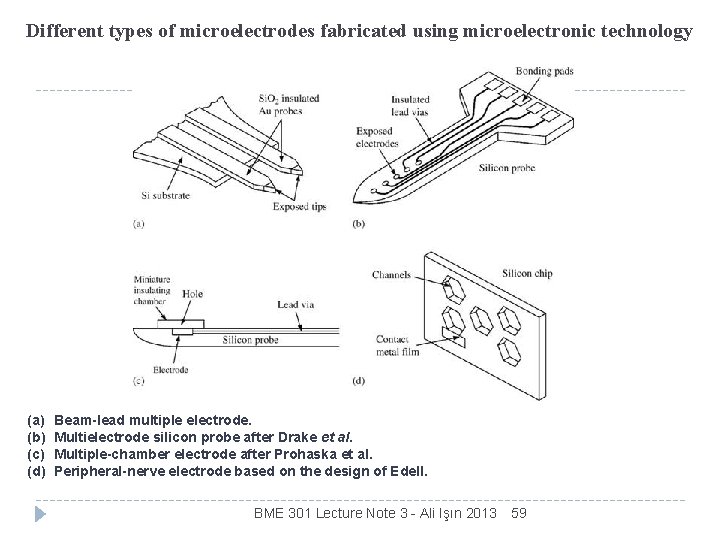
Different types of microelectrodes fabricated using microelectronic technology (a) (b) (c) (d) Beam-lead multiple electrode. Multielectrode silicon probe after Drake et al. Multiple-chamber electrode after Prohaska et al. Peripheral-nerve electrode based on the design of Edell. BME 301 Lecture Note 3 - Ali Işın 2013 59
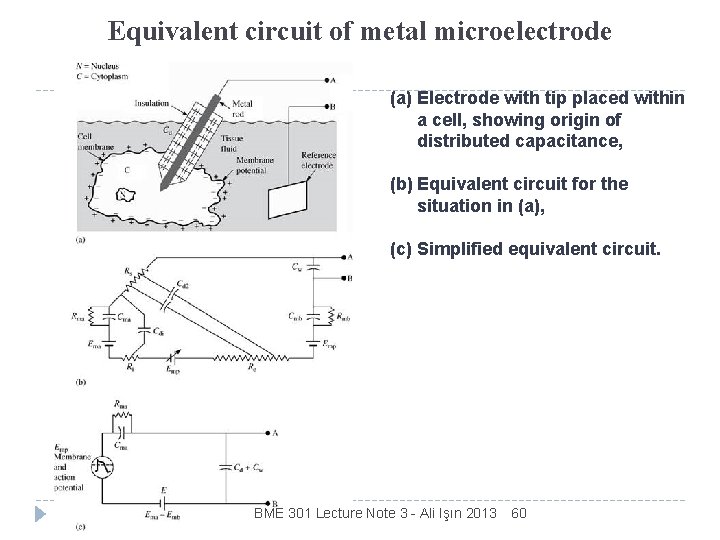
Equivalent circuit of metal microelectrode (a) Electrode with tip placed within a cell, showing origin of distributed capacitance, (b) Equivalent circuit for the situation in (a), (c) Simplified equivalent circuit. BME 301 Lecture Note 3 - Ali Işın 2013 60
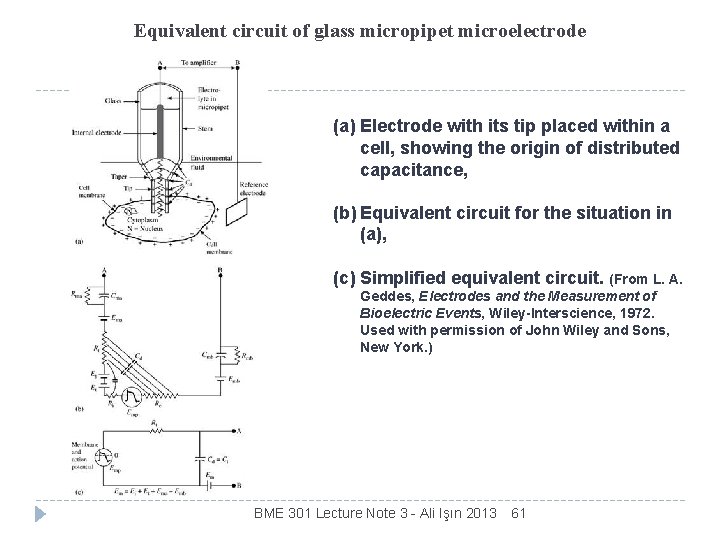
Equivalent circuit of glass micropipet microelectrode (a) Electrode with its tip placed within a cell, showing the origin of distributed capacitance, (b) Equivalent circuit for the situation in (a), (c) Simplified equivalent circuit. (From L. A. Geddes, Electrodes and the Measurement of Bioelectric Events, Wiley-Interscience, 1972. Used with permission of John Wiley and Sons, New York. ) BME 301 Lecture Note 3 - Ali Işın 2013 61
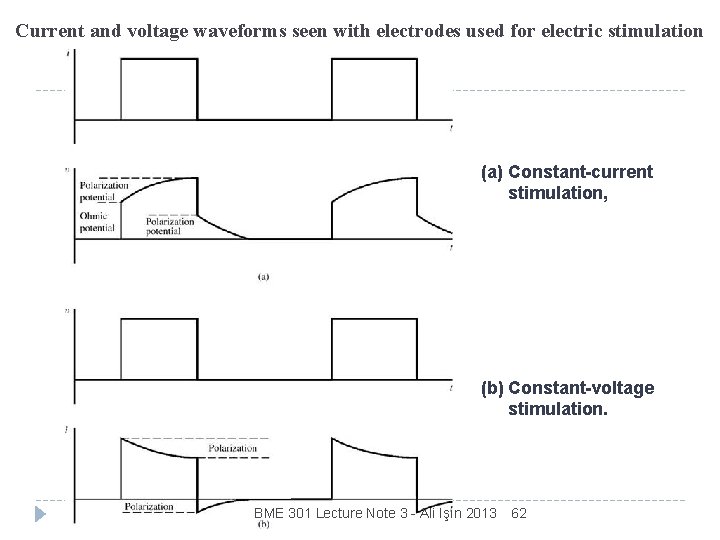
Current and voltage waveforms seen with electrodes used for electric stimulation (a) Constant-current stimulation, (b) Constant-voltage stimulation. BME 301 Lecture Note 3 - Ali Işın 2013 62
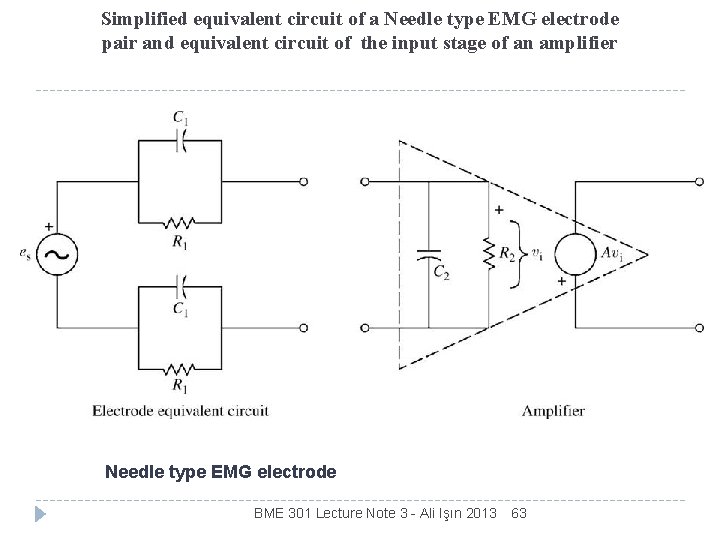
Simplified equivalent circuit of a Needle type EMG electrode pair and equivalent circuit of the input stage of an amplifier Needle type EMG electrode BME 301 Lecture Note 3 - Ali Işın 2013 63
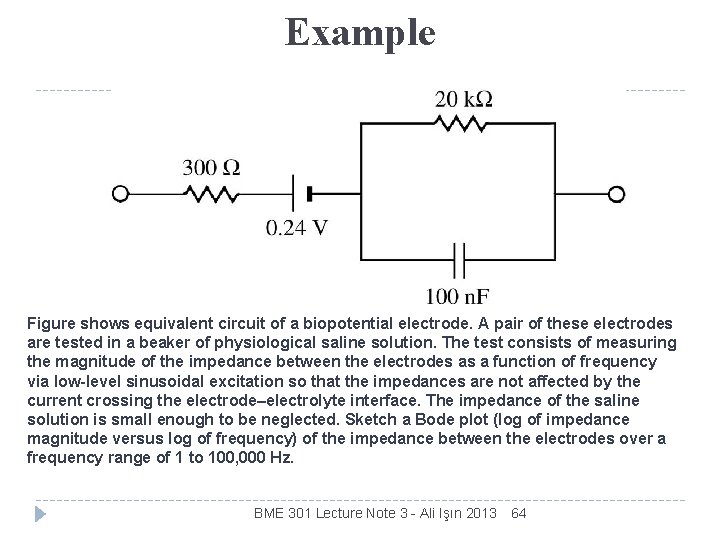
Example Figure shows equivalent circuit of a biopotential electrode. A pair of these electrodes are tested in a beaker of physiological saline solution. The test consists of measuring the magnitude of the impedance between the electrodes as a function of frequency via low-level sinusoidal excitation so that the impedances are not affected by the current crossing the electrode–electrolyte interface. The impedance of the saline solution is small enough to be neglected. Sketch a Bode plot (log of impedance magnitude versus log of frequency) of the impedance between the electrodes over a frequency range of 1 to 100, 000 Hz. BME 301 Lecture Note 3 - Ali Işın 2013 64
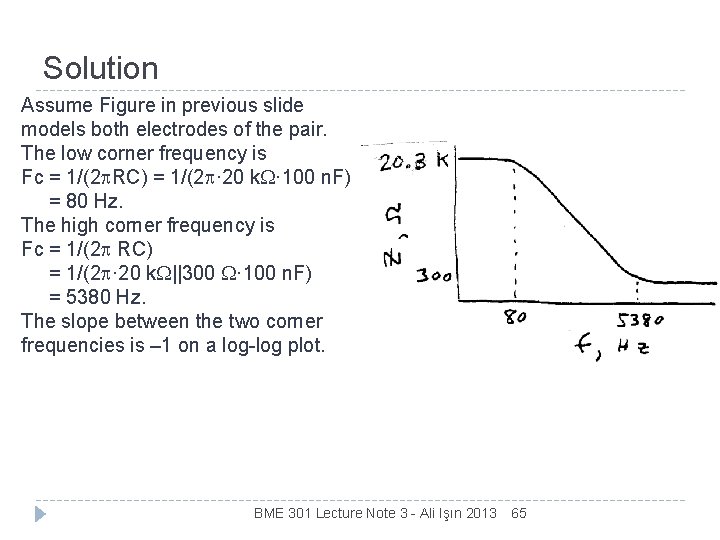
Solution Assume Figure in previous slide models both electrodes of the pair. The low corner frequency is Fc = 1/(2 RC) = 1/(2 · 20 k. W· 100 n. F) = 80 Hz. The high corner frequency is Fc = 1/(2 RC) = 1/(2 · 20 k. W||300 W· 100 n. F) = 5380 Hz. The slope between the two corner frequencies is – 1 on a log-log plot. BME 301 Lecture Note 3 - Ali Işın 2013 65

Example A pair of biopotential electrodes are implanted in an animal to measure the electrocardiogram for a radiotelemetry system. One must know the equivalent circuit for these electrodes in order to design the optimal input circuit for the telemetry system. Measurements made on the pair of electrodes have shown that the polarization capacitance for the pair is 200 n. F and that the half-cell potential for each electrode is 223 m. V. The magnitude of the impedance between the two electrodes was measured via sinusoidal excitation at several different frequencies. The results of this measurement are given in the accompanying table. On the basis of all of this information, draw an equivalent circuit for the electrode pair. State what each component in your circuit represents physically, and give its value. BME 301 Lecture Note 3 - Ali Işın 2013 66

Solution The 600 W is the tissue impedance plus the electrode/electrolyte highfrequency interface impedance. The 19400 W is the electrode/electrolyte low-frequency interface impedance. The 200 n. F is the electrode/electrolyte interface capacitance. The 223 m. V is the electrode/electrolyte polarization voltage. BME 301 Lecture Note 3 - Ali Işın 2013 67
- Slides: 67