BLOODDEVICE INTERACTIONS Thrombosis and Complement Thrombosis CARDIAC VALVES

BLOOD-DEVICE INTERACTIONS Thrombosis and Complement

Thrombosis CARDIAC VALVES AND VASCULAR GRAFTS
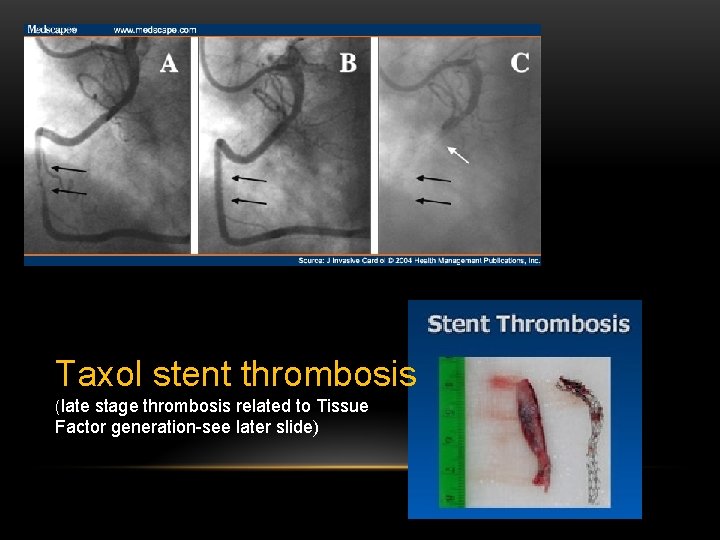
Taxol stent thrombosis (late stage thrombosis related to Tissue Factor generation-see later slide)

CLINICAL MANIFESTATIONS • Thrombotic events can range between 2% and 10% depending upon the device • Sudden and complete obstruction within weeks • Sub-acute thrombotic occlusion • Long-term “chronic” thrombosis • Embolism

THROMBOSIS-WHY? • Vascular grafts • Midgraft thrombosis • Lack of endothelium • Intimal narrowing at anastamosis • Surgical trauma • Variations in flow • Low flow-augmentation of anastomotic intimal hyperplasia • Shear stress • Diameter mismatches
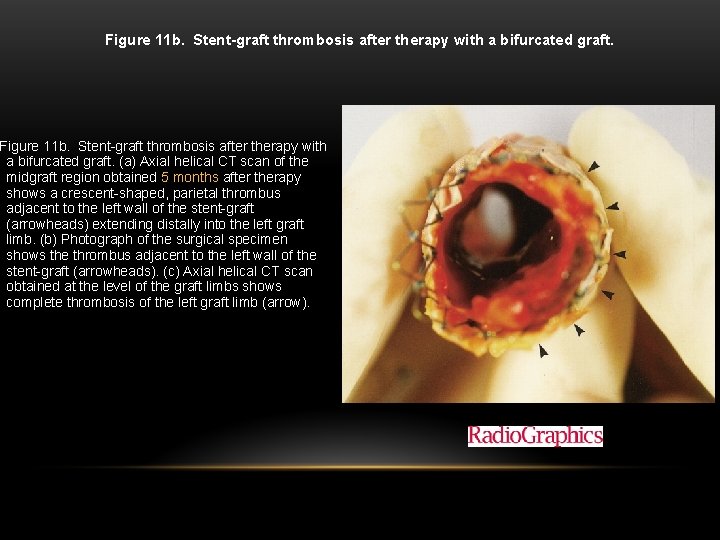
Figure 11 b. Stent-graft thrombosis after therapy with a bifurcated graft. (a) Axial helical CT scan of the midgraft region obtained 5 months after therapy shows a crescent-shaped, parietal thrombus adjacent to the left wall of the stent-graft (arrowheads) extending distally into the left graft limb. (b) Photograph of the surgical specimen shows the thrombus adjacent to the left wall of the stent-graft (arrowheads). (c) Axial helical CT scan obtained at the level of the graft limbs shows complete thrombosis of the left graft limb (arrow). Tillich M et al. Radiographics 1999; 19: 1573 -1583
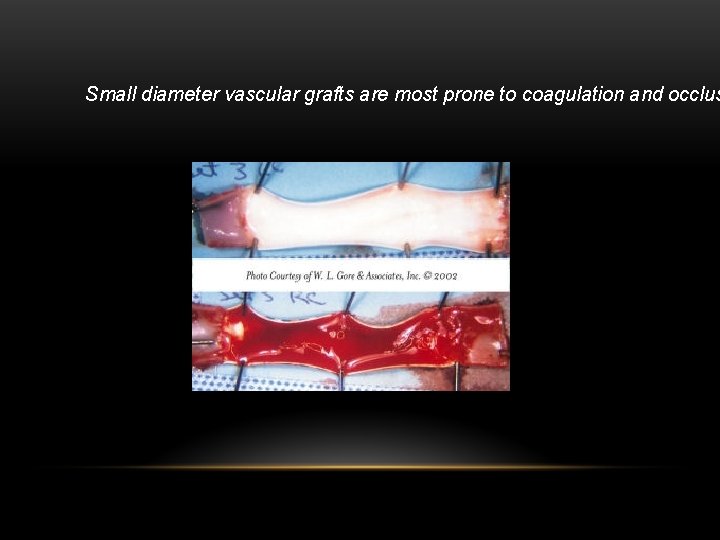
Small diameter vascular grafts are most prone to coagulation and occlus

THROMBOSIS-WHY? • Cardiac Valves • Intimal narrowing at anastamosis • Surgical trauma • Lack of endothelium • Variations in flow and turbulence • Shear stress • Diameter mismatches
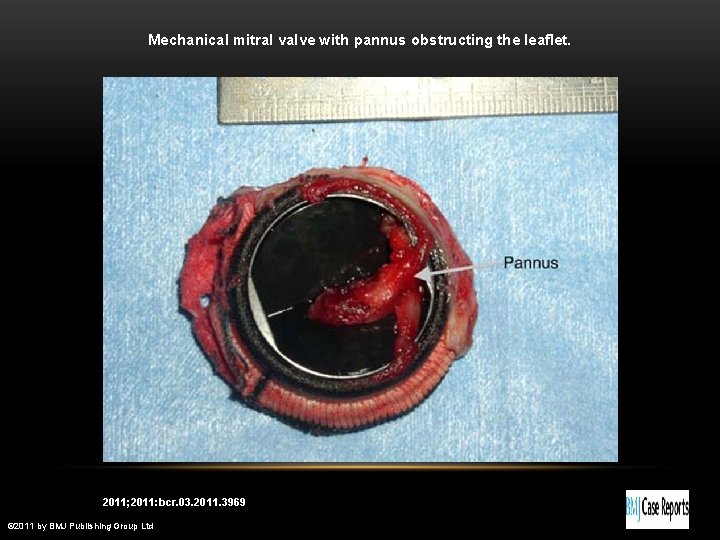
Mechanical mitral valve with pannus obstructing the leaflet. Imran U H et al. BMJ Case Reports 2011; 2011: bcr. 03. 2011. 3969 © 2011 by BMJ Publishing Group Ltd
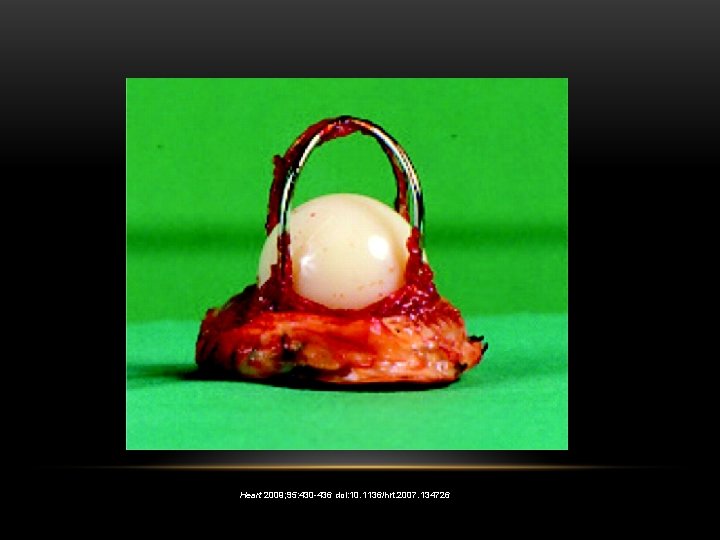
Heart 2009; 95: 430 -436 doi: 10. 1136/hrt. 2007. 134726

THE INITIAL STEPS… (PLATELET ACTIVATION) • Exposure of biomaterial surface or platelet trauma • Initiation of the clotting cascade automatically initiates wound healing (soft tissue lecture) • Binding of Factor XII • Activation of platelets and release of phospholipids and platelet factor 3 • Activation of Factor X • Culimination in common pathway • Fibrin clot formed from bound and circulating fibrinogen Contact phase proteins
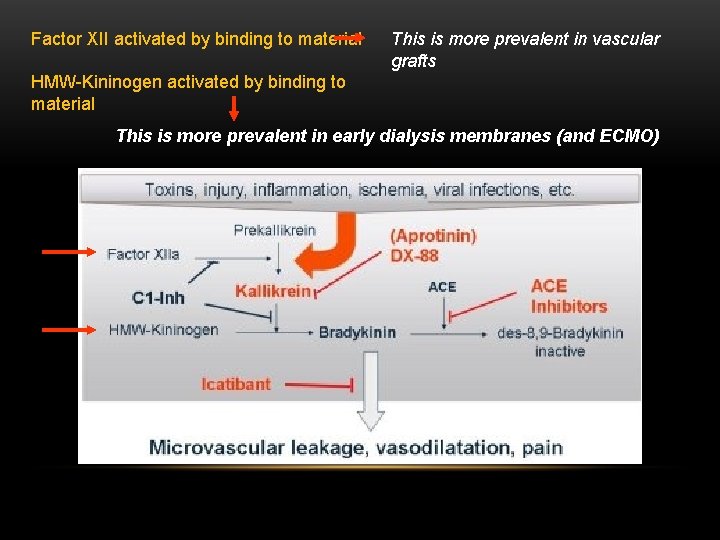
Factor XII activated by binding to material This is more prevalent in vascular grafts HMW-Kininogen activated by binding to material This is more prevalent in early dialysis membranes (and ECMO)
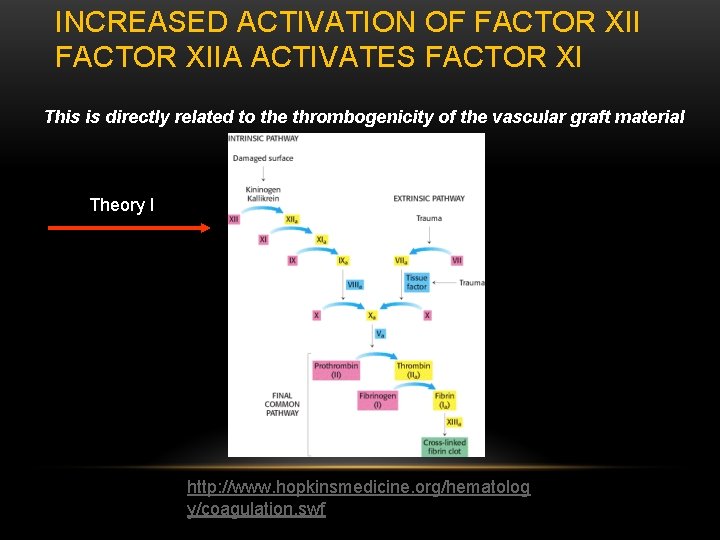
INCREASED ACTIVATION OF FACTOR XIIA ACTIVATES FACTOR XI This is directly related to the thrombogenicity of the vascular graft material Theory I http: //www. hopkinsmedicine. org/hematolog y/coagulation. swf
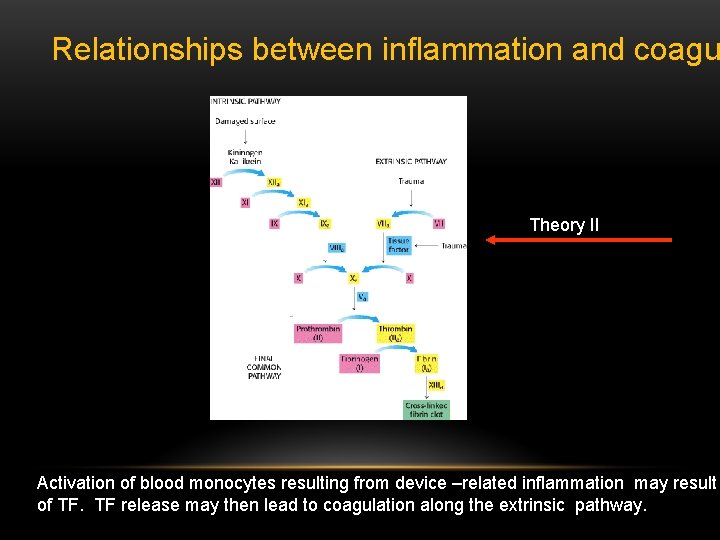
Relationships between inflammation and coagu Theory II Activation of blood monocytes resulting from device –related inflammation may result of TF. TF release may then lead to coagulation along the extrinsic pathway.

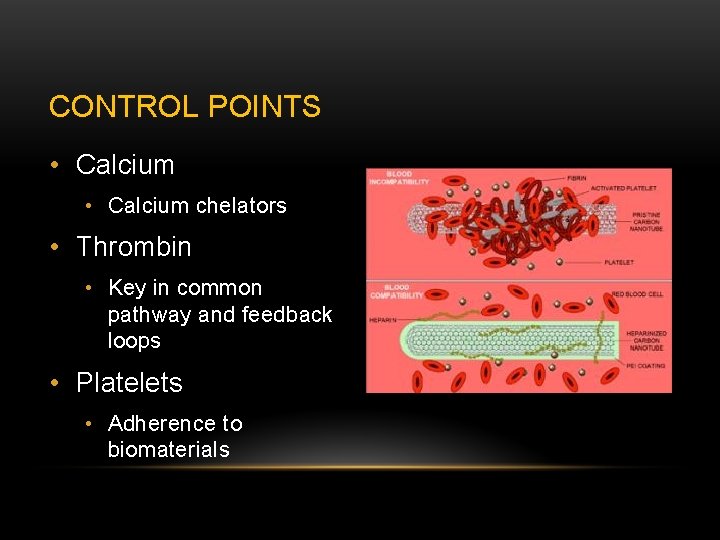
CONTROL POINTS • Calcium chelators • Thrombin • Key in common pathway and feedback loops • Platelets • Adherence to biomaterials
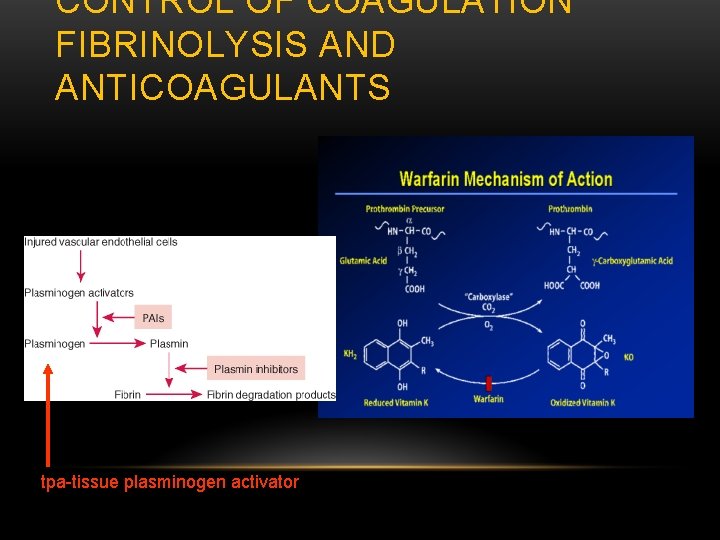
CONTROL OF COAGULATION FIBRINOLYSIS AND ANTICOAGULANTS tpa-tissue plasminogen activator

The Intersection Between Coagulation and Complement Activ

Complement Activation DIALYSIS MEMBRANES/ECMO
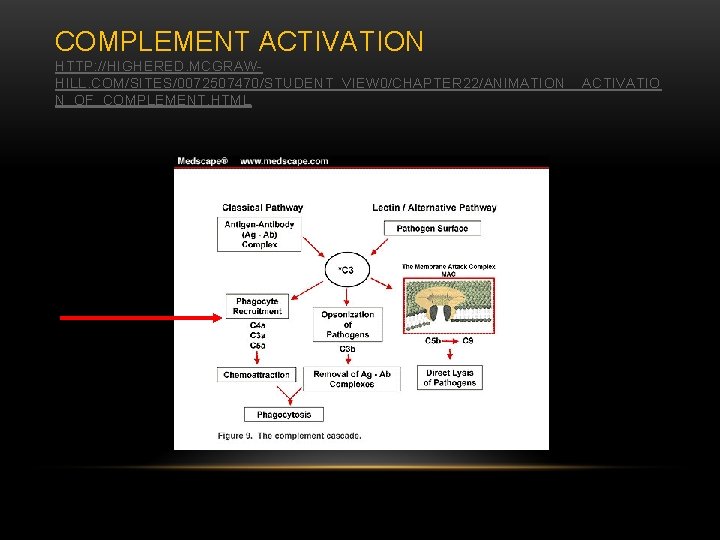
COMPLEMENT ACTIVATION HTTP: //HIGHERED. MCGRAWHILL. COM/SITES/0072507470/STUDENT_VIEW 0/CHAPTER 22/ANIMATION__ACTIVATIO N_OF_COMPLEMENT. HTML
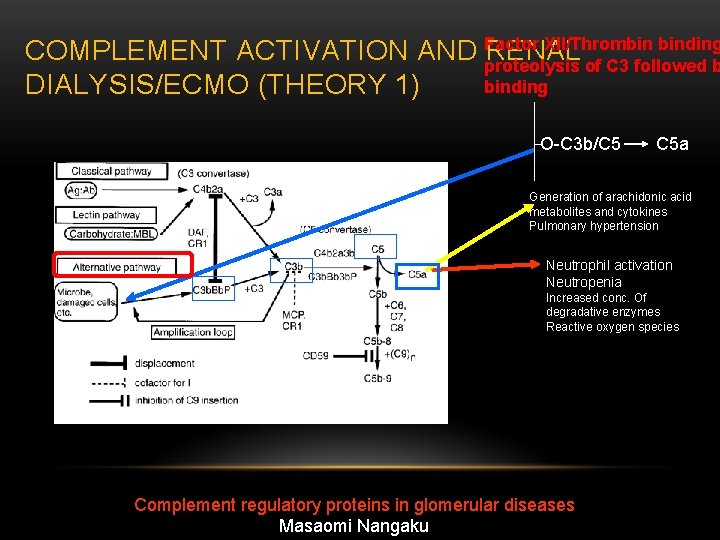
XII/Thrombin binding COMPLEMENT ACTIVATION AND Factor RENAL proteolysis of C 3 followed b binding DIALYSIS/ECMO (THEORY 1) O-C 3 b/C 5 C 5 a Generation of arachidonic acid metabolites and cytokines Pulmonary hypertension Neutrophil activation Neutropenia Increased conc. Of degradative enzymes Reactive oxygen species Complement regulatory proteins in glomerular diseases Masaomi Nangaku
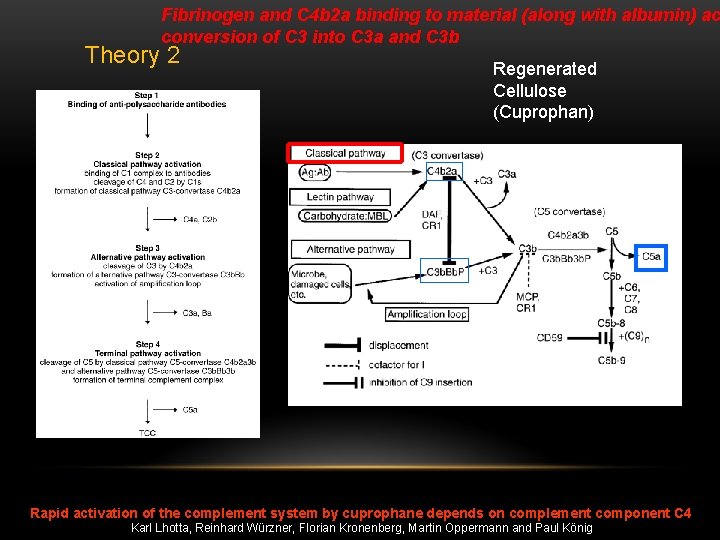
Fibrinogen and C 4 b 2 a binding to material (along with albumin) ac conversion of C 3 into C 3 a and C 3 b Theory 2 Regenerated Cellulose (Cuprophan) Rapid activation of the complement system by cuprophane depends on complement component C 4 Karl Lhotta, Reinhard Würzner, Florian Kronenberg, Martin Oppermann and Paul König

Thrombosis and Complement: Crosstalk
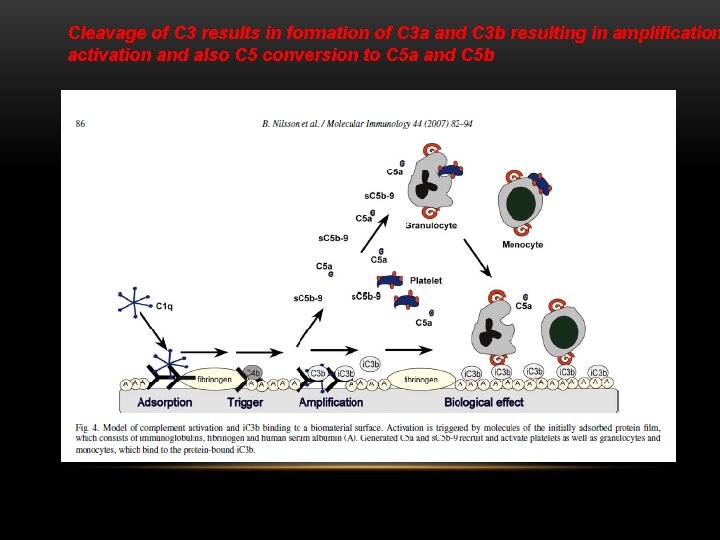
Cleavage of C 3 results in formation of C 3 a and C 3 b resulting in amplification activation and also C 5 conversion to C 5 a and C 5 b
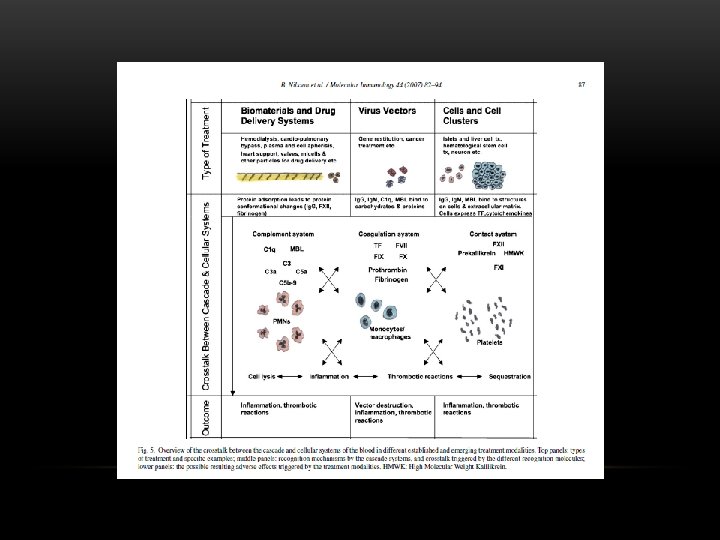
THE HOST’S RESPONSE • Protein deposition on membrane • Boundary layer/secondary layer • Cellular Activation • Platelet/leukocyte aggregates • Immune stimulation • Complement • Hypersensitivity reactions • Residual ETO • Hemodynamic Effects • Contact phase formation of bradykinin

PROTEIN DEPOSITION • Deposition of protein occurs instantaneously • A protein layer forms on the surface of the membrane as the levels of solution phase proteins increase • Composition of adsorbed proteins depends upon membrane type • Hydrophobic membranes tend to adsorb more proteins • Proteins adsorb and then detach until permanent adsorption and denaturing occurs • This may result in the formation of a boundary layer • Protein adsorption determines cellular responses • Protein adsorption may serve as a way to successfully remove unwanted proteins from patients or may negatively effect the diffusivity of the membrane • Low MW-interleukins, inflammatory cytokines • High MW-albumin, fibrinogen, Ig. G

PLATELET/LEUKOCYTE AGGREGATES • Platelets aggregate on membrane surface resulting in further aggregation of leukocytes • Aggregation causes activation of the platelets as well as the leukocytes and can result in release of platelet-derived factors and cytokines • Results: superoxide release, cytokine release and leukopenia • Superoxide release resulting from chronic dialysis has been implicated in atherosclerosis • Cytokine release and leukopenia can result in general feeling of malaise UPenn

HYPERSENSITIVITY AND HEMODYNAMICS “First use syndrome”-Inflammation and Hypersensitivity Toxins-ETO (sterilization) residuals Plasticizers-Membrane mfg Leachables-Membrane mfg Post-perfusion syndrome (neucleophiles) Contact phase activation Bradykinin is formed due to contact with the membrane Bradykinin system activated by Factor XII (clotting cascade) Vasodilation Anaphylaxis
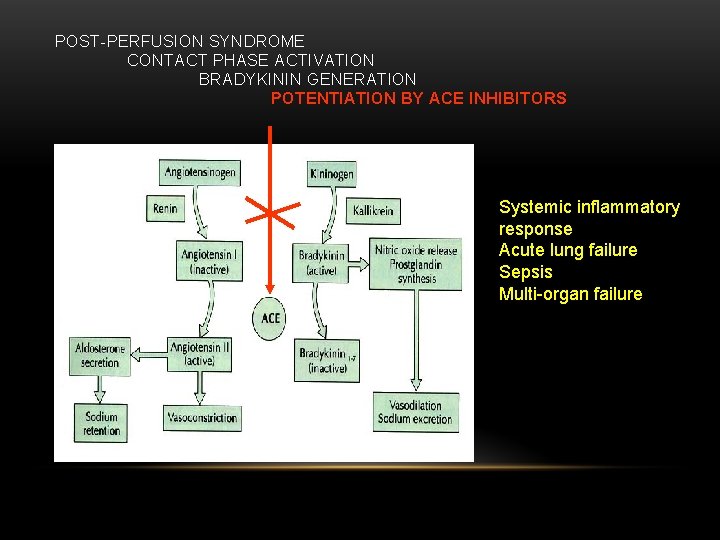
POST-PERFUSION SYNDROME CONTACT PHASE ACTIVATION BRADYKININ GENERATION POTENTIATION BY ACE INHIBITORS Systemic inflammatory response Acute lung failure Sepsis Multi-organ failure
- Slides: 30