Blood sample processing for serum total cholesterol HDL
Blood sample processing for serum total cholesterol, HDL cholesterol and plasma glucose, Hb. A 1 c and DNA extraction

Based on EHES Manual, Part B. Fieldwork Procedures, 2 nd edition (2016) Available at: http: //urn. fi/URN: ISBN: 978 -952 -302 -701 -5 These slides can be used freely, translated and adapted to national use (e. g. concerning the equipment). However, it is important to keep in mind that no changes should be made to the measurement techniques, which need to be standardized.

Equipment • Centrifuge • Empty tubes for recentrifugation (only needed if gel serum tubes are used) • Storage tubes • Pipette • Storage boxes • Disposable gloves
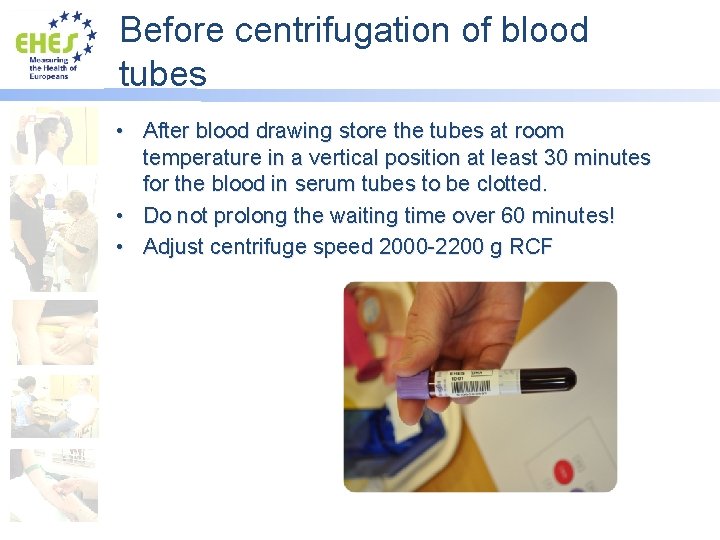
Before centrifugation of blood tubes • After blood drawing store the tubes at room temperature in a vertical position at least 30 minutes for the blood in serum tubes to be clotted. • Do not prolong the waiting time over 60 minutes! • Adjust centrifuge speed 2000 -2200 g RCF
Centrifugation of blood tubes (1/2) After the waiting period, centrifuge The serum tube l The fluoride-citrate tube l The 1 st EDTA tube l Do not centrifuge the last two EDTA tubes designated for Hb. A 1 c and DNA extraction!

Centrifugation of blood tubes (2/2) • Place the serum tube and the fluoride-citrate and the 1 st EDTA plasma tube in the centrifuge • Check that all tubes are resting on the bottom of the centrifuge rack • Check that the centrifuge rotor and tubes are in balance. • Centrifuge the tubes 2000 g for 10 minutes at room temperature
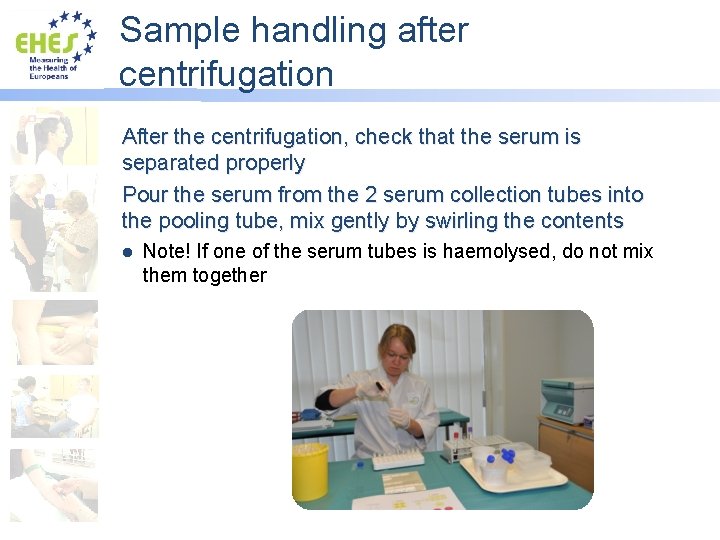
Sample handling after centrifugation After the centrifugation, check that the serum is separated properly Pour the serum from the 2 serum collection tubes into the pooling tube, mix gently by swirling the contents l Note! If one of the serum tubes is haemolysed, do not mix them together

Labelling storage tubes • Place the storage tubes in a tube rack according to the pipetting scheme • No need to label all tubes if blood collection is incomplete • Place the bar code labels in an upright direction, so that the scale marks remain visible
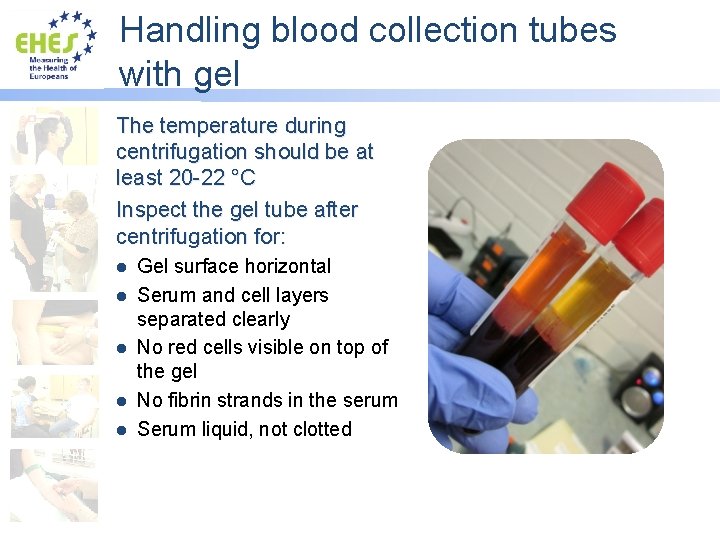
Handling blood collection tubes with gel The temperature during centrifugation should be at least 20 -22 °C Inspect the gel tube after centrifugation for: l l l Gel surface horizontal Serum and cell layers separated clearly No red cells visible on top of the gel No fibrin strands in the serum Serum liquid, not clotted

Hemolysed samples • Do not pool a hemolysed serum sample with non-hemolysed samples • Several hemolysed samples can be pooled • Document hemolysed aliquots • Use the colour guide to document the degree of hemolysis

Pipette aliquots into storage tubes • 2 x 1, 5 ml serum from the pooling serum tube into the 3 ml plastic tube and 1, 5 ml aliquots into the 1, 5 ml storage cryotubes • 2 x 1 ml from fluoride-citrate plasma tube into two 1, 5 ml cryotubes • 2 x 1, 5 ml from the EDTA tube into two 1, 5 ml cryotubes • Close the cryotubes with caps and freeze the tubes at once

Transferring storage tubes into freezers Place the tubes without delay into the boxes in the freezer Label boxes before placing them into the freezer because labels will not stick on a wet/cold surface

Sample shipment To the national HES laboratory – In dry ice – Packed in leak proof secondary packaging To the EHES Reference Laboratory (if possible) – Packed according to the IATA regulations, according to Pecking Instruction 650 for diagnostic specimens – Place one tube upside down in every box

Recording • The amount of the collected serum and plasma • Visible attributes (haemolysis, lipemia, icterius) • Deviations from the sample processing protocols, if any • Your initials/personnel code to identify who handled the samples

Acknowledgements Slides prepared by: Laura Råman Photographs: Hanna Tolonen Sample processing demonstrations by Saara Vallivaara Experiences and feedback from the EHES network have been utilized in the preparation of these slides Funding: Preparation of the slides is part of the activities of the EHES Coordinating Centre which has received funding from the EC/DG SANTÉ in 2009 -2012 through SANCO/2008/C 2/02 -SI 2. 538318 EHES and Grand Agreement number 2009 -23 -01, and in 2015 -2017 through Grand Agreement number 664691/BRIDGE Health

Disclaimer The views expressed here are those of the authors and they do not represent the Commission’s official position.
- Slides: 16