Blood Oxygen physically diffused by 0 2 ml


Blood • • Oxygen physically diffused by 0. 2 ml / 100 ml blood By Hb 20 ml / 100 ml blood So it’s the main function.
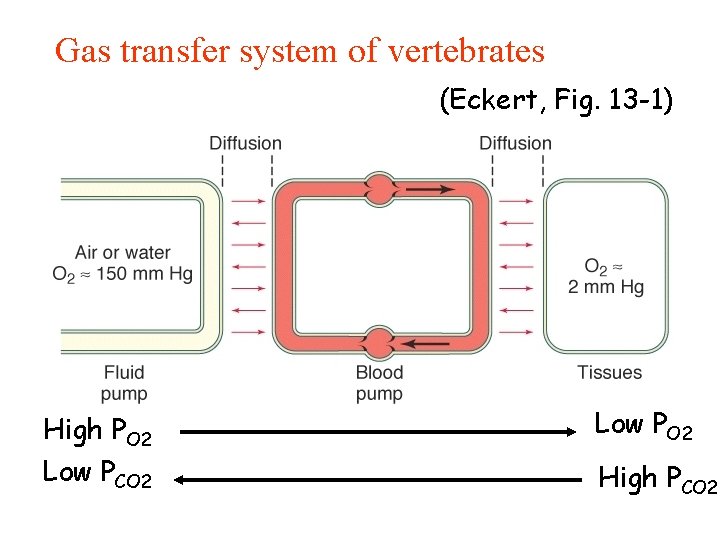
Gas transfer system of vertebrates (Eckert, Fig. 13 -1) High PO 2 Low PCO 2 Low PO 2 High PCO 2

Main functions of blood • * transport : • *Oxygen and carbon dioxide » » » - nutrients Hormones Metabolites Force ( hydraulic ) Temperature • Immunity • Coagulation

Transport of O 2 in the blood q. Respiratory pigments § After O 2 diffuses across the respiratory epithelium it is bound by a respiratory pigment § Respiratory pigments are complexes of proteins and metal ions

• • 1. hemoglobin 2. chlorocruorin 3. hemerythrin 4. hemocyanin • 5. myoglobin Which in solution heavy, In cells lighter

Why Hb inside the cell • VISCOSITY • FIGURE 2, 1 • CHEMISTRY O 2 BINDING

Chlorocruorin • • Class Polychaeta contain Fe green in low oxygen, red when saturated In solution

Hemocyanin • some , molluscs , crustaceans and arachnids • blue when oxygenated and colorless when deoxygenated • contain Cu • No porphyrin ring • In solution

Hemerythrin • Present in the blood of certain marine invertebrates (e. g. sipunculoid worms, polychaetes) • Contains iron but not a porphyrin ring • violet-pink when oxygenated, colorless when deoxygenated • In cells

Hemoglobin (Hb) § Hemoglobin (Hb) • Increases the carrying capacity of blood for O 2 • Four protein (globin) chains, each attached to an iron -containing heme group • Each heme group consists of a porphyrin ring with an iron atom in the center • In humans, there are forms of globin: , , in adults (2 , 2 ) • Form dimers 1 1 2 2 • each Subunits bonded to other by links ( hydrogen , …)

• Fe bind to the 4 N with coordinate covalent bond ( the bond e pair is formally contributed by only of the atoms forming the bonds) • The fifth is his residue from globin • The sixth is oxygen if exist • To give oxyhemoglobin • Each hb can carry 4 o • Subunit interactions altered when Hb binds to O 2 or to other molecules
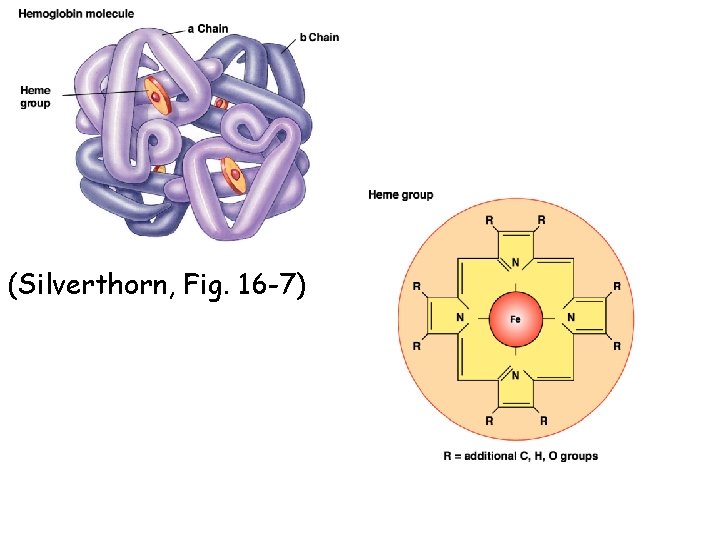
(Silverthorn, Fig. 16 -7)


Hemoglobin • Hemoglobin loads up with oxygen in the lungs, forming oxyhemoglobin. • As blood passes through the capillaries, some of the oxyhemoglobin releases oxygen and become deoxyhemoglobin. - Carboxyhemoglobin: Hb saturated with CO

(Hemoglobin (Hb • Partially saturated hemoglobin – when one to three hemes are bound to oxygen • The rate that hemoglobin binds and releases oxygen is regulated by: – PO 2, temperature, blood p. H, PCO 2, and the concentration of BPG (an organic chemical) • These factors ensure adequate delivery of oxygen to tissue cells

Hemoglobin • • Fetal hemoglobin: gamma chains (not β) w/ higher affinity for O (enhance O transfer from mother to fetus) Affinity for CO = 200 x’s greater than for O

• ( No, CO, H 2 S) poisoning even at low partial pressures Oxidation of Fe yields 3+ charge - ferric iron -methemyoglobin ( Met Hb) does NOT bind oxygen. ON THE SIXTH PART AWATER MOLECULE

• Normally Fe may be oxidized ( few) • So what : • RBCs have a methemoglobin reductase , which convert small amount to – Hb normal.

Myoglobin • • Mb is a monomeric heme protein a store oxygen protein facilitate diffusion - found in skeletal & cardiac muscle - contain one heme binds one O 2 Globin: - single polypeptide chain; 154 hemoglobin a. a. ; 17. 2 k. D (monomer) 141= α - 8 helicies (label A-H) 20 Β= 146

Myoglobin • • Surface: polar; inside: non polar • • Fe in Mb is Fe 2+ - ferrous iron - the form that binds oxygen • • Oxygen binds as the sixth ligand to Fe • Higher affinity for O 2 than Hb

Myoglobin • Oxidation of Fe yields 3+ charge - ferric iron -metmyoglobin ( Met. Mb) does NOT bind oxygen • Cardiac muscle of diving mammals contain higher (8%) amounts of Myoglobin than most mammals (e. g. 0. 5% for a dog)

Who's haven't: Antarctic Icefish body temperature: -1. 7 o. C

Who's haven't Antarctic ice fish lack pigment • low metabolic needs = low metabolism • high cardiac output, blood volume • large heart • O 2 solubility 0. 3% • Blood has lower viscosity – Easier to pump

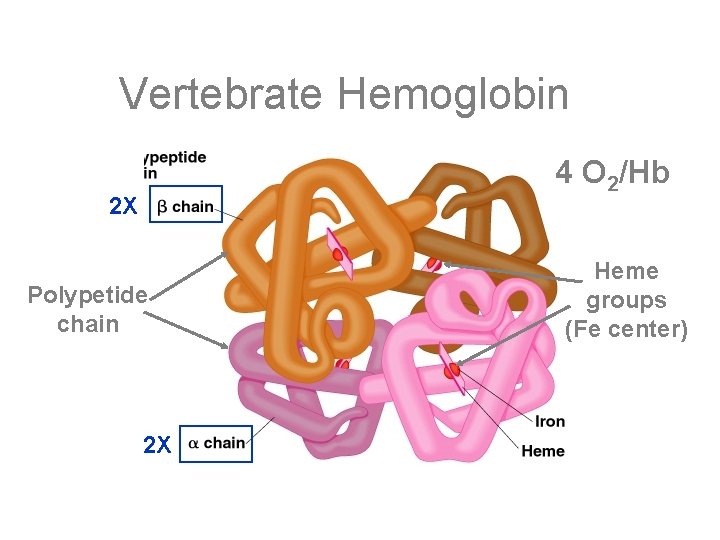
Vertebrate Hemoglobin 4 O 2/Hb 2 X Polypetide chain 2 X Heme groups (Fe center)

How do respiratory pigments ? work • Reversibly Bind O 2: heme group • Hb + O 2 « Hb. O 2 • To work effectively Hb should: 1. Bind O 2 at high PO 2 (lung, eqn. shifts right) 2. Release O 2 at low PO 2 (working tissue, eqn. shifts left)

• Oxygen binding changes the Hb conformation • – Without oxygen bound, Fe is out of heme plane • – Oxygen binding pulls the Fe into the heme plane • Fe pulls its His F 8 ligand along with it • The F helix moves when oxygen binds • This change means little to Mb, • but lots to Hb!


Mechanism of allosteric change • Heme iron move <= 0. 04 nm induces • conformation change in Hb • • One alpha-beta pair moves relative to the other by 15 degrees upon oxygen binding • • This massive change is induced by movement of Fe by 0. 039 nm when oxygen binds

• Oxy and deoxy form represent 2 different • conformation states • - Hb binds O 2 (from T R)


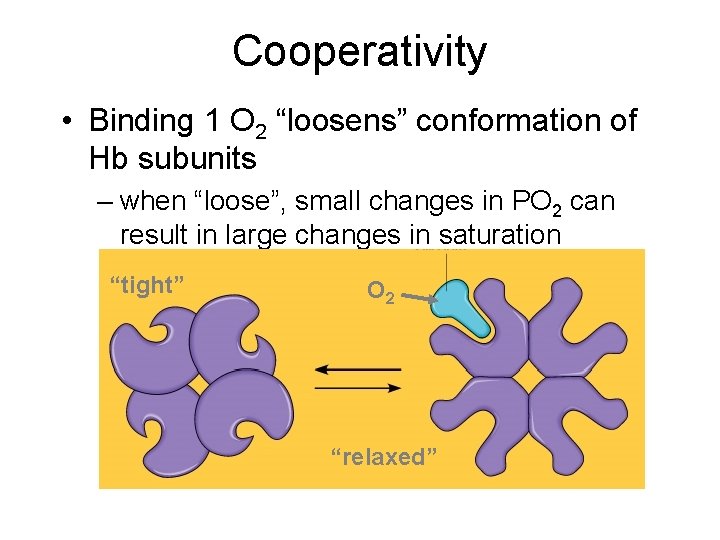
Cooperativity • Binding 1 O 2 “loosens” conformation of Hb subunits – when “loose”, small changes in PO 2 can result in large changes in saturation “tight” O 2 “relaxed”
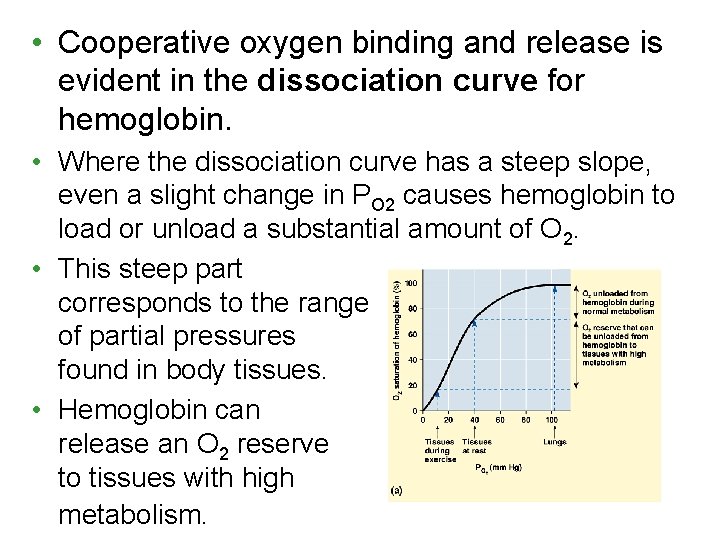
• Cooperative oxygen binding and release is evident in the dissociation curve for hemoglobin. • Where the dissociation curve has a steep slope, even a slight change in PO 2 causes hemoglobin to load or unload a substantial amount of O 2. • This steep part corresponds to the range of partial pressures found in body tissues. • Hemoglobin can release an O 2 reserve to tissues with high metabolism.

Influence of PO 2 on Hemoglobin Saturation • Hemoglobin saturation plotted against PO 2 produces a oxygen-hemoglobin dissociation curve • 98% saturated arterial blood contains 20 ml oxygen per 100 ml blood (20 vol %) • As arterial blood flows through capillaries, 5 ml oxygen are released
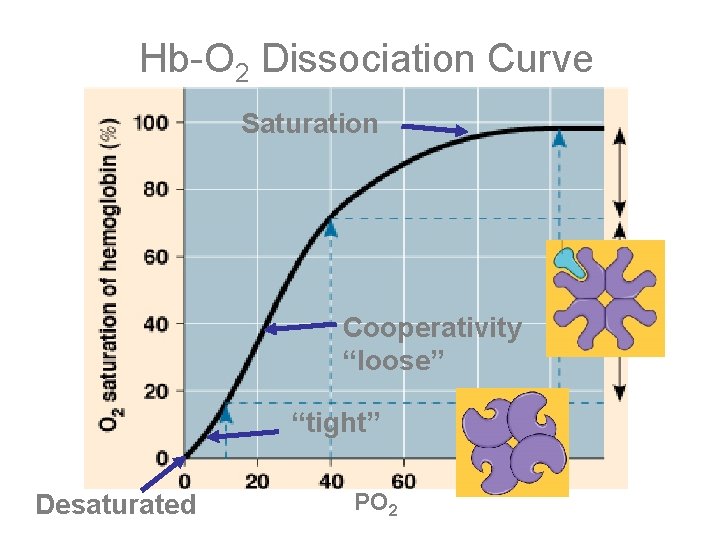
Hb-O 2 Dissociation Curve Saturation Cooperativity “loose” “tight” Desaturated PO 2
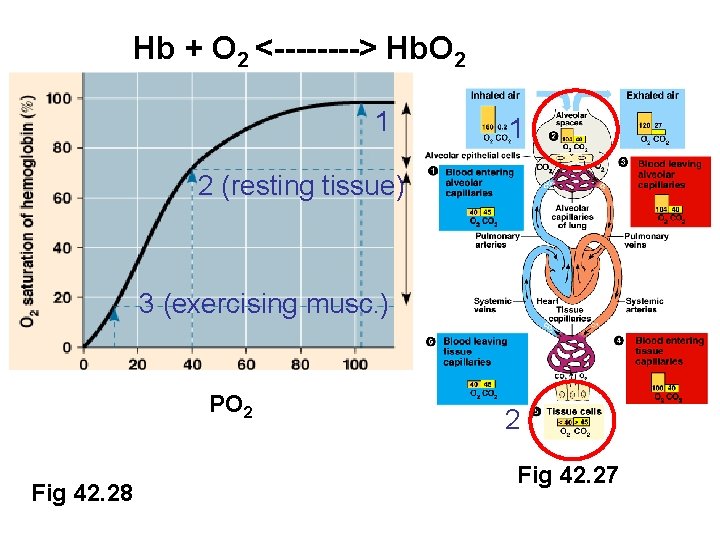
Hb + O 2 <----> Hb. O 2 1 1 2 (resting tissue) 3 (exercising musc. ) PO 2 Fig 42. 28 2 Fig 42. 27

Hemoglobin Saturation Curve • Hemoglobin is almost completely saturated at a PO 2 of 70 mm Hg • Further increases in PO 2 produce only small increases in oxygen binding • Oxygen loading and delivery to tissue is adequate when PO 2 is below normal levels

Hemoglobin Saturation Curve • Only 20– 25% of bound oxygen is unloaded during one systemic circulation • If oxygen levels in tissues drop: – More oxygen dissociates from hemoglobin and is used by cells – Respiratory rate or cardiac output need not increase
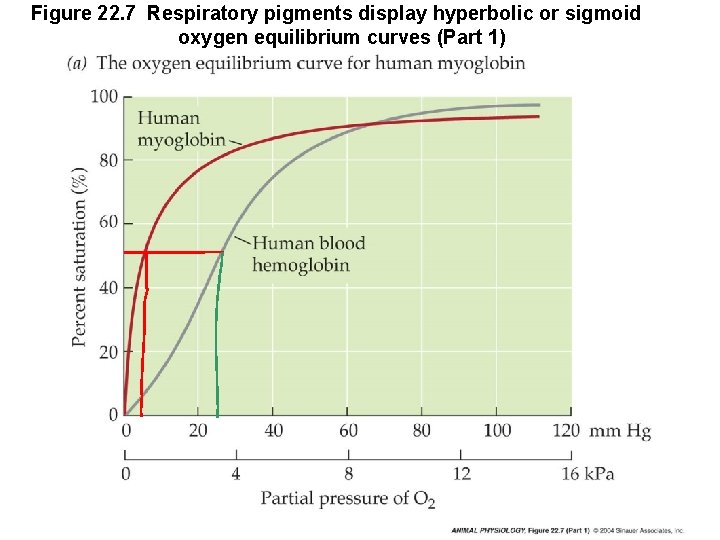
Figure 22. 7 Respiratory pigments display hyperbolic or sigmoid oxygen equilibrium curves (Part 1)

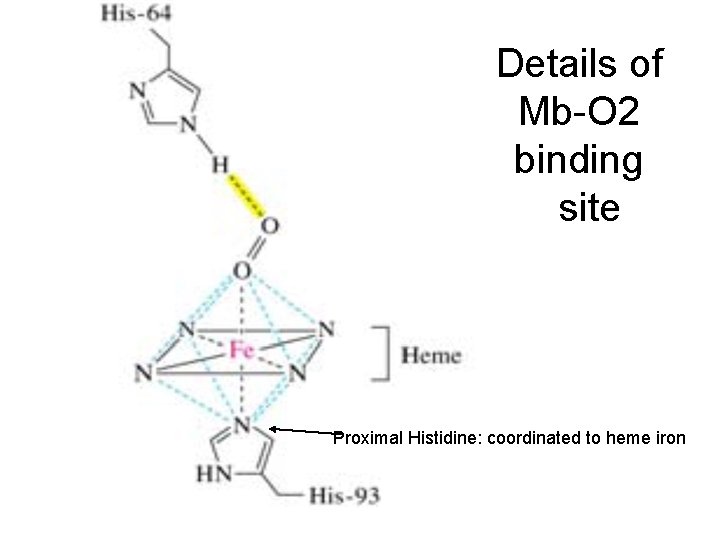
Details of Mb-O 2 binding site Proximal Histidine: coordinated to heme iron
- Slides: 43