Blood gas Workshop VTA 2019 Acid A substance

Blood gas Workshop VTA 2019

Acid • A substance that dissociates in H 20 H+ • HCl H+ + Cl- (complete dissociation) • Acetic acid H+ + Acetate- Base • Substance that accepts a H+ Buffer • Mixture of a weak acid and its conjugate base

Bicarbonate buffer system (ECF) • H 2 C 03 / HC 03 • Strong acid : HC 03 - + H+ • Strong Base: H 2 C 03 + OH- H 2 C 03 H 20 • p. H – measure of [H+ ] or Log 1/ [H+ ] • H/H equation relates the p. H of a buffer to the concentration of its buffer acid and base p. H = p. Ka + log [base] / [acid]
![• p. H = p. Ka + log [HC 03 -] / [H • p. H = p. Ka + log [HC 03 -] / [H](http://slidetodoc.com/presentation_image_h/2122002ca29e22f816a2fa4762b4548d/image-4.jpg)
• p. H = p. Ka + log [HC 03 -] / [H 2 C 03] • p. H = 6. 1 + log 20/1 = 7. 4 As long as the ratio 20: 1 is kept, irrespective of concentrations, p. H is normal • H 2 C 03 H 20 + C 02 oversimplification • H 2 C 03 p. C 02 p. H [HC 03 -] /p. C 02
![Components of acid base status • p. H [HC 03 -] /p. C 02 Components of acid base status • p. H [HC 03 -] /p. C 02](http://slidetodoc.com/presentation_image_h/2122002ca29e22f816a2fa4762b4548d/image-5.jpg)
Components of acid base status • p. H [HC 03 -] /p. C 02 Non Respiratory component • If disease causes changes in one component, the other component responds in an attempt to correct the ratio of 20: 1 • See if you can work out the direction of the compensatory component should one component change

Equations • • Metabolic acidosis Metabolic alkalosis Respiratory acidosis Respiratory alkalosis Delta ratio – Increase in AG / Decrease in HC 03 AG calculated- (12) / 24 - (HC 03 measured ) Interpretation <0. 4 – Normal AG Met Acid 0. 4 – 08 – Mixed Normal AG , High Metabolic Met Acid 1 -2 - High AG met Acid >2 - HC 03 is high to start with

Case 1 p. H 7. 08 Pa. CO 2 18, 8 mm. Hg Pa. O 2 100 mm. Hg HC 03 5, 4 Na 136 mmol/l Cl 102 mmol/l Fi. O 2 0. 3

Case 2 p. H 7. 48 Pa. CO 2 47 mm. Hg Pa. O 2 68 mm. Hg HC 03 34 Fi. O 2 0. 4 K = 2, 8

Metabolic Alkalosis • High p. H • HC 03 - excess • Compensation? p. H [HC 03 -] /p. C 02

Causes of Metabolic alkalosis • H+ loss – GIT: • Vommiting / NG suction – Renal: • • Diuretics Mineralocorticoid excess Low PTH/ Post chronic hypercapnea – Hypokalemia • Transcellular shift/ Loss

• HCO 3 excess – Na. HCO 3 admin – Massive transfusion • Contraction alkalosis
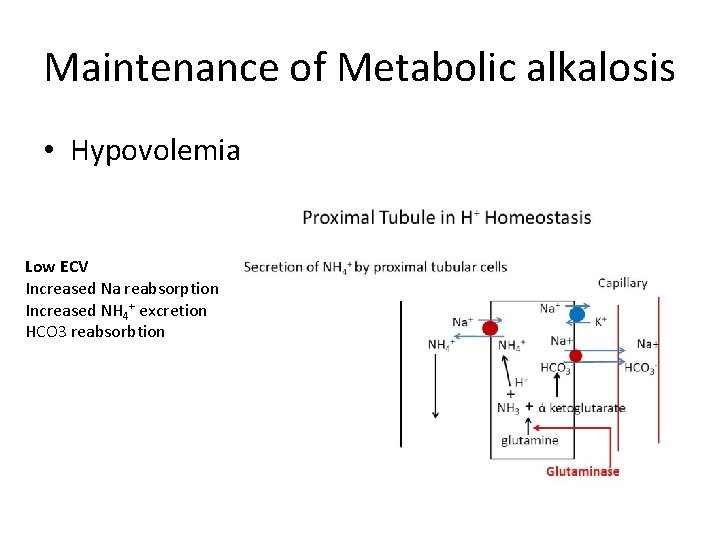
Maintenance of Metabolic alkalosis • Hypovolemia Low ECV Increased Na reabsorption Increased NH 4+ excretion HCO 3 reabsorbtion
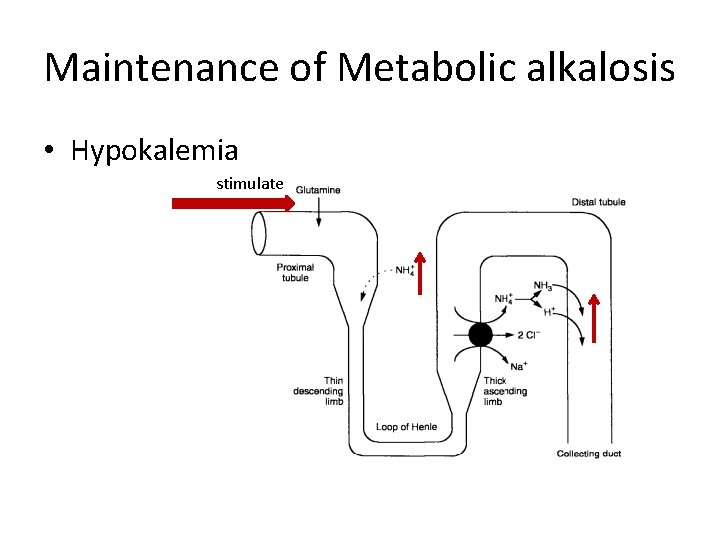
Maintenance of Metabolic alkalosis • Hypokalemia stimulate
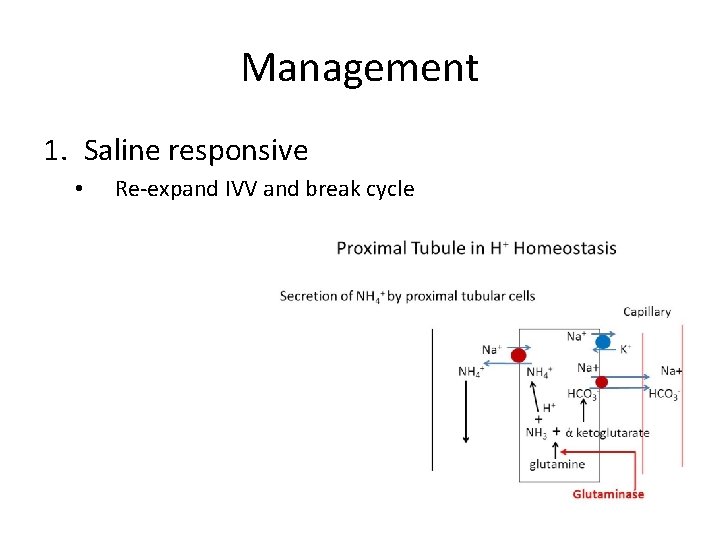
Management 1. Saline responsive • Re-expand IVV and break cycle
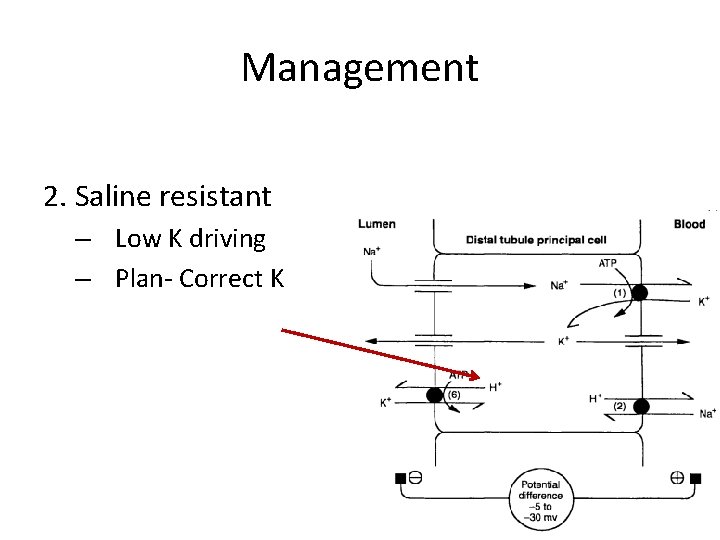
Management 2. Saline resistant – Low K driving – Plan- Correct K

Case 3 p. H 7. 50 Pa. CO 2 41 mm. Hg Pa. O 2 145 mm. Hg HC 03 34 Fi. O 2 0. 4 K = 2, 8

Causes: 1. vomiting in pregnant patient, 2. diuretics or vomiting in patient with chronic respiratory alkalosis typical of cirrhosis, 3. postcardiac arrest (hyperventilation, bicarbonate therapy and conversion of lactate to bicarbonate

Case 4 • • • Elderly COPD p. H 7. 55 Pa. CO 2 53 mm. Hg Pa. O 2 63 mm. Hg Bicarbonate 48 mmol/l Fi. O 2 0. 21

Alveolar arterial gradient Hypoxemia Pa. O 2 < 80 mm. Hg Due to imbalance between: Pulmonary ventilation and Pulmonary capillary blood flow Alveolar O 2 = Fi. O 2 x( Atm Pressure – H 2 O) – Pa. CO 2/RQ Decrease if : Pa. CO 2 increases Pa. CO 2 = Pa. CO 2 production/ RR x (Vt-Vd)

Case 5 • • • p. H 7. 44 Pa. CO 2 63 mm. Hg Pa. O 2 52 mm. Hg Bicarbonate 42 mmol/l Fi. O 2 0. 28

A-a. DO 2 • A-a DO 2 in hypoxemia 1. High: • Diffusion/shunt/dead space 2. Normal: Altitude/Fi 02 low or Pa. CO 2 high Alveolar O 2 = Fi. O 2 x( Atm Pressure – H 2 O) – Pa. CO 2/RQ • Resp depressants/ sedatives/ms relaxants/NM ds/Skeletal abnormalities

Paeds BASIC ICU Course • Email: • paedsbasicjhb@gmail. com
- Slides: 22